‘What Housewife Isn’t On Ozempic?’ How A Weight-Loss Craze Is Sweeping Across America
Stars and producers of Bravo’s hit franchise are amplifying a debate about whether new drugs that show promise for weight loss are being prescribed too liberally. ‘What Housewife Isn’t On Ozempic?’ How A Weight-Loss Craze Is Sweeping Across America
Is The Weight-Loss Drug Revolution Causing A Frailty Epidemic?
Weight-loss drugs, particularly GLP-1 agonists like Ozempic and Wegovy, can lead to a loss of muscle mass alongside fat loss.
Studies indicate that approximately 20-35% of the weight lost may come from lean tissue, including muscle.
Impact Of Weight-Loss Drugs On Muscle Mass
Weight-loss medications, especially GLP-1 agonists like Ozempic and Wegovy, are effective for reducing body weight.
However, they can also lead to a loss of muscle mass alongside fat loss.
Muscle Loss Statistics
Research indicates that when using these medications, approximately 20-35% of the total weight lost may come from lean tissue, which includes muscle.
This muscle loss can have significant implications for overall health and metabolism.
Reasons For Muscle Loss
Several Factors Contribute To Muscle Loss During Weight Reduction With GLP-1 Drugs:
Reduced Appetite: These medications suppress appetite, which can lead to lower protein intake.
Since protein is crucial for muscle maintenance, inadequate consumption can result in muscle breakdown.
Rapid Weight Loss: Losing weight too quickly, especially without strength training, can lead to significant muscle mass loss.
The body may break down muscle tissue for energy along with fat.
Decreased Physical Activity: Some individuals may experience fatigue while on these medications, leading to reduced physical activity, which can accelerate muscle atrophy.
Health Implications
The Loss Of Muscle Mass Is Not Just An Aesthetic Concern; It Can Lead To:
Slower Metabolism: Muscle is metabolically active, and losing muscle can lower the metabolic rate, making it harder to maintain weight loss.
Decreased Strength And Mobility: Muscle atrophy can result in weakness and reduced functional ability, increasing the risk of falls, especially in older adults.
By implementing these strategies, individuals can help preserve muscle mass while benefiting from the weight-loss effects of GLP-1 medications.
The Impact Of Weight-Loss Drugs On Your Muscles Vs Your Fatty Tissue
Weight-loss drugs, particularly GLP-1 receptor agonists like Ozempic, can lead to a significant reduction in both fat and muscle mass during weight loss.
Research indicates that while these medications effectively reduce fat, they may also cause a loss of lean body mass, which includes muscle, potentially affecting strength and metabolism.
Overview of Weight-Loss Drugs
Weight-loss drugs, particularly GLP-1 receptor agonists like Ozempic and Wegovy, are designed to help individuals manage obesity and type 2 diabetes.
These medications work by reducing appetite and improving blood sugar control, leading to significant weight loss.
Muscle vs. Fat Loss
Muscle Loss
Lean Mass Reduction: Weight loss from GLP-1 medications often includes a reduction in lean body mass, which can account for 15-40% of total weight loss.
Muscle Preservation: New combination therapies, such as bimagrumab with semaglutide, show promise in enhancing fat loss while preserving muscle mass.
This approach aims to minimize the loss of muscle during weight reduction.
Strength Concerns: Some studies indicate that while muscle mass may not decrease significantly, muscle strength can still decline.
This is particularly concerning for older adults, who are at higher risk for muscle loss and reduced mobility.
Fat Loss
Fat Mass Reduction: The majority of weight lost with GLP-1 medications is from fat tissue. For instance, studies show that patients can lose up to 60% fat and 40% muscle mass during treatment.
Body Composition Changes: GLP-1 therapies preferentially reduce visceral fat, which is harmful to metabolic health, while also promoting a healthier body composition overall.
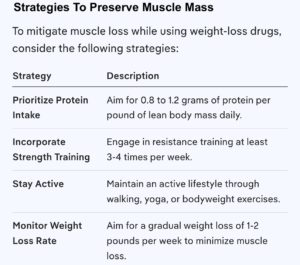
Strategies To Minimize Muscle Loss
To Mitigate Muscle Loss While Using Weight-Loss Drugs, Consider The Following:
Increase Protein Intake: Aim for 0.8 to 1.2 grams of protein per pound of lean body mass daily to support muscle maintenance.
Engage In Strength Training: Incorporate resistance exercises at least 3-4 times a week to help preserve muscle mass.
Stay Active: Regular physical activity, including walking and other forms of exercise, can help maintain muscle and overall health during weight loss.
Understanding the balance between fat and muscle loss is crucial for optimizing health outcomes while using weight-loss medications.
“You’re looking thin,” Andy Cohen told “Real Housewives of New Jersey” star Dolores Catania on a recent episode of his late-night Bravo talk show.
Mr. Cohen was talking about Ozempic, a Type 2 diabetes drug that is part of a group of injections changing weight loss as we know it. Ms. Catania said that yes, she was taking the drug, but clarified in a segment of the show posted online that it was actually Mounjaro, a competitor.
“That’s the amped-up version,” said Marysol Patton, a “Real Housewives of Miami” cast member who was seated beside Ms. Catania. “That’s the fast-forward to Skinnyville.”
Though these drugs are relatively new, reality TV stars speak about them with familiarity and frequency, as if they were unofficial characters in Bravo’s “Real Housewives” universe. Mr. Cohen, the franchise’s executive producer, first helped establish the connection in September, when he tweeted to his 2.4 million followers, “Everyone is suddenly showing up 25 pounds lighter.
What happens when they stop taking #Ozempic ?????” In the “Watch What Happens Live With Andy Cohen” segment with Ms. Catania, Mr. Cohen stoked the flames again: “What Housewife isn’t on Ozempic?” he said. “Not one,” Ms. Catania replied. “Half your cast,” Mr. Cohen said.
Every week, another star from the show’s orbit speaks out about GLP-1s—a class of drugs that includes Novo Nordisk’s Ozempic and Wegovy, as well as Eli Lilly’s Mounjaro. Mentions need no introductions from producers, podcasters or posters in online forums.
While the Bravo juggernaut has been known for candid discussions about sensitive topics like plastic surgery and eating disorders, the spotlight on these injections is amplifying a debate about whether they are being prescribed too liberally.
(Wegovy is approved by the FDA to treat obesity. Ozempic and Mounjaro are not, though providers may prescribe them off-label for weight loss.)
Bravo stars who speak openly about using the drugs say they’re doing what they were hired to do: Be real. While there has been speculation about widespread Hollywood use, reality TV stars are among the rare celebrities who have been upfront about weight loss resulting from GLP-1s.
“I think Housewives on the whole are very honest. So, everybody weaponizes everything they do against them,” said Margaret Josephs, a Real Housewife of New Jersey, who has been taking compounded semaglutide—the active ingredient in Ozempic and Wegovy—since last spring, in addition to peptides.
“It wasn’t about, ‘Oh I want to be thin.’ I really want to feel good and looking great was a bonus.” Still, her weight loss has drawn tabloid coverage. “I really have no shame in it.”
Fans and the celebrity press have made a sport of speculating on who might be taking the drugs, and some stars have faced backlash as a result. Several Housewives have denied use, with some offering up alternative explanations. Some fans aren’t convinced.
“Apparently a lot of people think I’m taking Ozempic,” Kyle Richards of “The Real Housewives of Beverly Hills” said on a January podcast hosted by two women of “Housewives” fame. “
To clarify, I’ve never taken Ozempic, never tried Ozempic, and I’m not taking the other one that starts with an ‘M.’” She said she’d had a breast reduction, curbed sugar and carbs, cut out alcohol, in addition to working out.
On a recent episode of “The Real Housewives of Miami,” which streams on Peacock, Ms. Patton and her co-star Alexia Nepola visit a “fat doctor” for a “fat-burning shot” that is meant to be taken weekly, just like Ozempic and its cousin drugs.
The name of the clinic is prominently displayed on the show, and its website lists GLP-1 injections among its offerings. “Am I tripping or did Marysol introduce us to her Ozempic prescribing doctor?” one person tweeted.
In a statement, Ms. Nepola said, “We affectionately call him the ‘Fat Doctor’ because that’s what he’s known for in Miami… We were there to get a vitamin shot.” Ms. Patton has denied taking Ozempic and could not be reached for comment. The clinic did not return requests for comment.
“I just think everyone should just be honest about what they’re doing because I think it sends the wrong message to say ‘Look at me, this is diet and exercise,’” said David Yontef, whose daily podcast “Behind the Velvet Rope” dissects reality shows including the “Real Housewives” franchise.
He clips segments of the show for YouTube with headlines like: “Why Won’t These Specific Real Housewives Admit They Are on Ozempic?” Mr. Yontef, who is taking a GLP-1 himself, speaks candidly on his show about his own experience.
He said that the drugs are rumored to have come up during filming of the coming season of “The Real Housewives of Beverly Hills,” which is under way. A representative for the show declined to comment.
Ms. Catania, who was frank about using Mounjaro on “Watch What Happens Live,” walked back her words in a subsequent interview, as did her doctor, Rocio Salas-Whalen.
“I have no control of what she’s going to say on live TV,” said Dr. Salas-Whalen, an endocrinologist and obesity physician. “There’s a reason behind why the patient is on the medication. Without going into details about her health, the way that she presented it to the public, that is different.”
Mr. Cohen’s exchange with Ms. Catania received some backlash, which he acknowledged on his Sirius XM radio show. He didn’t want to glorify thinness, he said, but he felt it was important to stay true to the show’s candor around bodies.
“This was, I think, one of the first shows that showed people getting Botox and talking about their breast enhancements and all that stuff,” he said on-air. “I think that there are millions of women at home who look at these women and want to know, ‘What did they have done?’”
“I’m trying to deliver the goods, though, for my audience who are watching,” said Mr. Cohen, who had several listeners call in to share their perspectives on the topic. “If Dolores shows up 15 pounds lighter, I gotta find out how it happened.” Through a representative, Mr. Cohen declined to comment.
Dr. Reshmi Srinath, medical director of Mount Sinai Hospital’s weight and metabolism management program, said that popular culture can help increase awareness about health treatments.
But a quick mention in a TV show can also have an adverse effect, leading viewers to believe a pharmaceutical approved to treat specific diseases is a “magic drug.”
“We have to keep in mind what the data shows, and that there are some patients that don’t respond at all,” Dr. Srinath said.
She also cited side effects such as gastrointestinal issues, ranging from upset stomach and nausea to severe reflux and gallstones, and stressed the importance of adequate nutrition. According to experts, the drugs may need to be taken long-term to keep weight off.
Gossip about the drugs has become so rampant that moderators of a “Real Housewives” Reddit forum banned speculative “Ozempic” discussion last month.
(If a Housewife has commented on use, it’s fine to talk about that.) Dozens of posts a day are flagged by users for mentioning Ozempic, according to a moderator.
Jackie Goldschneider of “The Real Housewives of New Jersey” said she first learned about Ozempic in the fall, after the show’s current season had stopped filming. “All of a sudden I feel like out of nowhere, I started seeing it and hearing about it everywhere.”
She is in recovery for anorexia and said it was jarring to see her peers eating such reduced portions. (The drugs have an appetite-curbing effect.)
“To go out to dinner and be the only person really eating—that, for me, was very difficult,” she said, noting that she confronted the issue but received pushback.
“I don’t think anyone wants to be judged for their decisions,” she said. “And I get that, because for a very long time, I had the most unhealthy relationship with food and I never wanted to be judged for it.”
Ms. Goldschneider knows the focus on reality stars comes with the territory of being public figures. She sees them as a microcosm of a conversation that is taking place around the world, as people looking to lose weight clamor for Ozempic, Wegovy and Mounjaro prescriptions.
“It’s so much bigger than the ‘Housewives’ franchise,” she said.
Updated: 3-13-2023
European Medicines Agency Warns of Diabetics’ Risk Amid Ozempic Shortage
* Some Doctors Have Been Prescribing Ozempic For Weight Loss
* Use As Slimming Treatment May Endanger Diabetics, EMA says
The increasing use of diabetes drug Ozempic as a weight-loss treatment prompted the European Medicines Agency to urge doctors to prioritize the injection made by Novo Nordisk A/S for diabetics, who may be at risk if they don’t get it.
Demand for Ozempic has surged because it’s the same drug as Novo Nordisk’s Wegovy weight-loss treatment, but at a different dose.
Doctors have been prescribing Ozempic off-label to patients desperate to try the most recent weight-loss treatment. While Ozempic is sold across Europe, Wegovy is yet to launch in most of the region and has also faced shortages in the US.
“Increased demand for Ozempic has led to shortages, which are expected to continue throughout 2023,” the EMA said Monday.
“Ozempic is only indicated for the treatment of adults with insufficiently controlled type 2 diabetes mellitus,” and any other use may endanger those patients’ health.
Diabetes patients who may run out of Ozempic should be switched to an alternative to avoid hyperglycemic reactions, the EMA said.
Wegovy is the first new obesity medicine to reach the market in years and has been found to help overweight people lose about 15% of their total body weight as it dampens the appetite.
Demand for Wegovy has been spurred by use by Hollywood actors as well as Elon Musk. Sales for the weight-loss drug quadrupled to 6.2 billion Danish kroner ($892 million) last year, helping turn Novo Nordisk into Europe’s second-biggest company by market value.
Updated: 4-3-2023
The ‘King Kong’ of Weight-Loss Drugs Is Coming
Eli Lilly’s Mounjaro could outpace Ozempic as the most powerful treatment on the market. To develop it, the drug company needed to overhaul long-held but failing practices.
People who are overweight are flocking to the drug Ozempic to slim down. Looming is an even more powerful weight-loss treatment.
The drug Mounjaro helped a typical person with obesity who weighed 230 pounds lose up to 50 pounds during a test period of nearly 17 months.
No anti-obesity drug has ever safely made such a difference. In the coming months, it is widely expected to get the go-ahead from U.S. health regulators to be prescribed for losing weight and keeping it off, and some patients are already using it unapproved for that purpose.
The advance of Mounjaro, which is already on the market to treat Type 2 diabetes, has excited doctors and patients who have been waiting decades for effective treatments, while helping turn its maker, Eli Lilly & Co., into the most valuable standalone pharmaceutical company in the U.S. with a market value of more than $300 billion.
It’s a product of Lilly’s recent, sometimes painful overhaul of how it develops drugs. After several costly drug failures, Lilly abandoned some of its long-held practices, including waiting for multiple committees to weigh in before advancing a drug.
The company had also been prioritizing its existing successful drug franchises at all costs, sometimes at the expense of promising new treatments.
That now discarded approach would have stifled the development of Mounjaro. Some people inside Lilly discouraged pursuing the drug in the mid-2010s because it might compete with a Lilly product that was already selling well.
Overriding these concerns, Lilly pushed its labs to move fast, pursue ambitious projects and worry less about the business ramifications, even if that would mean cannibalizing sales of high-selling products with years of lucrative patent protection left. Lilly scientists were able to chase Mounjaro, and they worked quickly.
“Every program we do, we look at what our competitors have done, who’s done it the fastest, and then we set a goal to go even faster,” said Daniel Skovronsky, Lilly’s chief scientific and medical officer.
“Speed becomes our No. 1 incentive, which is hard because it’s a cultural change.”
Since it shifted its approach, in stages over the past decade, Lilly’s overall R&D output has been among the industry’s most prodigious.
The company has had new prescription drugs approved in the U.S. or other countries since 2014 for conditions such as cancer, migraines and Covid-19. It has cut their development timelines to an average of six years from 11.
The revamp has produced medicines that could make big differences in diseases that have long frustrated researchers and debilitated patients.
Mounjaro helped people who have difficulty losing pounds despite dieting cut their weight by up to 22.5% over 72 weeks during testing.
In comparison, Ozempic and its sister drug, Wegovy, made by Novo Nordisk AS, which share the same active ingredient, induced weight loss of up to around 17% in studies.
Also up for approval from Lilly is an experimental Alzheimer’s drug that slowed the disease’s progress in a key study. The experimental Alzheimer’s drug, if approved, could reach $12 billion in yearly sales, according to analysts.
Mounjaro could be one of the highest-selling drugs of all time with annual sales exceeding $25 billion. Novo’s Ozempic and Wegovy brought in close to $10 billion last year, with prescriptions rapidly growing.
‘We gotta get out of this’
Lilly, founded in 1876 in Indianapolis, was the first drug company to sell insulin and distribute the polio vaccine globally.
Starting in the 1980s, it became known for groundbreaking psychiatric drugs including the antidepressant Prozac.
By the early 2010s, however, the company’s labs were striking out. Experimental drugs for heart disease, schizophrenia, depression and Alzheimer’s failed in large, expensive clinical trials.
Big-selling products such as the antipsychotic Zyprexa began facing competition from lower-cost generics. Company shares sank.
“Oh man, we gotta get out of this,” John Lechleiter, then Lilly’s chief executive, recalled thinking on a walk home from work in 2009 after the stock hit a decades low.
To innovate, Lilly would need to let go of its single-minded focus on protecting its existing lucrative drug franchises, maximizing their sales until patents ran out and then chasing further sales with new products that weren’t all that different from the ones they replaced.
The company also needed to move faster. One internal committee after another second-guessed every recommendation to advance a promising drug candidate. “The decisions got revisited every step of the way,” recalled J. Anthony Ware, who led product development at Lilly before retiring in 2017.
The committees were intended to ensure thorough vetting, but in practice became a limiting process that squeezed out bold ideas, according to Dr. Skovronsky.
Dr. Skovronsky, who joined Lilly after it acquired his brain-imaging firm in 2010, was accustomed to moving quickly because money was tight at the startup. Lilly lacked the same urgency, Dr. Skovronsky said, and the slowness made it miss out on huge opportunities.
Lilly’s scientists, for instance, were among the first to see potential for a new type of breast cancer drug targeting proteins known as CDK4 and CDK6 that play a role in tumor growth.
It took them too long to get internal funding for clinical trials, however, handing competitors Novartis AG and Pfizer Inc. the advantage of bringing their therapies to market first. Lilly’s drug, Verzenio, was approved by the FDA in 2017, after Pfizer’s Ibrance in 2015 and Novartis’s Kisqali earlier in 2017.
Lilly also missed out on cancer immunotherapies, ceding the treatments to rivals such as Merck & Co. and Bristol-Myers Squibb Co. Their drugs have saved many skin and lung cancer patients and are now among the industry’s biggest sellers.
Dr. Skovronsky was frustrated with Lilly’s slow pace. “Let me understand this,” he recalled saying at a committee meeting setting timetables for getting experimental drugs to market.
“Our goal is to be slower than average, and we’re failing at that goal? This can’t be the way to do things.”
In 2015, Lilly’s board of directors asked Dr. Skovronsky, then senior vice president of clinical and product development, to help analyze Lilly’s research flops over the prior 10 years and figure out how to do R&D better.
A big reason for the failures, Dr. Skovronsky found, was that Lilly’s business-unit heads, focusing on sales potential, were making decisions about which drugs to promote to late-stage studies.
The result: The company advanced into the large, expensive studies candidates that had mixed results in earlier testing. Dr. Skovronsky found that drugs that had earlier mixed results often failed the later studies.
The business-unit officials overplayed “what would really be great for sales representatives and underplayed what would be great science and great for patients,” he said.
Dr. Skovronsky recommended Lilly pursue drug projects where it best understood the science, and lean less on commercial sales estimates.
Lilly was not very good at predicting a drug’s sales over time anyway, he concluded, but could better predict the scientific probability of a drug’s success.
Lilly jettisoned research on diseases where it was tougher to deliver an advance, including osteoporosis and psychiatric conditions, and doubled down in areas where it had expertise: diabetes, oncology and Alzheimer’s disease.
“We had to hunker down in a lot of ways to free up resources for other priorities,” said Chief Executive David Ricks, who led Lilly divisions including its China and U.S. businesses during this period, before becoming CEO in 2017.
Lilly also tried to become more open to outsiders to help bring in fresh ideas. In 2018, it promoted Dr. Skovronsky to the chief scientific officer role. When Lilly acquired cancer-drug developer Loxo Oncology in 2019, it put Loxo’s leaders in charge of Lilly’s cancer research.
Along with its successes, Lilly has had setbacks, including pulling a new cancer drug from the market in 2019 after a study found it wasn’t helping patients.
Last year, U.S. drug regulators rejected a proposed new cancer therapy co-developed by Lilly and a Chinese biotech company because of concerns about the medicine’s testing in China.
Then, in January, the U.S. Food and Drug Administration hit the brakes on speedily approving Lilly’s experimental Alzheimer’s therapy, saying it would wait for more study data before making a decision. Still, these types of setbacks are happening less frequently than before.
‘King Kong’
The new obesity drug grew out of long-running efforts at Lilly to promote the body’s production of insulin, the hormone used to control blood-sugar levels. Lack of insulin or insufficient insulin are hallmarks of diabetes.
In 2014, Lilly introduced a drug that helped people release more insulin when they eat. The drug, named Trulicity, did that by mimicking a hormone in the gut called GLP-1 that naturally mobilized the release of insulin. Scientists also found it suppressed appetite and made people feel full when they eat.
Patients with the most common form of diabetes needed to inject Trulicity once a week, not every day like older medicines. And not only did the drug significantly reduce blood sugar levels, it helped patients lose some weight.
Doctors and patients began flocking to the new drug. Analysts projected it would be a big seller for Lilly, perhaps reaching $2 billion in annual sales. And the company could look forward to patents protecting those hefty sales for years.
When Lilly scientists proposed, in 2014, pursuing a drug that promised to lower blood sugar more than Trulicity and cut weight by even larger amounts, company leaders hesitated.
“It was controversial among senior colleagues at Lilly,” recalled David Moller, a former company head of diabetes research. “There were those who thought Trulicity was the best we could do.”
Lilly scientists expressed hope their drug candidate could do much more than that. The experimental drug combined a synthesized GLP-1 gut hormone like the one in Trulicity with a cousin called GIP, which the scientists theorized could produce even more insulin and suppress appetite further.
Two weeks after starting to get the compound, chubby laboratory mice given the compound lost 20% to 25% of their weight.
Drug effects in mice, such as weight loss, often don’t carry over to humans. Despite the unknowns, Lilly went ahead and greenlighted the experimental drug for human testing.
“It was the largest degree of weight loss I had ever seen in a mouse model of obesity. It felt pretty compelling,” Dr. Skovronsky said.
Initially, the plan was to get the drug candidate through clinical testing and approved for marketing as a diabetes treatment in 2024, Dr. Skovronsky said. Then Lilly reorganized to move more quickly.
To stop the second-guessing of decisions, Lilly established independent internal units operating like biotech companies—with less bureaucracy and faster decision-making—to manage each of its high-priority drug projects.
Lilly dubbed the new project “GIP Bio,” said Ruth Gimeno, a biologist who joined from Pfizer in 2011 and now serves as Lilly’s vice president of diabetes, obesity and cardiometabolic research.
GIP Bio had its own board of directors, made up of senior researchers and executives from Lilly’s diabetes business unit. They were given a budget, and charged with making quick decisions on their own.
After a Lilly researcher proposed a last-minute change to the design of the second phase of human testing, the GIP Bio board met within 24 hours and approved the change so the study could start on time, Dr. Gimeno said.
Results from the study in people echoed the findings in overweight laboratory mice. The drug candidate, which Lilly was then referring to by the chemical name tirzepatide and later branded as Mounjaro, not only cut blood sugar levels sharply in people with diabetes but also helped them lose much more weight than older diet drugs could achieve.
Lilly released the Phase 2 tirzepatide results publicly in October 2018 at a diabetes conference in Berlin.
Julio Rosenstock, a veteran diabetes doctor, took the microphone to share his reaction. Dr. Rosenstock, senior scientific advisor at clinical-trial site operator Velocity Clinical Research and a clinical professor of medicine at University of Texas Southwestern Medical Center in Dallas, wasn’t involved in the study but has worked with Lilly on other studies.
He said he had nicknamed Ozempic, the drug from Lilly’s rival Novo Nordisk, the “gorilla” because it had been the most potent GLP-1 containing drug to that point.
“But tirzepatide is really a King Kong,” Dr. Rosenstock said.
Just as the Phase 2 testing was getting off the ground, Lilly started spending money to prepare for the third and final phase of testing required to gain regulatory approval.
Typically, companies wait before starting the last-stage studies because they can cost several hundred million dollars.
Lilly decided it was worth the risk for certain high-priority drugs, however, because that could hasten their speed to market.
The Phase 3 studies began in late 2018. The decision to go ahead with the investments ultimately cut about nine to 12 months off the development timeline, Dr. Skovronsky said.
In May 2022, the FDA approved Mounjaro for the treatment of Type 2 diabetes. Lilly expects to complete the application for Mounjaro’s use treating obesity after results of another study are available by the end of April, which could lead to approval later this year or early 2024.
Though doctors consider it to be safe, Mounjaro does have side effects, with the most common being nausea and other gastrointestinal issues. Similar side effects have been reported for Ozempic and Wegovy.
Lilly is studying the drug for additional uses like treating a liver disease, and is monitoring whether the weight loss it induces has downstream benefits including heart health.
“To me, tirzepatide in my career may be the most important drug Lilly’s been a part of,” said Mr. Ricks, the chief executive, who has worked at Lilly for more than 25 years.
“It is one of the rare ones that has a chance to move the life expectancy of the population.”
Now, Lilly is developing a drug that adds a third component, called glucagon, to GLP-1 and GIP, to see if that induces even greater weight loss. Phase 3 studies are set to begin this year.
The drug could be up for FDA approval in 2026, well before Mounjaro’s key U.S. patent expires in 2036.
Updated: 4-9-2023
WeightWatchers Doesn’t Want To Get Netflixed by Ozempic
The WW deal for telehealth provider Sequence underscores the seismic shift in obesity management.
Do the new weight-loss drugs spell doom for companies like WeightWatchers?
The news that the company — now known as WW — has acquired Sequence, a telehealth provider that for a monthly fee can prescribe drugs like Wegovy and Ozempic, certainly suggests that today’s drugs pose an existential threat to companies focused on behavioral approaches to weight loss.
And Insider reported that Noom, an app that helps users track their eating and exercise habits, has quietly started a service to prescribe weight-loss drugs to some customers.
As BMO Capital Markets analyst Evan Seigerman put it in a note to clients, “Today’s announcement by WW feels reminiscent of the demise of Blockbuster, whose business model was made irrelevant when digital streaming became a reality.”
WW says the acquisition simply follows the science. If you compare the weight loss effects of lifestyle modification (including WW users), bariatric surgery, older pharmaceutical interventions and the new class of drugs, the differences are clear, says Fatima Cody Stanford, an obesity doctor at Harvard Medical School and Massachusetts General Hospital. Wegovy handily beats intensive behavioral therapy across all levels of weight loss.
But there’s still a role for diet and exercise, and that’s what WW and Noom are banking on. Combining the new drugs with lifestyle changes provides (modest) improvements over the drugs alone.
And obviously, what we eat and how much we exercise is critical to improving long-term health outcomes like lowering the risk of heart disease or diabetes.
And there could be more a practical upside to integrating apps that track behavioral changes and pharmaceutical interventions: It might help convince insurance providers to pay for these expensive drugs.
Currently, insurance coverage for Wegovy and its competitors is spotty to nonexistent. That’s a huge barrier when the regimen can cost more than $1,600 per month — and when patients must take the drug indefinitely to keep the weight off.
WW said as much to investors, noting that the combination creates an approach that “may appeal to payors, employers, and health systems who bear risk to reduce total cost of care.”
And notably, the US Food and Drug Administration approved the drugs with the proviso that they should be used as an add-on to behavioral changes, a position echoed in the positions doctors have taken.
For example, when the American Academy of Pediatrics recently made the controversial (but, as I wrote, reasonable) call to include pharmaceutical interventions in its recommendations for treatment of children with obesity, the AAP was clear that drugs should only be prescribed alongside lifestyle changes, noting that “no current evidence supports weight loss medication use as a monotherapy.”
Beyond providing a potential way to convince more insurers that the drugs are worth paying for, the moves by WW and Noom could offer some other advantages for patients.
Telemedicine could improve access for people who live far away from obesity medicine specialists, or those who are on long wait lists to see one.
None of this means there aren’t plenty of things to worry about when it comes to the quickly changing landscape for weight-loss management, including components of this deal.
For example, WW and Sequence have a responsibility to ensure the drugs are only prescribed to those who genuinely fall under their recommended use.
If TikTok trends and Hollywood fads are any indicator, misuse of Ozempic is already wildly out of control. The weight-loss industry needs to be clear that these drugs are not a temporary life hack, but part of disease management.
But the deal is tacit acknowledgment that even weight-loss companies understand that slimming down is about more than sheer willpower. The big question is whether insurance companies are willing to admit that, too.
Updated: 4-10-2023
The $76 Billion Diet Industry Asks: What To Do About Ozempic?
Weight-loss businesses, which long pushed calorie-counting and willpower, are grappling with the surging popularity of new drugs.
Annick Lenoir-Peek, a lawyer from Durham, N.C., has struggled with her weight since adolescence. She has tried Atkins and keto and spent thousands of dollars over decades on weight-loss efforts and programs such as Noom, Nutrisystem and WeightWatchers.
Since starting Ozempic in late November, she has lost around 30 pounds. Her cholesterol and glucose levels have improved, and she can eat far fewer calories without feeling hungry, she says. She has felt few side effects and has more energy than when she tried calorie-restricted diets.
Currently on a trip through Eastern Europe, she says she is doing more tours than she would have at a higher weight.
People such as Ms. Lenoir-Peek—among the diet business’s most reliable customers—are sparking an existential crisis for the industry, which rang up $76 billion in sales in 2022 from weight loss and medical programs, diet soda and low-calorie frozen food, gym memberships and other categories, according to research firm Marketdata LLC.
Drugs such as Ozempic, Wegovy and Mounjaro have upended the business of losing weight in America. They are shaping up to be blockbusters for Novo Nordisk, which makes Ozempic and Wegovy, and Eli Lilly, which makes Mounjaro.
The drugs are also ripping up long-held beliefs that diet, exercise and willpower are the way to weight loss.
Drugs such as Ozempic, Wegovy and Mounjaro have upended the business of losing weight in America. They are shaping up to be blockbusters for Novo Nordisk, which makes Ozempic and Wegovy, and Eli Lilly, which makes Mounjaro. The drugs are also ripping up long-held beliefs that diet, exercise and willpower are the way to weight loss.
“I think they [the new drugs] are going to transform the industry of weight loss in a pretty big way,” says Alex Fuhrman, senior research analyst at Craig-Hallum Capital Group LLC.
“The traditional approach to weight loss has been the only game in town for a very long time except for more drastic surgical interventions. The behavioral approach to weight loss is going to be under pressure now.”
Some big players, including WeightWatchers, are embracing the drugs, sometimes referred to as GLP-1s, and betting on them as a critical part of the industry’s future.
Others are positioning their diet and exercise programs as a natural alternative to pharmaceuticals, championing the notion that there is no quick fix.
Some are noting the medications’ downsides: Side effects such as nausea and diarrhea, and the prospect of having to be on them long-term to keep the weight off. At stake for companies are their business models; for consumers, their health.
“Most of my clients have been on a ton of different diets. They’ve been on everything,” says Alix Turoff, a registered dietitian and personal trainer. “No one can follow them because they’re so insanely restrictive.”
An Industry Shift
Companies have profited off Americans’ weight-loss dreams for decades. WeightWatchers, founded in 1963, brought its promise of weight loss through food-tracking and lifestyle changes to generations of Americans.
In the ’80s, Jane Fonda told Americans to feel the burn; Atkins later surged to popularity urging people to cut carbs.
Weight-loss drugs have come and gone, often felled by links to health problems. The fen-phen diet-drug combination was thought to be a miracle treatment in the 1990s but was taken off the market when some users developed heart issues.
Dexatrim, a once-popular over-the-counter pill, was linked to an increased risk of strokes.
And still the country got heavier, with about 42% of American adults having obesity, according to data collected between 2017 and March 2020 by the Centers for Disease Control and Prevention. That is up from about 31% in 1999-2000.
What is striking about Ozempic, Wegovy and Mounjaro is that they do lead to significant weight loss, according to research, doctors and patients. Ozempic and Mounjaro are approved to treat Type 2 diabetes, but patients also take them off-label for weight loss.
Wegovy is approved for weight loss for people who are obese, or overweight with a related condition. Ozempic’s list price is about $892 for a roughly one-month supply, without insurance. Wegovy’s is about $1,350.
For weight-loss companies, the question now is whether to embrace the drugs, stick with pushing diet and lifestyle changes, or find a middle ground.
No moment better illustrates the industry shift than WeightWatchers’ decision to buy Sequence, a telehealth company that connects members to doctors who can prescribe Ozempic, Wegovy, Mounjaro and other drugs used for weight loss.
WeightWatchers has spent the past 60 years telling members they can lose weight by tracking the food they eat, doing frequent weight checks and making other lifestyle changes. But fewer people are banking on that promise:
The company’s membership declined to 3.5 million at the end of 2022 from 4.2 million a year earlier and down from its record high of 5.03 million subscribers at the end of the first quarter of 2020.
Now, WeightWatchers sees an opportunity to market access to the new drugs to its former members.
“There are probably people we could have served better,” says Sima Sistani, the company’s chief executive. “For some, these medications can help them adhere to a program like WeightWatchers.” Many current members are potential candidates for the medications, too, she says.
WeightWatchers’s embrace of the drugs has upset some within its own community, with some members expressing disappointment and even a sense of betrayal. Ms. Sistani, who has received some angry member messages, says she is surprised by the backlash.
“There are parts of our community that believe because I did it the hard way, quote unquote, that that’s what everybody else should do,” she says.
WeightWatchers also plans to create programs geared to members who are using the GLP-1 medications, since when people lose weight quickly they often lose important muscle mass.
The drug can cause people’s appetite to change dramatically, so they may need additional guidance to get adequate nutrition.
Noom Inc., which has an online dieting program that color-codes foods based on calories, quietly launched a program offering GLP-1 drugs to clients last fall, hiring its first medical director to manage the service in December.
Noom’s main weight-loss program can cost from $70 a month to $209 a year depending on the plan.
(Novo Holdings, the controlling shareholder in Ozempic and Wegovy maker Novo Nordisk, is an investor in Noom.) Clients who sign up for Noom’s core dieting and coaching program can sign up for weight-loss drugs if they qualify. For now, there is no mention of the service on Noom’s website and it isn’t advertising the program.
Linda Anegawa, Noom’s new medical director, says the program is in pilot mode. “We feel treating weight loss from a medical standpoint is a natural complement to Noom’s behavioral-change tool that’s been in development for over a decade,” Dr. Anegawa says.
Noting The Downsides
Other weight-loss companies are pointing out the drawbacks of the new drugs.
“The early side effects are massive nausea and diarrhea. The more concerning parts are the unstudied side effects,” says Dan Chard, chairman and chief executive officer of Medifast, whose company’s Optavia program generated $1.6 billion in revenue in 2022.
Mr. Chard notes that a medication’s long-term side effects may take years to fully emerge. He also says that the drugs’ off-label prescribing is a concern.
Mr. Chard says he sees Optavia as an alternative or a complement to the medications. On Optavia’s most popular plan, users consume between 800 and 1,000 calories by eating five “fuelings” a day, Medifast products such as shakes, bars and soups, and one home-cooked “Lean and Green” meal.
Users are paired with coaches, most of whom started as Optavia users, who earn a commission on sales of the company’s products and provide support and information on healthy habits. “We feel confident with our approach,” says Mr. Chard.
Herbalife, a global direct-selling company that offers products including meal-replacement shakes and bars and nutritional supplements, sees its role as both an alternative and adjunct to the new drugs, says Kent Bradley, the company’s chief health and nutrition officer.
He says that long-term, sustainable change requires a lifestyle shift.
Dr. Bradley rejects the notion that the medications may make his company’s meal-replacement products obsolete. His brother is an optometrist, and Dr. Bradley likens the introduction of the GLP-1s to what happened in the eye-care field with the introduction of Lasik surgery.
When Lasik came out, his brother’s ophthalmologist colleagues speculated that optometrists, who prescribe glasses but generally don’t do surgery, would soon be out of business, Dr. Bradley says.
Instead, his business increased, he says, due to patients seeking screening for surgery and needing glasses after the procedures.
Simply Good Foods Co.’s Atkins brand, which sells products based on its protein-rich, low-carbohydrate diet, says its program may be a “good partner” for the drugs, according to Colette Heimowitz, the company’s vice president of nutrition and education.
Protein can prevent the loss of lean muscle mass, she says, and the high fiber in the Atkins approach can help reduce some of the side effects of GLP-1s such as diarrhea. Ms.
Heimowitz says the healthy fats in the Atkins method can potentially reduce the risk of what has been dubbed “Ozempic face,” a haggard, aged appearance that can sometimes occur with rapid weight loss.
Ms. Heimowitz also sees Atkins as an off-ramp for people who need to stop the medications.
At Canyon Ranch, longtime medical director Stephen C. Brewer says he is hoping the craze over new drugs for weight loss subsides.
He has prescribed the drugs to some guests who had diabetes and were overweight and for whom it made sense but is reluctant to do so for others who haven’t exhausted other avenues for weight loss.
While being overweight puts people at higher risk for chronic disease, he notes that there are other aspects of a person’s health to consider, as well.
“I’m always hoping that there are places like us that are the voice of reason,” Dr. Brewer says.
Updated: 4-16-2023
WeightWatchers Gets A Makeover With Obesity-Drug Business
The decision to get into the business for drugs such as Ozempic makes sense.
If you can’t beat them, join them.
WeightWatchers acquisition of the drug-prescribing business Sequence, which closed last Monday, is a clear sign that the center of gravity in the weight-loss industry is shifting.
Whereas conventional wisdom for decades was that obesity could only be addressed through lifestyle changes, powerful new drugs are starting to change that narrative, forcing the old guard to adapt or die.
Obesity-diabetes drugs such as Ozempic, Wegovy and Mounjaro are well on their way to becoming one of the largest drug classes in the history of the pharmaceutical business, with some analysts seeing annual sales peaking in the $100 billion range.
That will mostly benefit the drugmakers such as Eli Lilly and Novo Nordisk, whose stocks have more than doubled in the past three years.
On Thursday, Danish pharma company Novo Nordisk raised its full-year guidance due primarily to strong sales of Wegovy. In a country struggling with obesity, Novo and Lilly can barely keep up with the rapid increase in demand for these drugs, known as GLP-1s.
The remarkable weight loss patients experience when taking these injectables might also have something to do with the decline in subscriptions at WeightWatchers, which helps customers trim their waistline through changes in lifestyle and diet.
WeightWatchers’ business has been in deep trouble in recent years as its subscriber base has shrunk from about five million a few years ago to 3.5 million in the last quarter. The stock is down nearly 90% over the past five years.
It is only natural, then, that WeightWatchers wants a piece of the GLP-1 action.
For $106 million, the company gets Sequence, whose 24,000 members pay $99 a month for access to telehealth appointments with doctors who can prescribe drugs such as Ozempic.
The program also includes an app where people can track their weight loss and have access to meetings with dietitians and fitness coaches.
Now that Sequence is attached to WeightWatchers, its growth should accelerate. Goldman Sachs thinks its subscriber base can rise to 645,000 by 2025, though that could be a conservative estimate given the unrelenting demand for these drugs in the U.S.
The key point is that WeightWatchers already possesses an extensive database of users to which it can now market this new offering, says Goldman analyst Jason English.
“We believe the launch of this solution set will be effective as it targets its marketing message to its roughly 20 million database of lapsed WW users,” he wrote, noting that this is a “cohort of consumers who have historically been overweight or obese, [and] have demonstrated a willingness to pay for help.”
There is no doubt an opportunity for companies who can help patients navigate the insurance maze with relative ease while offering a broader package that goes beyond just medication.
As effective as these drugs may be, patients will put the weight back on if they don’t eventually transition to a new lifestyle.
Sequence could, in theory, help bring some of those users to the legacy platform, though that requires a lot of faith in the willpower of the American consumer.
Regardless, it will be extremely important that the company avoid the pitfalls of aggressive marketing that many telehealth companies have been accused of in recent years.
The main competition to online services such as Sequence is the traditional doctor. UBS’s Michael Lasser writes that “many of those in need of a medicinal alternative for weight loss might just seek out support from their primary care physician.”
While the stock is soaring as investors price in the large size of the addressable market, Mr. Lasser cautions that reality will eventually come into focus, and “the company will need to show what’s realistic.”
The treatment of obesity is destined to be upended as these new drugs continue to gain traction. While the need for exercise and dieting will always exist, traditional weight-loss businesses that broaden out their approach to make room for medication are likely to prosper.
Updated: 4-17-2023
Demand For Obesity Drugs Has Insurers Paying Close Attention
Strong sales of obesity-diabetes drugs known as GLP-1s could affect insurance companies’ financial results.
Not too much is standing in the way of a new class of obesity-diabetes drugs from becoming one of the biggest blockbusters the pharmaceutical industry has ever seen.
Doctors are excited about the potential health benefits, the social-media hype just won’t abate and the weight loss can be dramatic.
There is one major question still looming over this drug class, known as GLP-1s, though: What do insurers, who ultimately foot the bill, think about this new revolution in the treatment of obesity?
If this class of drugs can truly surpass $100 billion in annual sales, as many analysts expect, insurers (and ultimately employers and the government) will have to foot the bill.
On Friday, UnitedHealth Group Chief Executive Officer Andrew Witty fired a first salvo, signaling there will be a tough approach both on pricing as well as on just who gets to take these drugs.
While GLP-1s are expected to continue to gain pace as a treatment for diabetes, just who gets covered for the treatment of obesity will be up for debate.
There is no question the drugs have captured the national imagination in a way that only few others, such as Viagra or Adderall, have done. And with so many overweight Americans, demand won’t be a problem.
That means insurers will be making some tough decisions on who gets to take drugs such as Ozempic and Mounjaro, which is expected to receive Food and Drug Administration approval for obesity this year.
“We need to really be clear about which patients really do benefit from these medicines and make sure we properly understand how they’re going to use those medicines,” Mr. Witty said during a call with Wall Street analysts following the company’s earnings release. “So there’s a lot still to learn, I think, as these things progress through their final phases.”
In more blunt terms, what Mr. Witty seems to have meant is that UnitedHealth isn’t going to foot the bill for every American looking to shed a few pounds just to look good in a swimsuit at the beach this summer.
As Lisa Gill, the JPMorgan analyst who asked the GLP-1 question during the call explains, these are expensive drugs, so medical necessity will be scrutinized.
“If you have a very high BMI and you’re obese, you’re probably going to be in a place where there’s a higher probability you might have heart disease and these drugs could help you,” she says.
“But they’re not just going to start giving a green light to every doctor that writes a prescription for drugs that cost $6,000 a month.”
Looking at this year, UnitedHealth executives didn’t seem too concerned about an impact from these drugs on their financial results, noting that pharmaceutical spending is 20% of their overall expenditure and any one drug only makes up as much as 1% of overall expenses. Not every insurer is so nonchalant.
In late March, the chief financial officer of Elevance ELV 2.88%increase; green up pointing triangle, formerly known as Anthem, John Gallina spooked investors during an investor day conference when he suggested that diabetes and weight-loss drugs were running “a bit hotter than we had expected.”
That comment briefly hit stock prices across the entire insurance sector. Strong results from Novo Nordisk and Eli Lilly will likely pressure Elevance shares, David Windley, an analyst at Jefferies, wrote following the event.
Ms. Gill from JPMorgan says that vertically integrated insurers that also own large pharmacy-benefit managers—the middlemen negotiating drug prices—may come out just fine since higher volume for these drugs drives up their PBM profits via rebates.
That should help insulate UnitedHealth from too big of a negative impact, but Elevance, which owns a smaller PBM, is less protected.
Mr. Gallina’s comments about demand running hot were supported by Novo Nordisk last week. In a pre-announcement on Thursday, Novo, which will report its full quarterly results in May, raised its full-year guidance due primarily to strong sales of Wegovy, a GLP-1.
Perhaps just as important as current prescription trends will be a key study from Novo Nordisk that could have a big impact on how insurers decide to provide coverage for this new class of drugs.
The late-stage study, named Select, aims to determine whether Novo Nordisk’s Ozempic can cut cardiovascular risks in overweight patients. Proving that obesity drugs not only help people trim waistlines but also reduce mortality will be key to getting broader buy-in from insurers.
For drugs to reach true blockbuster status, popular buzz alone isn’t enough. Insurers need to play ball, too.
Updated: 4-18-2023
‘I Hate You, Kathie Lee Gifford!’ Ozempic Users Report Bizarre Dreams
Viral weight-loss drugs trigger vivid, celebrity-filled night visions; buying cabinets from Clint Eastwood.
Chery McLemore was recently enjoying watching a parade of cows and bulls at a cattle auction with actor Matthew McConaughey. Then he started climbing atop the small pet llama she had brought, and she had to raise her voice to tell him to stop.
“Fine, you can stay with your llama, I’m leaving!” Mr. McConaughey screamed before storming off.
Then Ms. McLemore woke up.
Her trip to a cattle auction with one of Hollywood’s leading men was one of the dreams Ms. McLemore has had since February, when she began taking one of the powerful, new weight-loss drugs that have become viral sensations in recent months.
Ozempic and other similar medications are doing more than helping people tighten belts and fit into old outfits. Many users are reporting bizarre, vivid and eerily realistic night visions that bear no resemblance to their past dreams.
The Ozempic dreams they have been reporting are downright wacky. Among the plots people have posted on social-media groups: joining the cast of “The Golden Girls” and preparing to rob a museum with Jennifer Lopez and Ben Affleck.
“I am carrying your baby,” one dreamer recalled telling Dwayne “The Rock” Johnson, who she thought, judging by his reaction, was open to blending their families.
The overnight spectacles are a side effect of drugs such as Ozempic and Wegovy from Novo Nordisk A/S and Mounjaro from Eli Lilly & Co.
The companies originally developed the drugs, which require weekly injections, to treat diabetes, only to discover they were much better than older diet drugs at helping people lose weight, with manageable risks.
The Food and Drug Administration has approved Wegovy for losing weight, while diabetes treatment Mounjaro is under review for that use. Ozempic, which has the same main ingredient as Wegovy, is approved to treat diabetes but is widely used for weight loss.
Fueled by celebrity endorsements, use has taken off. On Facebook and other social-media platforms, thousands have traded tips about where to find scarce supplies and how to manage side effects, including sulfur-tasting burps.
They also have celebrated “non-scale victories” such as buckling a seat belt more easily.
Strange dreams are another hot topic, prompting some users to start social-media groups dedicated to recalling the strange, colorful details.
The TikTok account “ozempicdreams” posts brief videos of text describing dreams submitted by followers, setting them to music to match the mood. “Spent the night at Home Depot ordering new cabinets and appliances for my kitchen.
My salesman, Clint Eastwood, helped me pick out everything I would need,” read one, accompanied by a sped-up version of the song “Escapism,” by the singers Raye and 070 Shake.
Ms. McLemore’s Facebook post about her cattle-auction dream drew more than 160 comments, including several admonishing her dream self for not letting the imaginary Mr. McConaughey sit on the little llama in the first place.
Celebrities star in many of the reported dreams. In one woman’s, according to a Facebook post, actor Jonah Hill waited tables on a cruise, divulged to the diners that he had changed his name to “Sundaze” and planned to quit film acting for a cabaret career.
Oprah Winfrey rode in a go-kart to rescue an Ozempic user who was playing in a baseball game before a hostile crowd, according to a Facebook post.
Another Ozempic dreamer was about to give birth in a bar and wanted actor Will Ferrell, standing nearby, to deliver the baby.
A Novo Nordisk spokeswoman said the company has received reports of abnormal dreams among users of Ozempic and Wegovy, but it doesn’t have enough information to establish whether the drugs are causing the dreams.
An Eli Lilly spokeswoman declined to comment on the reports of strange dreams among Mounjaro users.
Dr. Caroline Apovian, a professor of medicine at Harvard Medical School and co-director of a weight-loss center at Brigham & Women’s Hospital in Boston, suspects the dreams are related to how the drugs work promoting production of a gut hormone that has receptors in the brain.
The drugs might also increase a user’s energy expenditure during sleep, which could contribute to vivid dreams, she said.
It also is possible the drugs are helping users’ recall dreams they would have had normally but used to forget, said Deirdre Leigh Barrett, a dream researcher at Harvard Medical School and author of “Pandemic Dreams.”
As for those celebrity guest appearances, Dr. Barrett said, those are unlikely to be specific to the drugs. She said posts on social media about celebrities in strange dreams might be prompting others to respond with similar recollections.
Other medicines have been known to cause strange or vivid dreams. Antidepressants including Paxil and smoking-cessation drug Chantix are associated with abnormal dreams.
The drugs’ labels advise doctors to consider adjusting the dose if patients experience strange dreams.
Arnetrice Knight, a beauty consultant from Oxon Hill, Md., who has been taking Ozempic for diabetes since January, said she told her doctor about her recent dreams, but the doctor told her it wasn’t a problem unless she starts sleepwalking.
Often her dreams involve taking a peaceful drive on a sunny day and having a conversation with her passenger. “None of it made sense, but it was OK because we were driving and it was nice outside,” she said.
One night, however, she dreamed she was sitting in a dark house watching TV when the doors disappeared. A creepy figure lurked in the shadows, and she tried to escape. “It was Stephen King scary,” Ms. Knight said.
The dreams have been so lifelike, she said, that she wakes up still thinking she is in the dream, before she realizes she’s in bed next to her sleeping husband.
Ms. McLemore, a mother of two from Amarillo, Texas, who takes Wegovy, said she typically used to dream about her family, but since taking Wegovy has had slumber meetups with celebrities.
In addition to attending the cattle auction with Mr. McConaughey, she has shopped for shoes with “Jurassic Park” actor Jeff Goldblum, seen an eye doctor with the “Pirates of the Caribbean” star Johnny Depp and toured North Korea with Minnie Pearl of “Hee Haw” fame.
On a camping trip in Yellowstone National Park, Ms. McLemore couldn’t agree with her celebrity dream partner on how to set up the tent. “I hate you, Kathie Lee Gifford!” she recalled screaming.
Updated: 10-2-2023
If Ozempic Leads People To Eat Less, Maker of Cheez-It Will Be Ready
* Kellanova Prepared For Mitigation If Needed, Cahillane Says
* Former Kellogg Co. Just Completed Separation From Cereal Arm
Diabetes drug Ozempic and other appetite-suppressing medications are set to help overweight Americans cut down on their calorie intake. US food companies are unlikely to sit idly by.
“We’re by no means complacent,” Kellanova Chief Executive Officer Steve Cahillane said in an interview Monday.
The recently renamed Kellogg Co. just completed its separation from its North American cereal business, now called WK Kellogg Co. Kellanova is a global snacking company, with brands like Cheez-It, Pringles and Rice Krispies Treats. Snack consumption is on the rise and is expected to continue growing.
Cahillane called it “very, very early days” for the drug but said the company was studying its potential impact on dietary behaviors so it could respond if necessary.
“Like everything that potentially impacts our business, we’ll look at it, study it and, if necessary, mitigate,” Cahillane said, emphasizing that it was premature to make predictions.
Discussing how his business has responded to similar threats, Cahillane pointed to the UK’s High Fat Sugar Salt regulations, which restricted how certain foods could be marketed.
In response, the company changed its formulations and merchandising strategies, Cahillane said. “What happened is, our business wasn’t affected,” he said.
Although prescriptions for these drugs skyrocketed 300% from 2020 to 2022, it’s still too early to know how they will affect consumer behaviors like eating habits on a large scale. Ozempic is made by Novo Nordisk A/S.
Last week, an analyst report from Jefferies said Kellogg was among the snack makers with the most potential exposure but noted that the firm couldn’t yet measure any volume shift.
Kellanova is looking at potential penetration levels in both the US and other markets, he said. “Who’s gonna cover it? Who’s gonna be on it? Do they stay on it?” he said, listing questions the company will try to answer. “There’s growing stories about the secondary effects of it, so we’re studying that,” he added.
Updated: 10-3-2023
Short Fast Food Stocks Because of Ozempic, Barclays Says
* Demand For Snacks, Cigarettes May Be Hit As Drugs Proliferate
* Stocks Seem To Have Been Hit Hard By Fear of Loss of Appetite
Drugs used for weight loss like Ozempic pose a real risk to companies ranging from fast food restaurants to cigarette makers, and credit market prices don’t fully reflect the potential downside, according to a report from Barclays strategists.
Pharmaceuticals known as GLP-1 agonists help people lose weight while anecdotal evidence suggests they also cut urges to consume addictive substances, including alcohol, and cigarettes.
The growing popularity of the drugs could hurt demand for companies including PepsiCo Inc., the maker of Pepsi soda and Lay’s potato chips, McDonald’s Corp., and Altria Group Inc,, the cigarette maker, according to Barclays strategists led by Jigar Patel in a note on Tuesday.
Those concerns may be reflected in stock prices: an index of packaged foods is down by about 14% this year, even as the S&P 500 index has risen by around 10%. But in credit derivatives markets, investors seem to believe most of these companies have grown less risky.
For example the cost of insuring McDonald’s debt against default for five years has fallen about 6 basis points, or 0.06 percentage point, over the last year to 36 bps.
“The impacts of GLP-1s potentially introduce disruption into a number of industries,” the strategists wrote.
Some companies will benefit from the proliferation of these drugs, the strategists wrote. CVS Health Corp., for example, could benefit if more consumers get prescriptions for these drugs, and its Aetna health insurance division could benefit if people lose weight.
A spokesperson for Pepsi declined to comment, citing a quiet period before earnings. Representatives for Altria, McDonald’s, and CVS didn’t respond to requests for comment.
Barclays suggests selling credit protection on a basket of companies that might benefit from GLP-1 drugs and buying protection on those that might get hurt.
The prices on these two baskets are only about 5 basis points apart, the tighter end of the one-year range, signaling that now is a relatively good time to enter the trade.
Management teams are still determining how the drugs will affect their businesses. Steve Cahillane, the chief executive officer of Kellanova, the snack maker recently separated from cereal maker WK Kellogg Co., said on Monday that the company is studying the impact of these drugs on consumer diets so it can adjust its business as necessary.
The rise of GLP-1 drugs is one theme that Barclays is focusing on for the next several years, along with the rise of artificial intelligence, energy transition, and telecom, media and technology disintermediation.
Updated: 10-4-2023
Ozempic Is Making People Buy Less Food, Walmart Says
* Retailer Is Analyzing Shopping Behavior Of People On Drug
* Prescriptions For GLP-1s, Like Ozempic, Are Boosting Sales
Walmart Inc. says it’s already seeing an impact on food-shopping demand from people taking the diabetes drug Ozempic, Wegovy and other appetite-suppressing medications.
“We definitely do see a slight change compared to the total population, we do see a slight pullback in overall basket,” John Furner, the chief executive officer of Walmart’s sprawling US operation, said in an interview Wednesday. “Just less units, slightly less calories.”
The Bentonville, Arkansas-based retailer is studying changes in sales patterns using anonymized data on shopper populations. It can look at the purchasing changes among people taking the drug and can also compare those habits to similar people who aren’t taking the shots.
Furner said it’s too early to draw any definitive conclusions about the appetite-suppressing drugs made by Novo Nordisk A/S, and similar medicines.
An increasing number of CEOs and investors are talking about how popular weight-loss drugs might change the economy and business. Earlier this week, the CEO of the maker of Pringles and Cheez-Its said the company is studying their potential impact on dietary behaviors.
“Like everything that potentially impacts our business, we’ll look at it, study it and, if necessary, mitigate,” Steve Cahillane, the CEO of Kellanova, said in an interview.
Walmart sells GLP-1 drugs, a category that includes Ozempic, through its pharmacies. In August, it said they were giving the retailer a revenue boost. US sales for those medicines increased 300% between 2020 and 2022, according to a recent report from Trilliant Health.
“We still expect food, consumables, and health and wellness primarily due to the popularity of some GLP-1 drugs to grow as a percent of total in the back half,” Walmart CEO Doug McMillon said on a call with analysts in August.
There are other potential positive effects on sales as well, Chief Financial Officer John David Rainey said at the time. Customers taking weight-loss drugs “tend to spend more with us overall” even as they buy less food, Rainey told CNBC.
Updated: 10-5-2023
America’s Food Giants Confront The Ozempic Era
Nearly 7% of the population is projected to be on weight-loss drugs in 2035.
You just started taking Ozempic. Will you still crave that bag of potato chips?
Big food companies and investors are watching as Ozempic and other similar weight-loss drugs flow to millions of people, upending America’s diet industry and raising new questions about how consumers will eat.
Executives at food manufacturers from Campbell Soup to Conagra Brands said they are fielding questions from investors about the drugs’ potential impact, as internal teams start to assess consumer behavior and brainstorm ways to respond.
The drugs, which suppress patients’ appetites, have exploded in popularity in the U.S., straining manufacturing capacity.
Morgan Stanley has projected that 24 million people, or nearly 7% of the U.S. population, will be taking such medications in 2035.
Those people could cut their daily calorie consumption by as much as 30%, according to the firm, which surveyed over 300 patients. For a person on a 2,000-calorie diet, that could mean eliminating a one-ounce bag of salted potato chips, a bottle of soda and more each day.
Carolyn MacBain-Waldo said she is eating significantly less since she started taking Eli Lilly’s Mounjaro—her family orders from restaurants less often and their grocery bills have dropped by as much as 20%.
The 50-year-old, who works as a senior director in retail, said the drug makes her feel full more quickly and that she is far less likely to overeat when stressed.
“I still have a fully stocked kitchen, there’s chips and pretzels in there,” MacBain-Waldo said. “I don’t find it tempting.”
Mark Clouse, chief executive of Campbell, which along with its namesake soups makes Goldfish crackers and Cape Cod potato chips, said he has been struck by the rapid rise of pharmaceutical companies behind the drugs.
“That’s a little bit of an ‘OK, wait a minute, something is going on here,’” Clouse said. Still, he said, he doesn’t buy the idea that the drugs will cut into food sales across the board.
Wall Street has been trying to project the drugs’ impact on sales for food and beverage makers, restaurants and grocery stores. Among food companies, snack and candy manufacturers such as Hershey, Mondelez, Hostess and Campbell might be the most exposed, as patients cut back most on foods that are high in sugar and fat such as cookies and salty snacks, Morgan Stanley said.
A Bernstein report said goods such as candy, where about one-third of sales are to people who consume the products at least once a day, could be most at risk.
Karyn Carlton, 47, who takes Mounjaro, said she doesn’t think about food all the time anymore and eats far fewer snacks.
“The other day I had a single jelly bean, which is unheard of for me,” she said, adding that she also recently ordered a kids’ meal from a fast-food restaurant and felt satiated.
The women said their doctors didn’t recommend specific diets while taking the drug.
The rise of Ozempic and other weight-loss drugs comes as sales growth is slowing for big food companies as consumers begin to balk at higher prices.
Sales roared for food companies as consumers stocked pantries with major brands during the Covid-19 pandemic, and paid more for groceries when inflation took hold.
Now, concerns about companies’ growth prospects in the age of Ozempic are adding to worries over declining sales volumes, increasing pressure on food companies’ stock prices. The S&P 500 Packaged Food & Meat subindex has dropped 14% so far this year, while the S&P 500 has climbed 11%.
Nicholas Fereday, executive director of food and consumer trends for agricultural lender Rabobank, said the drugs pose a new threat to the packaged-food industry’s growth.
“If two-thirds of Americans are the target audience, it’s a huge thing,” Fereday said, referring to the portion of the U.S. population that is considered overweight.
Some Wall Street analysts and food-industry consultants said they don’t view Ozempic, made by Novo Nordisk, and other weight-loss drugs as a big risk for now.
The drugs remain expensive and inaccessible to many Americans, they said, and widespread or long-term adoption isn’t guaranteed, with some people experiencing unpleasant side effects such as nausea and diarrhea.
It is also still unclear which foods patients may opt to eat while taking the drugs, they said.
Many food executives said the drugs have gotten their attention, but that after navigating previous trends, from low-fat to low-carb diets, they aren’t panicking.
Conagra CEO Sean Connolly said that any significant drop in calorie consumption due to the drugs is a long way off. If they do gain traction, Connolly said, food companies could respond with new products, including smaller packaging sizes.
“Snacking tends to be one of the most profitable businesses in food,” Connolly said, and among the fastest-growing. “It’s not necessarily bad news.”
Bob Nolan, Conagra’s senior vice president of demand science, said his team began studying Ozempic and other drugs last fall. He said consumers aren’t discussing them on social media as much as other weight-loss programs such as the keto diet, though that could change once the drugs become available in pill form instead of as injections.
If patients are consuming fewer calories, Nolan said, they will turn to higher quality food, such as Conagra’s Healthy Choice and Marie Callender’s frozen meals, and Birds Eye frozen vegetables. “We have lots of products that will fit this already, but we’ll design new ones if we don’t,” he said.
Mondelez said it is expanding its “portion control” snacks, which are 200 calories or less and individually wrapped. The company also offers products such as gluten-free Oreos and Hu paleo-friendly chocolate bars for specific dietary needs.
Campbell’s Clouse said it is too early to determine whether overall food consumption will decline as a result of the drugs, and that previous changes to American diets have created both challenges and opportunities.
Morgan Stanley’s survey found that patients taking weight-loss drugs ate more fruits, vegetables, poultry and fish, as well as weight-management products such as protein bars and nutritional shakes. The bank said packaged-food companies could adapt by changing their offerings.
MacBain-Waldo said she craves foods such as french fries less since starting Mounjaro, and eats more salads and protein. “I want to make sure I’m putting the right things in,” she said.
Mark Smucker, chief executive of Smucker, which recently agreed to pay $4.6 billion to acquire Twinkies maker Hostess, said the company is monitoring research on weight-loss drugs.
“At this point, I still just feel very bullish that that sweet sort of instant reward that some folks are seeking is never really going to go away,” he said.
Responding to analysts’ questions in June, executives at Simply Good Foods Co., the maker of Atkins-branded food products, said the drugs are driving renewed interest in weight management and bringing consumers into pharmacies where its products are also sold.
They said the company plans to market Atkins products to people who are on the drugs and are seeking more nutritious calories, as well as to patients coming off them, who are trying to keep weight off.
Geoff Tanner, who took over as Simply Good’s CEO in July, said the company had already begun work to find patients and market products directly to them.
“We think this is a wave and we’re going to catch it,” he said.
Updated: 5-17-2026
Weight-Loss Drugs Cause Loss Of Muscle Comparable To A Decade Or More Of Aging!!
Is the weight-loss drug revolution causing a frailty epidemic?
“We are curing obesity by encouraging frailty,” said Daniel Green, principal research fellow at the University of Western Australia, who contributed to the analysis.
The drugs can cause rapid and significant loss of lean muscle mass, up to 10%, comparable to a decade or more of aging, according to an analysis published by the American Diabetes Association.
As millions flock to GLP-1s, doctors warn the drugs can cause rapid and significant muscle loss.
For at least some of the 13 million Americans taking them, losing muscle along with fat is an unexpected downside that isn’t broadly discussed or immediately apparent.
The loss of lean tissue is similar to weight loss from dieting, but the magnitude over a short period can lead to frailty, instability and lack of coordination, doctors and researchers say.
Chanel Robinson achieved exactly what the gold rush of blockbuster weight-loss drugs promised: She lost nearly 100 pounds, lowered her cholesterol to normal levels and reined in her polycystic ovary syndrome.
Yet, nearly three years into her journey on Mounjaro, the 30-year-old from Atlanta, Ga., is discovering the hidden costs of the slimmed-down life.
Robinson experiences muscle fatigue daily, feeling physically weak, frail and often cold.
Robinson said she experiences bursts of sluggishness sporadically during the day, and has trouble with basic tasks like opening a jar. “It shouldn’t be this difficult,” she said.
GLP-1 drugs like Ozempic, Mounjaro and Zepbound have been a success for public health and the pharmaceutical companies that make them.
Obesity rates are falling, the volume of food consumed in America is declining and retailers report a slump in sales of plus-size apparel. It has improved health and happiness for millions of people.
Another concern is that losing muscle could slow down patients’ metabolism, leading to weight regain.
Many taking weight-loss medications initially lose fat and feel great, but quickly start to feel weak and lethargic, he said.
Green’s research showed that the rate of muscle loss could be slowed significantly by regular strength workouts. “It should say ‘must be taken with resistance training’ on the box,” he said.
Drugmakers say weight-loss drugs should be taken only on the advice of a physician and as part of a long-term plan that includes diet and exercise.
A spokesperson for Eli Lilly, maker of Zepbound, said Food and Drug Administration guidelines say it should be used “with increased physical activity.”
The spokesperson added: “Sustainable weight loss is about more than a number on a scale.”
Both Eli Lilly and Novo Nordisk said clinical trials showed users did lose some lean muscle tissue, though at far lower rates than fat.
Liz Skrbkova, a spokeswoman for Novo Nordisk, said that trials for its drug Wegovy showed changes in muscle mass didn’t “significantly differ” from patients who took a placebo.
Eli Lilly said users lost three times more fat weight than lean tissue.
Rayna Kingston, 30, from Denver, said her injections of Zepbound left her feeling so tired the following day that she struggled to complete anything other than basic tasks.
She said she shifted her dose to a Sunday because Mondays were her least busy day. Her partner would bring her meals in bed because she felt so weak.
She stopped exercising, and said her doctor didn’t give her any guidance on strength training or muscle maintenance. “I was relying on Reddit forums to understand what was happening to my body,” she said.
She got so frustrated with the fatigue she came off the medication just under two months later.
Experts say that losing muscle at such a rate can be especially dangerous for those over 50 or with osteoporosis or limited mobility as it could lead to an increased risk of injury.
“Loss of muscle mass is detrimental to moving around and quality of life, but it is also not safe,” said Katsu Funai, associate professor at the University of Utah.
Elderly Americans are set to be able to get GLP-1s from Medicare from July.
There is also pushback from doctors and regulators against using weight-loss drugs as a “quick fix” to lose a bit of weight.
People who take GLP-1s regain weight four times faster than those who lose weight through lifestyle interventions, and weight regained is often mostly fat, according to a recent analysis published in the British Medical Journal.
There currently are few, if any, guidelines or studies on de-prescribing the drugs, researchers say.
The nurse practitioner who prescribed Robinson the medication didn’t warn her that resistance training is essential to maintaining muscle mass, Robinson said. She said she regrets not exercising and now does Pilates once a week.
In the haste to disrupt the obesity epidemic, weight loss has been treated as the singular, undisputed metric of success, which experts say is problematic.
“People worship body weight as an outcome measure because it’s simple, quick and inexpensive,” said Green. “But what matters is fat and muscle mass, which is more expensive to measure as it requires an MRI.”
Grace Parkin, 34, a property manager from Sheffield, England, has lost 125 pounds after she started taking Mounjaro in 2024. “I don’t care about my muscle mass as long as I’m a healthy weight,” she said.
The doctor who prescribed the drug didn’t tell her to exercise, though the pharmacy that sold the medication gave her information on exercise and protein intake, she said.
She didn’t exercise and said she soon felt side effects: a “deathly cold, from the inside” likely because of the drug. Still, she vowed to keep going, saying the weight loss was worth it.
In response to some of the side effects, drug companies are hoping to develop weight-loss treatments aimed at preserving or even building lean muscle mass.
German drugmaker Boehringer Ingelheim recently said it had promising results from one such drug. Eli Lilly last September halted a trial of a similar drug.
While weight-loss medications are designed as lifelong treatments for chronic diseases, namely obesity and Type 2 diabetes, they are increasingly marketed as lifestyle fixes.
Tennis superstar Serena Williams, who used GLP-1s to slim down after having children, was featured in this year’s Super Bowl commercial promoting telehealth company Ro’s weight-loss medication.
Women may be particularly vulnerable to the drugs’s side effects, which can also include nausea, diarrhea, migraines and rarer cases of pancreatitis.
A study last year from a university hospital in Turin, Italy, showed that women are more prone to adverse reactions to weight-loss drugs than men, including muscle loss.
Green, the researcher, said the issue is of particular concern to those taking GLP-1s recreationally and who don’t have much muscle mass to begin with.
Others say a lack of oversight is compounding the issue.
“Patients are self-reporting, and telehealth companies don’t have the patient in front of them to conduct a proper medical assessment,” said Rupal Mathur, an internist in Houston whose practice specializes in weight loss.
She said medical spas are prescribing off-label drugs that don’t meet the criteria set out by the FDA that justify a prescription.
The number of people taking weight-loss drugs who are not living with obesity or Type 2 diabetes is difficult to track since it is unregulated.
However, an analysis by the FDA from 2023 found that more than half of new Ozempic and Mounjaro users didn’t have Type 2 diabetes.
Scientists are calling for more clinical trials to pin down the full effects of weight-loss drugs on muscle loss in different demographics.
“The only studies that have been done have looked at people living with obesity or Type 2 diabetes,” said Green.
“That makes it all the more concerning for those using weight-loss drugs in an ad hoc or unregistered way.”
Related Articles:
Bitcoin Information & Resources (#GotBitcoin)
Snoop Dogg’s Net Worth Is Almost As High As He (Usually) Is
Is It Just Me Or Is America Having A Mental Breakdown? Joker
CFPB (Idiots) Says Staffer Sent 250,000 Consumers’ Data To Personal Account #GotBitcoin
Global Bitcoin Game Theory Is Now Playing Out
Operation Choke Point 2.0 Could Be Bitcoin’s Biggest Banking Crackdown And Regulatory Battle
The US Cracked A $3.4 Billion Crypto Heist—And Bitcoin’s Pseudo-Anonymity😹🤣😂
Twitter To Launch Bitcoin And Stock Trading In Partnership With eToro
Rich Chinese Splashing Out On Luxury In Singapore
Apple Sues NSO Group To Curb The Abuse Of State-Sponsored Spyware
Harvard Quietly Amasses California Vineyards—And The Water Underneath (#GotBitcoin)
US Says China Backed Hackers Who Targeted COVID-19 Vaccine Research
Ultimate Resource For Covid-19 Vaccine Passports
Companies Plan Firings For Anti-Vaxers And Giveaways For Covid-19 Vaccine Recipients
US Bank Lending Slumps By Most On Record In Final Weeks Of March And It’s Impact On Home Buyers
California Defies Doom With No. 1 U.S. Economy
California Wants Its Salton Sea Located In The Imperial Valley To Be ‘Lithium Valley’
Ultimate Resource For Nationwide Firsts Taking Place In California (#GotBitcoin)
Flight To Money Funds Is Adding To The Strains On Banks #GotBitcoin
The Fed Loses Money For The First Time In 107 Years – Why It Matters #GotBitcoin
African Safari Vacation Itinerary (2024 Proposal)
The Next Fountain-of-Youth Craze? Peptide Injections
This Ocean Monster Offers A Potential Climate Solution
What Are Credit Default Swaps, How Do They Work, And How They Go Wrong
Cyberattack Sends Quadrillion Dollars Derivative’s Trading Markets Back To The 1980s #GotBitcoin
What Is Dopamine Fasting? Meet The Dangerous Fad Among Silicon Valley’s Tech Geniuses
Bitcoin Community Leaders Join Longevity Movement
Sean Harribance Shares His Psychic Gifts With The Public
Scientists Achieve Real-Time Communication With Lucid Dreamers In Breakthrough
Does Getting Stoned Help You Get Toned? Gym Rats Embrace Marijuana
Marijuana In Africa Is Like The Gold Rush For America In The 1800s
The Perfect Wine And Weed To Get You Through The Coronavirus Pandemic Lockdown
Mike Tyson’s 420-Acre Weed Ranch Rakes In $500K A Month
What Sex Workers Want To Do With Bitcoin
“Is Bitcoin Reacting To The Chaos Or Is Bitcoin Causing The Chaos?” Max Keiser
How To Safely Store Deposits If You Have More Than $250,000
How To Host A Decentralized Website
Banks Lose Billions (Approx. $52 Billion) As Depositors Seek Higher Deposit Yields #GotBitcoin
Crypto User Recovers Long-Lost Private Keys To Access $4M In Bitcoin
Stripe Stops Processing Payments For Trump Campaign Website
Bitcoin Whales Are Profiting As ‘Weak Hands’ Sell BTC After Price Correction
Pentagon Sees Giant Cargo Cranes As Possible Chinese Spying Tools
Bitcoin’s Volatility Should Burn Investors. It Hasn’t
Bitcoin’s Latest Record Run Is Less Volatile Than The 2017 Boom
“Lettuce Hands” Refers To Investors Who Can’t Deal With The Volatility Of The Cryptocurrency Markets
The Bitcoiners Who Live Off The Grid
US Company Now Lets Travelers Pay For Passports With Bitcoin
After A Year Without Rowdy Tourists, European Cities Want To Keep It That Way
Director Barry Jenkins Is The Travel Nerd’s Travel Nerd
Four Stories Of How People Traveled During Covid
Who Is A Perpetual Traveler (AKA Digital Nomad) Under The US Tax Code
Tricks For Making A Vacation Feel Longer—And More Fulfilling
Travel Has Bounced Back From Coronavirus, But Tourists Stick Close To Home
Nurses Travel From Coronavirus Hot Spot To Hot Spot, From New York To Texas
How To Travel Luxuriously Post- Covid-19, From Private Jets To Hotel Buyouts
Ultimate Travel Resource Covering Business, Personal, Cruise, Flying, Etc.)
Does Bitcoin Boom Mean ‘Better Gold’ Or Bigger Bubble?
Bitcoin’s Slide Dents Price Momentum That Dwarfed Everything
Retail Has Arrived As Paypal Clears $242M In Crypto Sales Nearly Double The Previous Record
Jarlsberg Cheese Offers Significant Bone & Heart-Health Benefits Thanks To Vitamin K2, Says Study
Chrono-Pharmacology Reveals That “When” You Take Your Medication Can Make A Life-Saving Difference
Ultimate Resource For News, Breakthroughs And Innovations In Healthcare
Ultimate Resource For Cooks, Chefs And The Latest Food Trends
Ethereum Use Cases You Might Not Know
Will 1% Yield Force The Fed Into Curve Control?
Ultimate Resource On Hong Kong Vying For World’s Crypto Hub #GotBitcoin
France Moves To Ban Anonymous Crypto Accounts To Prevent Money Laundering
Numerous Times That US (And Other) Regulators Stepped Into Crypto
Where Does This 28% Bitcoin Price Drop Rank In History? Not Even In The Top 5
Traditional Investors View Bitcoin As If It Were A Technology Stock
3 Reasons Why Bitcoin Price Abruptly Dropped 6% After Reaching $15,800
As Bitcoin Approaches $25,000 It Breaks Correlation With Equities
UK Treasury Calls For Feedback On Approach To Cryptocurrency And Stablecoin Regulation
Bitcoin Rebounds While Leaving Everyone In Dark On True Worth
Slow-Twitch vs. Fast-Twitch Muscle Fibers
Biden, Obama Release Campaign Video Applauding Their Achievements
Joe Biden Tops Donald Trump In Polls And Leads In Fundraising (#GotBitcoin)
Trump Gets KPOP’d And Tic Toc’d As Teens Mobilized To Derail Trump’s Tulsa Rally
Schwab’s $200 Million Charge Puts Scrutiny On Robo-Advising
TikTok Is The Place To Go For Financial Advice If You’re A Young Adult
TikTok Is The Place To Go For Financial Advice If You’re A Young Adult
American Shoppers Just Can’t Pass Up A Bargain And Department Stores Pay The Price #GotBitcoin
Motley Fool Adding $5M In Bitcoin To Its ‘10X Portfolio’ — Has A $500K Price Target
Mad Money’s Jim Cramer Invests 1% Of Net Worth In Bitcoin Says, “Gold Is Dangerous”
Ultimate Resource For Financial Advisers By Financial Advisers On Crypto
Anti-ESG Movement Reveals How Blackrock Pulls-off World’s Largest Ponzi Scheme
The Bitcoin Ordinals Protocol Has Caused A Resurgence In Bitcoin Development And Interest
Bitcoin Takes ‘Lion’s Share’ As Institutional Inflows Hit 7-Month High
Bitcoin’s Future Depends On A Handful of Mysterious Coders
Billionaire Hedge Fund Investor Stanley Druckenmiller Says He Owns Bitcoin In CNBC Interview
Bitcoin Billionaire Chamath Palihapitiya Opts Out Of Run For California Governor
Billionaire Took Psychedelics, Got Bitcoin And Is Now Into SPACs
Billionaire’s Bitcoin Dream Shapes His Business Empire In Norway
Trading Firm Of Richest Crypto Billionaire Reveals Buying ‘A Lot More’ Bitcoin Below $30K
Simple Tips To Ensure Your Digital Surveillance Works As It Should
Big (4) Audit Firms Blasted By PCAOB And Gary Gensler, Head Of SEC (#GotBitcoin)
What Crypto Users Need Know About Changes At The SEC
The Ultimate Resource For The Bitcoin Miner And The Mining Industry (Page#2) #GotBitcoin
How Cryptocurrency Can Help In Paying Universal Basic Income (#GotBitcoin)
Gautam Adani Was Briefly World’s Richest Man Only To Be Brought Down By An American Short-Seller
Global Crypto Industry Pledges Aid To Turkey Following Deadly Earthquakes
Money Supply Growth Went Negative Again In December Another Sign Of Recession #GotBitcoin
Here Is How To Tell The Difference Between Bitcoin And Ethereum
Crypto Investors Can Purchase Bankruptcy ‘Put Options’ To Protect Funds On Binance, Coinbase, Kraken
Bitcoin Developers Must Face UK Trial Over Lost Cryptoassets
Google Issues Warning For 2 Billion Chrome Users
How A Lawsuit Against The IRS Is Trying To Expand Privacy For Crypto Users
IRS Uses Cellphone Location Data To Find Suspects
IRS Failed To Collect $2.4 Billion In Taxes From Millionaires
Treasury Calls For Crypto Transfers Over $10,000 To Be Reported To IRS
Can The IRS Be Trusted With Your Data?
US Ransomware Attack Suspect Hails From A Small Ukrainian Town
Alibaba Admits It Was Slow To Report Software Bug After Beijing Rebuke
Japan Defense Ministry Finds Security Threat In Hack
Raoul Pal Believes Institutions Have Finished Taking Profits As Year Winds Up
Yosemite Is Forcing Native American Homeowners To Leave Without Compensation. Here’s Why
What Is Dollar Cost Averaging Bitcoin?
Ultimate Resource On Bitcoin Unit Bias
Best Travel Credit Cards of 2022-2023
A Guarded Generation: How Millennials View Money And Investing (#GotBitcoin)
Bitcoin Enthusiast And CEO Brian Armstrong Buys Los Angeles Home For $133 Million
Nasdaq-Listed Blockchain Firm BTCS To Offer Dividend In Bitcoin; Shares Surge
Ultimate Resource On Kazakhstan As Second In Bitcoin Mining Hash Rate In The World After US
Ultimate Resource On Solana Outages And DDoS Attacks
How Jessica Simpson Almost Lost Her Name And Her Billion Dollar Empire
Sidney Poitier, Actor Who Made Oscars History, Dies At 94
Green Comet Will Be Visible As It Passes By Earth For First Time In 50,000 Years
FTX (SBF) Got Approval From F.D.I.C., State Regulators And Federal Reserve To Buy Tiny Bank!!!
Joe Rogan: I Have A Lot Of Hope For Bitcoin
Teen Cyber Prodigy Stumbled Onto Flaw Letting Him Hijack Teslas
Spyware Finally Got Scary Enough To Freak Lawmakers Out—After It Spied On Them
The First Nuclear-Powered Bitcoin Mine Is Here. Maybe It Can Clean Up Energy FUD
The World’s Best Crypto Policies: How They Do It In 37 Nations
Tonga To Copy El Salvador’s Bill Making Bitcoin Legal Tender, Says Former MP
Wordle Is The New “Lingo” Turning Fans Into Argumentative Strategy Nerds
Prospering In The Pandemic, Some Feel Financial Guilt And Gratitude
Is Art Therapy The Path To Mental Well-Being?
New York, California, New Jersey, And Alabama Move To Ban ‘Forever Chemicals’ In Firefighting Foam
The Mystery Of The Wasting House-Cats
What Pet Owners Should Know About Chronic Kidney Disease In Dogs And Cats
Pets Score Company Perks As The ‘New Dependents’
Why Is My Cat Rubbing His Face In Ants?
Natural Cure For Hyperthyroidism In Cats Including How To Switch Him/Her To A Raw Food Diet
Ultimate Resource For Cat Lovers
FDA Approves First-Ever Arthritis Pain Management Drug For Cats
Ultimate Resource On Duke of York’s Prince Andrew And His Sex Scandal
Walmart Filings Reveal Plans To Create Cryptocurrency, NFTs
Bitcoin’s Dominance of Crypto Payments Is Starting To Erode
T-Mobile Says Hackers Stole Data On About 37 Million Customers
Jack Dorsey Announces Bitcoin Legal Defense Fund
More Than 100 Millionaires Signed An Open Letter Asking To Be Taxed More Heavily
Federal Regulator Says Credit Unions Can Partner With Crypto Providers
What’s Behind The Fascination With Smash-And-Grab Shoplifting?
Train Robberies Are A Problem In Los Angeles, And No One Agrees On How To Stop Them
US Stocks Historically Deliver Strong Gains In Fed Hike Cycles (GotBitcoin)
Ian Alexander Jr., Only Child of Regina King, Dies At Age 26
Amazon Ends Its Charity Donation Program Amazonsmile After Other Cost-Cutting Efforts
Indexing Is Coming To Crypto Funds Via Decentralized Exchanges
Doctors Show Implicit Bias Towards Black Patients
Darkmail Pushes Privacy Into The Hands Of NSA-Weary Customers
3D Printing Make Anything From Candy Bars To Hand Guns
Stealing The Blood Of The Young May Make You More Youthful
Henrietta Lacks And Her Remarkable Cells Will Finally See Some Payback
AL_A Wins Approval For World’s First Magnetized Fusion Power Plant
Want To Be Rich? Bitcoin’s Limited Supply Cap Means You Only Need 0.01 BTC
Smart Money Is Buying Bitcoin Dip. Stocks, Not So Much
McDonald’s Jumps On Bitcoin Memewagon, Crypto Twitter Responds
America COMPETES Act Would Be Disastrous For Bitcoin Cryptocurrency And More
Lyn Alden On Bitcoin, Inflation And The Potential Coming Energy Shock
Inflation And A Tale of Cantillionaires
El Salvador Plans Bill To Adopt Bitcoin As Legal Tender
Miami Mayor Says City Employees Should Be Able To Take Their Salaries In Bitcoin
Vast Troves of Classified Info Undermine National Security, Spy Chief Says
BREAKING: Arizona State Senator Introduces Bill To Make Bitcoin Legal Tender
San Francisco’s Historic Surveillance Law May Get Watered Down
How Bitcoin Contributions Funded A $1.4M Solar Installation In Zimbabwe
California Lawmaker Says National Privacy Law Is a Priority
The Pandemic Turbocharged Online Privacy Concerns
How To Protect Your Online Privacy While Working From Home
Researchers Use GPU Fingerprinting To Track Users Online
Japan’s $1 Trillion Crypto Market May Ease Onerous Listing Rules
Ultimate Resource On A Weak / Strong Dollar’s Impact On Bitcoin
Fed Money Printer Goes Into Reverse (Quantitative Tightening): What Does It Mean For Crypto?
Crypto Market Is Closer To A Bottom Than Stocks (#GotBitcoin)
When World’s Central Banks Get It Wrong, Guess Who Pays The Price😂😹🤣 (#GotBitcoin)
“Better Days Ahead With Crypto Deleveraging Coming To An End” — Joker
Crypto Funds Have Seen Record Investment Inflow In Recent Weeks
Bitcoin’s Epic Run Is Winning More Attention On Wall Street
Ultimate Resource For Crypto Mergers And Acquisitions (M&A) (#GotBitcoin)
Why Wall Street Is Literally Salivating Over Bitcoin
Nasdaq-Listed MicroStrategy And Others Wary Of Looming Dollar Inflation, Turns To Bitcoin And Gold
Bitcoin For Corporations | Michael Saylor | Bitcoin Corporate Strategy
Ultimate Resource On Myanmar’s Involvement With Crypto-Currencies
‘I Cry Every Day’: Olympic Athletes Slam Food, COVID Tests And Conditions In Beijing
Does Your Baby’s Food Contain Toxic Metals? Here’s What Our Investigation Found
Ultimate Resource For Pro-Crypto Lobbying And Non-Profit Organizations
Ultimate Resource On BlockFi, Celsius And Nexo
Petition Calling For Resignation Of U.S. Securities/Exchange Commission Chair Gary Gensler
100 Million Americans Can Legally Bet on the Super Bowl. A Spot Bitcoin ETF? Forget About it!
Green Finance Isn’t Going Where It’s Needed
Shedding Some Light On The Murky World Of ESG Metrics
SEC Targets Greenwashers To Bring Law And Order To ESG
BlackRock (Assets Under Management $7.4 Trillion) CEO: Bitcoin Has Caught Our Attention
Canada’s Major Banks Go Offline In Mysterious (Bank Run?) Hours-Long Outage (#GotBitcoin)
On-Chain Data: A Framework To Evaluate Bitcoin
On Its 14th Birthday, Bitcoin’s 1,690,706,971% Gain Looks Kind of… Well Insane
The Most Important Health Metric Is Now At Your Fingertips
American Bargain Hunters Flock To A New Online Platform Forged In China
Why We Should Welcome Another Crypto Winter
Traders Prefer Gold, Fiat Safe Havens Over Bitcoin As Russia Goes To War
Music Distributor DistroKid Raises Money At $1.3 Billion Valuation
Nas Selling Rights To Two Songs Via Crypto Music Startup Royal
Ultimate Resource On Music Catalog Deals
Ultimate Resource On Music And NFTs And The Implications For The Entertainment Industry
Lead And Cadmium Could Be In Your Dark Chocolate
Catawba, Native-American Tribe Approves First Digital Economic Zone In The United States
The Miracle Of Blockchain’s Triple Entry Accounting
How And Why To Stimulate Your Vagus Nerve!
Housing Boom Brings A Shortage Of Land To Build New Homes
Biden Lays Out His Blueprint For Fair Housing
No Grave Dancing For Sam Zell Now. He’s Paying Up For Hot Properties
Cracks In The Housing Market Are Starting To Show
Ever-Growing Needs Strain U.S. Food Bank Operations
Food Pantry Helps Columbia Students Struggling To Pay Bills
Food Insecurity Driven By Climate Change Has Central Americans Fleeing To The U.S.
Housing Insecurity Is Now A Concern In Addition To Food Insecurity
Families Face Massive Food Insecurity Levels
US Troops Going Hungry (Food Insecurity) Is A National Disgrace
Everything You Should Know About Community Fridges, From Volunteering To Starting Your Own
Russia’s Independent Journalists Including Those Who Revealed The Pandora Papers Need Your Help
10 Women Who Used Crypto To Make A Difference In 2021
Happy International Women’s Day! Leaders Share Their Experiences In Crypto
Dollar On Course For Worst Performance In Over A Decade (#GotBitcoin)
Juice The Stock Market And Destroy The Dollar!! (#GotBitcoin)
Unusual Side Hustles You May Not Have Thought Of
Ultimate Resource On Global Inflation And Rising Interest Rates (#GotBitcoin)
The Fed Is Setting The Stage For Hyper-Inflation Of The Dollar (#GotBitcoin)
An Antidote To Inflation? ‘Buy Nothing’ Groups Gain Popularity
Why Is Bitcoin Dropping If It’s An ‘Inflation Hedge’?
Lyn Alden Talks Bitcoin, Inflation And The Potential Coming Energy Shock
Ultimate Resource On How Black Families Can Fight Against Rising Inflation (#GotBitcoin)
What The Fed’s Rate Hike Means For Inflation, Housing, Crypto And Stocks
Egyptians Buy Bitcoin Despite Prohibitive New Banking Laws
Archaeologists Uncover Five Tombs In Egypt’s Saqqara Necropolis
History of Alchemy From Ancient Egypt To Modern Times
Former World Bank Chief Didn’t Act On Warnings Of Sexual Harassment
Does Your Hospital or Doctor Have A Financial Relationship With Big Pharma?
Ultimate Resource Covering The Crisis Taking Place In The Nickel Market
Apple Along With Meta And Secret Service Agents Fooled By Law Enforcement Impersonators
Handy Tech That Can Support Your Fitness Goals
How To Naturally Increase Your White Blood Cell Count
Ultimate Source For Russians Oligarchs And The Impact Of Sanctions On Them
Ultimate Source For Bitcoin Price Manipulation By Wall Street
Russia, Sri Lanka And Lebanon’s Defaults Could Be The First Of Many (#GotBitcoin)
Will Community Group Buying Work In The US?
Building And Running Businesses In The ‘Spirit Of Bitcoin’
What Is The Mysterious Liver Disease Hurting (And Killing) Children?
Citigroup Trader Is Scapegoat For Flash Crash In European Stocks (#GotBitcoin)
Bird Flu Outbreak Approaches Worst Ever In U.S. With 37 Million Animals Dead
Financial Inequality Grouped By Race For Blacks, Whites And Hispanics
How Black Businesses Can Prosper From Targeting A Trillion-Dollar Black Culture Market (#GotBitcoin)
Ultimate Resource For Central Bank Digital Currencies (#GotBitcoin) Page#2
Meet The Crypto Angel Investor Running For Congress In Nevada (#GotBitcoin?)
Introducing BTCPay Vault – Use Any Hardware Wallet With BTCPay And Its Full Node (#GotBitcoin?)
How Not To Lose Your Coins In 2020: Alternative Recovery Methods (#GotBitcoin?)
H.R.5635 – Virtual Currency Tax Fairness Act of 2020 ($200.00 Limit) 116th Congress (2019-2020)
Adam Back On Satoshi Emails, Privacy Concerns And Bitcoin’s Early Days
The Prospect of Using Bitcoin To Build A New International Monetary System Is Getting Real
How To Raise Funds For Australia Wildfire Relief Efforts (Using Bitcoin And/Or Fiat )
Former Regulator Known As ‘Crypto Dad’ To Launch Digital-Dollar Think Tank (#GotBitcoin?)
Currency ‘Cold War’ Takes Center Stage At Pre-Davos Crypto Confab (#GotBitcoin?)
A Blockchain-Secured Home Security Camera Won Innovation Awards At CES 2020 Las Vegas
Bitcoin’s Had A Sensational 11 Years (#GotBitcoin?)
Sergey Nazarov And The Creation Of A Decentralized Network Of Oracles
Google Suspends MetaMask From Its Play App Store, Citing “Deceptive Services”
Christmas Shopping: Where To Buy With Crypto This Festive Season
At 8,990,000% Gains, Bitcoin Dwarfs All Other Investments This Decade
Coinbase CEO Armstrong Wins Patent For Tech Allowing Users To Email Bitcoin
Bitcoin Has Got Society To Think About The Nature Of Money
How DeFi Goes Mainstream In 2020: Focus On Usability (#GotBitcoin?)
Dissidents And Activists Have A Lot To Gain From Bitcoin, If Only They Knew It (#GotBitcoin?)
At A Refugee Camp In Iraq, A 16-Year-Old Syrian Is Teaching Crypto Basics
Bitclub Scheme Busted In The US, Promising High Returns From Mining
Bitcoin Advertised On French National TV
Germany: New Proposed Law Would Legalize Banks Holding Bitcoin
How To Earn And Spend Bitcoin On Black Friday 2019
The Ultimate List of Bitcoin Developments And Accomplishments
Charities Put A Bitcoin Twist On Giving Tuesday
Family Offices Finally Accept The Benefits of Investing In Bitcoin
An Army Of Bitcoin Devs Is Battle-Testing Upgrades To Privacy And Scaling
Bitcoin ‘Carry Trade’ Can Net Annual Gains With Little Risk, Says PlanB
Max Keiser: Bitcoin’s ‘Self-Settlement’ Is A Revolution Against Dollar
Blockchain Can And Will Replace The IRS
China Seizes The Blockchain Opportunity. How Should The US Respond? (#GotBitcoin?)
Jack Dorsey: You Can Buy A Fraction Of Berkshire Stock Or ‘Stack Sats’
Bitcoin Price Skyrockets $500 In Minutes As Bakkt BTC Contracts Hit Highs
Bitcoin’s Irreversibility Challenges International Private Law: Legal Scholar
Bitcoin Has Already Reached 40% Of Average Fiat Currency Lifespan
Yes, Even Bitcoin HODLers Can Lose Money In The Long-Term: Here’s How (#GotBitcoin?)
Unicef To Accept Donations In Bitcoin (#GotBitcoin?)
Former Prosecutor Asked To “Shut Down Bitcoin” And Is Now Face Of Crypto VC Investing (#GotBitcoin?)
Switzerland’s ‘Crypto Valley’ Is Bringing Blockchain To Zurich
Next Bitcoin Halving May Not Lead To Bull Market, Says Bitmain CEO
Bitcoin Developer Amir Taaki, “We Can Crash National Economies” (#GotBitcoin?)
Veteran Crypto And Stocks Trader Shares 6 Ways To Invest And Get Rich
Is Chainlink Blazing A Trail Independent Of Bitcoin?
Nearly $10 Billion In BTC Is Held In Wallets Of 8 Crypto Exchanges (#GotBitcoin?)
SEC Enters Settlement Talks With Alleged Fraudulent Firm Veritaseum (#GotBitcoin?)
Blockstream’s Samson Mow: Bitcoin’s Block Size Already ‘Too Big’
Attorneys Seek Bank Of Ireland Execs’ Testimony Against OneCoin Scammer (#GotBitcoin?)
OpenLibra Plans To Launch Permissionless Fork Of Facebook’s Stablecoin (#GotBitcoin?)
Tiny $217 Options Trade On Bitcoin Blockchain Could Be Wall Street’s Death Knell (#GotBitcoin?)
Class Action Accuses Tether And Bitfinex Of Market Manipulation (#GotBitcoin?)
Sharia Goldbugs: How ISIS Created A Currency For World Domination (#GotBitcoin?)
Bitcoin Eyes Demand As Hong Kong Protestors Announce Bank Run (#GotBitcoin?)
How To Securely Transfer Crypto To Your Heirs
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
Crypto News From The Spanish-Speaking World (#GotBitcoin?)
Financial Services Giant Morningstar To Offer Ratings For Crypto Assets (#GotBitcoin?)
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
The Original Sins Of Cryptocurrencies (#GotBitcoin?)
Bitcoin Is The Fraud? JPMorgan Metals Desk Fixed Gold Prices For Years (#GotBitcoin?)
Israeli Startup That Allows Offline Crypto Transactions Secures $4M (#GotBitcoin?)
[PSA] Non-genuine Trezor One Devices Spotted (#GotBitcoin?)
Bitcoin Stronger Than Ever But No One Seems To Care: Google Trends (#GotBitcoin?)
First-Ever SEC-Qualified Token Offering In US Raises $23 Million (#GotBitcoin?)
You Can Now Prove A Whole Blockchain With One Math Problem – Really
Crypto Mining Supply Fails To Meet Market Demand In Q2: TokenInsight
$2 Billion Lost In Mt. Gox Bitcoin Hack Can Be Recovered, Lawyer Claims (#GotBitcoin?)
Fed Chair Says Agency Monitoring Crypto But Not Developing Its Own (#GotBitcoin?)
Wesley Snipes Is Launching A Tokenized $25 Million Movie Fund (#GotBitcoin?)
Mystery 94K BTC Transaction Becomes Richest Non-Exchange Address (#GotBitcoin?)
A Crypto Fix For A Broken International Monetary System (#GotBitcoin?)
Four Out Of Five Top Bitcoin QR Code Generators Are Scams: Report (#GotBitcoin?)
Waves Platform And The Abyss To Jointly Launch Blockchain-Based Games Marketplace (#GotBitcoin?)
Bitmain Ramps Up Power And Efficiency With New Bitcoin Mining Machine (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Miss Finland: Bitcoin’s Risk Keeps Most Women Away From Cryptocurrency (#GotBitcoin?)
Artist Akon Loves BTC And Says, “It’s Controlled By The People” (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Co-Founder Of LinkedIn Presents Crypto Rap Video: Hamilton Vs. Satoshi (#GotBitcoin?)
Crypto Insurance Market To Grow, Lloyd’s Of London And Aon To Lead (#GotBitcoin?)
No ‘AltSeason’ Until Bitcoin Breaks $20K, Says Hedge Fund Manager (#GotBitcoin?)
NSA Working To Develop Quantum-Resistant Cryptocurrency: Report (#GotBitcoin?)
Custody Provider Legacy Trust Launches Crypto Pension Plan (#GotBitcoin?)
Vaneck, SolidX To Offer Limited Bitcoin ETF For Institutions Via Exemption (#GotBitcoin?)
Russell Okung: From NFL Superstar To Bitcoin Educator In 2 Years (#GotBitcoin?)
Bitcoin Miners Made $14 Billion To Date Securing The Network (#GotBitcoin?)
Why Does Amazon Want To Hire Blockchain Experts For Its Ads Division?
Argentina’s Economy Is In A Technical Default (#GotBitcoin?)
Blockchain-Based Fractional Ownership Used To Sell High-End Art (#GotBitcoin?)
Portugal Tax Authority: Bitcoin Trading And Payments Are Tax-Free (#GotBitcoin?)
Bitcoin ‘Failed Safe Haven Test’ After 7% Drop, Peter Schiff Gloats (#GotBitcoin?)
Bitcoin Dev Reveals Multisig UI Teaser For Hardware Wallets, Full Nodes (#GotBitcoin?)
Bitcoin Price: $10K Holds For Now As 50% Of CME Futures Set To Expire (#GotBitcoin?)
Bitcoin Realized Market Cap Hits $100 Billion For The First Time (#GotBitcoin?)
Stablecoins Begin To Look Beyond The Dollar (#GotBitcoin?)
Bank Of England Governor: Libra-Like Currency Could Replace US Dollar (#GotBitcoin?)
Binance Reveals ‘Venus’ — Its Own Project To Rival Facebook’s Libra (#GotBitcoin?)
The Real Benefits Of Blockchain Are Here. They’re Being Ignored (#GotBitcoin?)
CommBank Develops Blockchain Market To Boost Biodiversity (#GotBitcoin?)
SEC Approves Blockchain Tech Startup Securitize To Record Stock Transfers (#GotBitcoin?)
SegWit Creator Introduces New Language For Bitcoin Smart Contracts (#GotBitcoin?)
You Can Now Earn Bitcoin Rewards For Postmates Purchases (#GotBitcoin?)
Bitcoin Price ‘Will Struggle’ In Big Financial Crisis, Says Investor (#GotBitcoin?)
Fidelity Charitable Received Over $100M In Crypto Donations Since 2015 (#GotBitcoin?)
Would Blockchain Better Protect User Data Than FaceApp? Experts Answer (#GotBitcoin?)
Just The Existence Of Bitcoin Impacts Monetary Policy (#GotBitcoin?)
What Are The Biggest Alleged Crypto Heists And How Much Was Stolen? (#GotBitcoin?)
IRS To Cryptocurrency Owners: Come Clean, Or Else!
Coinbase Accidentally Saves Unencrypted Passwords Of 3,420 Customers (#GotBitcoin?)
Bitcoin Is A ‘Chaos Hedge, Or Schmuck Insurance‘ (#GotBitcoin?)
Bakkt Announces September 23 Launch Of Futures And Custody
Coinbase CEO: Institutions Depositing $200-400M Into Crypto Per Week (#GotBitcoin?)
Researchers Find Monero Mining Malware That Hides From Task Manager (#GotBitcoin?)
Crypto Dusting Attack Affects Nearly 300,000 Addresses (#GotBitcoin?)
A Case For Bitcoin As Recession Hedge In A Diversified Investment Portfolio (#GotBitcoin?)
SEC Guidance Gives Ammo To Lawsuit Claiming XRP Is Unregistered Security (#GotBitcoin?)
15 Countries To Develop Crypto Transaction Tracking System: Report (#GotBitcoin?)
US Department Of Commerce Offering 6-Figure Salary To Crypto Expert (#GotBitcoin?)
Mastercard Is Building A Team To Develop Crypto, Wallet Projects (#GotBitcoin?)
Canadian Bitcoin Educator Scams The Scammer And Donates Proceeds (#GotBitcoin?)
Amazon Wants To Build A Blockchain For Ads, New Job Listing Shows (#GotBitcoin?)
Shield Bitcoin Wallets From Theft Via Time Delay (#GotBitcoin?)
Blockstream Launches Bitcoin Mining Farm With Fidelity As Early Customer (#GotBitcoin?)
Commerzbank Tests Blockchain Machine To Machine Payments With Daimler (#GotBitcoin?)
Man Takes Bitcoin Miner Seller To Tribunal Over Electricity Bill And Wins (#GotBitcoin?)
Bitcoin’s Computing Power Sets Record As Over 100K New Miners Go Online (#GotBitcoin?)
Walmart Coin And Libra Perform Major Public Relations For Bitcoin (#GotBitcoin?)
Judge Says Buying Bitcoin Via Credit Card Not Necessarily A Cash Advance (#GotBitcoin?)
Poll: If You’re A Stockowner Or Crypto-Currency Holder. What Will You Do When The Recession Comes?
1 In 5 Crypto Holders Are Women, New Report Reveals (#GotBitcoin?)
Beating Bakkt, Ledgerx Is First To Launch ‘Physical’ Bitcoin Futures In Us (#GotBitcoin?)
Facebook Warns Investors That Libra Stablecoin May Never Launch (#GotBitcoin?)
Government Money Printing Is ‘Rocket Fuel’ For Bitcoin (#GotBitcoin?)
Bitcoin-Friendly Square Cash App Stock Price Up 56% In 2019 (#GotBitcoin?)
Safeway Shoppers Can Now Get Bitcoin Back As Change At 894 US Stores (#GotBitcoin?)
TD Ameritrade CEO: There’s ‘Heightened Interest Again’ With Bitcoin (#GotBitcoin?)
Venezuela Sets New Bitcoin Volume Record Thanks To 10,000,000% Inflation (#GotBitcoin?)
Newegg Adds Bitcoin Payment Option To 73 More Countries (#GotBitcoin?)
China’s Schizophrenic Relationship With Bitcoin (#GotBitcoin?)
More Companies Build Products Around Crypto Hardware Wallets (#GotBitcoin?)
Bakkt Is Scheduled To Start Testing Its Bitcoin Futures Contracts Today (#GotBitcoin?)
Bitcoin Network Now 8 Times More Powerful Than It Was At $20K Price (#GotBitcoin?)
Crypto Exchange BitMEX Under Investigation By CFTC: Bloomberg (#GotBitcoin?)
“Bitcoin An ‘Unstoppable Force,” Says US Congressman At Crypto Hearing (#GotBitcoin?)
Bitcoin Network Is Moving $3 Billion Daily, Up 210% Since April (#GotBitcoin?)
Cryptocurrency Startups Get Partial Green Light From Washington
Fundstrat’s Tom Lee: Bitcoin Pullback Is Healthy, Fewer Searches Аre Good (#GotBitcoin?)
Bitcoin Lightning Nodes Are Snatching Funds From Bad Actors (#GotBitcoin?)
The Provident Bank Now Offers Deposit Services For Crypto-Related Entities (#GotBitcoin?)
Bitcoin Could Help Stop News Censorship From Space (#GotBitcoin?)
US Sanctions On Iran Crypto Mining — Inevitable Or Impossible? (#GotBitcoin?)
US Lawmaker Reintroduces ‘Safe Harbor’ Crypto Tax Bill In Congress (#GotBitcoin?)
EU Central Bank Won’t Add Bitcoin To Reserves — Says It’s Not A Currency (#GotBitcoin?)
The Miami Dolphins Now Accept Bitcoin And Litecoin Crypt-Currency Payments (#GotBitcoin?)
Trump Bashes Bitcoin And Alt-Right Is Mad As Hell (#GotBitcoin?)
Goldman Sachs Ramps Up Development Of New Secret Crypto Project (#GotBitcoin?)
Blockchain And AI Bond, Explained (#GotBitcoin?)
Grayscale Bitcoin Trust Outperformed Indexes In First Half Of 2019 (#GotBitcoin?)
XRP Is The Worst Performing Major Crypto Of 2019 (GotBitcoin?)
Bitcoin Back Near $12K As BTC Shorters Lose $44 Million In One Morning (#GotBitcoin?)
As Deutsche Bank Axes 18K Jobs, Bitcoin Offers A ‘Plan ฿”: VanEck Exec (#GotBitcoin?)
Argentina Drives Global LocalBitcoins Volume To Highest Since November (#GotBitcoin?)
‘I Would Buy’ Bitcoin If Growth Continues — Investment Legend Mobius (#GotBitcoin?)
Lawmakers Push For New Bitcoin Rules (#GotBitcoin?)
Facebook’s Libra Is Bad For African Americans (#GotBitcoin?)
Crypto Firm Charity Announces Alliance To Support Feminine Health (#GotBitcoin?)
Canadian Startup Wants To Upgrade Millions Of ATMs To Sell Bitcoin (#GotBitcoin?)
Trump Says US ‘Should Match’ China’s Money Printing Game (#GotBitcoin?)
Casa Launches Lightning Node Mobile App For Bitcoin Newbies (#GotBitcoin?)
Bitcoin Rally Fuels Market In Crypto Derivatives (#GotBitcoin?)
World’s First Zero-Fiat ‘Bitcoin Bond’ Now Available On Bloomberg Terminal (#GotBitcoin?)
Buying Bitcoin Has Been Profitable 98.2% Of The Days Since Creation (#GotBitcoin?)
Another Crypto Exchange Receives License For Crypto Futures
From ‘Ponzi’ To ‘We’re Working On It’ — BIS Chief Reverses Stance On Crypto (#GotBitcoin?)
These Are The Cities Googling ‘Bitcoin’ As Interest Hits 17-Month High (#GotBitcoin?)
Venezuelan Explains How Bitcoin Saves His Family (#GotBitcoin?)
Quantum Computing Vs. Blockchain: Impact On Cryptography
This Fund Is Riding Bitcoin To Top (#GotBitcoin?)
Bitcoin’s Surge Leaves Smaller Digital Currencies In The Dust (#GotBitcoin?)
Bitcoin Exchange Hits $1 Trillion In Trading Volume (#GotBitcoin?)
Bitcoin Breaks $200 Billion Market Cap For The First Time In 17 Months (#GotBitcoin?)
You Can Now Make State Tax Payments In Bitcoin (#GotBitcoin?)
Religious Organizations Make Ideal Places To Mine Bitcoin (#GotBitcoin?)
Goldman Sacs And JP Morgan Chase Finally Concede To Crypto-Currencies (#GotBitcoin?)
Bitcoin Heading For Fifth Month Of Gains Despite Price Correction (#GotBitcoin?)
Breez Reveals Lightning-Powered Bitcoin Payments App For IPhone (#GotBitcoin?)
Big Four Auditing Firm PwC Releases Cryptocurrency Auditing Software (#GotBitcoin?)
Amazon-Owned Twitch Quietly Brings Back Bitcoin Payments (#GotBitcoin?)
JPMorgan Will Pilot ‘JPM Coin’ Stablecoin By End Of 2019: Report (#GotBitcoin?)
Is There A Big Short In Bitcoin? (#GotBitcoin?)
Coinbase Hit With Outage As Bitcoin Price Drops $1.8K In 15 Minutes
Samourai Wallet Releases Privacy-Enhancing CoinJoin Feature (#GotBitcoin?)
There Are Now More Than 5,000 Bitcoin ATMs Around The World (#GotBitcoin?)
You Can Now Get Bitcoin Rewards When Booking At Hotels.Com (#GotBitcoin?)
North America’s Largest Solar Bitcoin Mining Farm Coming To California (#GotBitcoin?)
Bitcoin On Track For Best Second Quarter Price Gain On Record (#GotBitcoin?)
Bitcoin Hash Rate Climbs To New Record High Boosting Network Security (#GotBitcoin?)
Bitcoin Exceeds 1Million Active Addresses While Coinbase Custodies $1.3B In Assets
Why Bitcoin’s Price Suddenly Surged Back $5K (#GotBitcoin?)
Zebpay Becomes First Exchange To Add Lightning Payments For All Users (#GotBitcoin?)
Coinbase’s New Customer Incentive: Interest Payments, With A Crypto Twist (#GotBitcoin?)
The Best Bitcoin Debit (Cashback) Cards Of 2019 (#GotBitcoin?)
Real Estate Brokerages Now Accepting Bitcoin (#GotBitcoin?)
Ernst & Young Introduces Tax Tool For Reporting Cryptocurrencies (#GotBitcoin?)
Recession Is Looming, or Not. Here’s How To Know (#GotBitcoin?)
How Will Bitcoin Behave During A Recession? (#GotBitcoin?)
Many U.S. Financial Officers Think a Recession Will Hit Next Year (#GotBitcoin?)
Definite Signs of An Imminent Recession (#GotBitcoin?)
What A Recession Could Mean for Women’s Unemployment (#GotBitcoin?)
Investors Run Out of Options As Bitcoin, Stocks, Bonds, Oil Cave To Recession Fears (#GotBitcoin?)
Goldman Is Looking To Reduce “Marcus” Lending Goal On Credit (Recession) Caution (#GotBitcoin?)
Your Questions And Comments Are Greatly Appreciated.
Monty H. & Carolyn A.
Go back



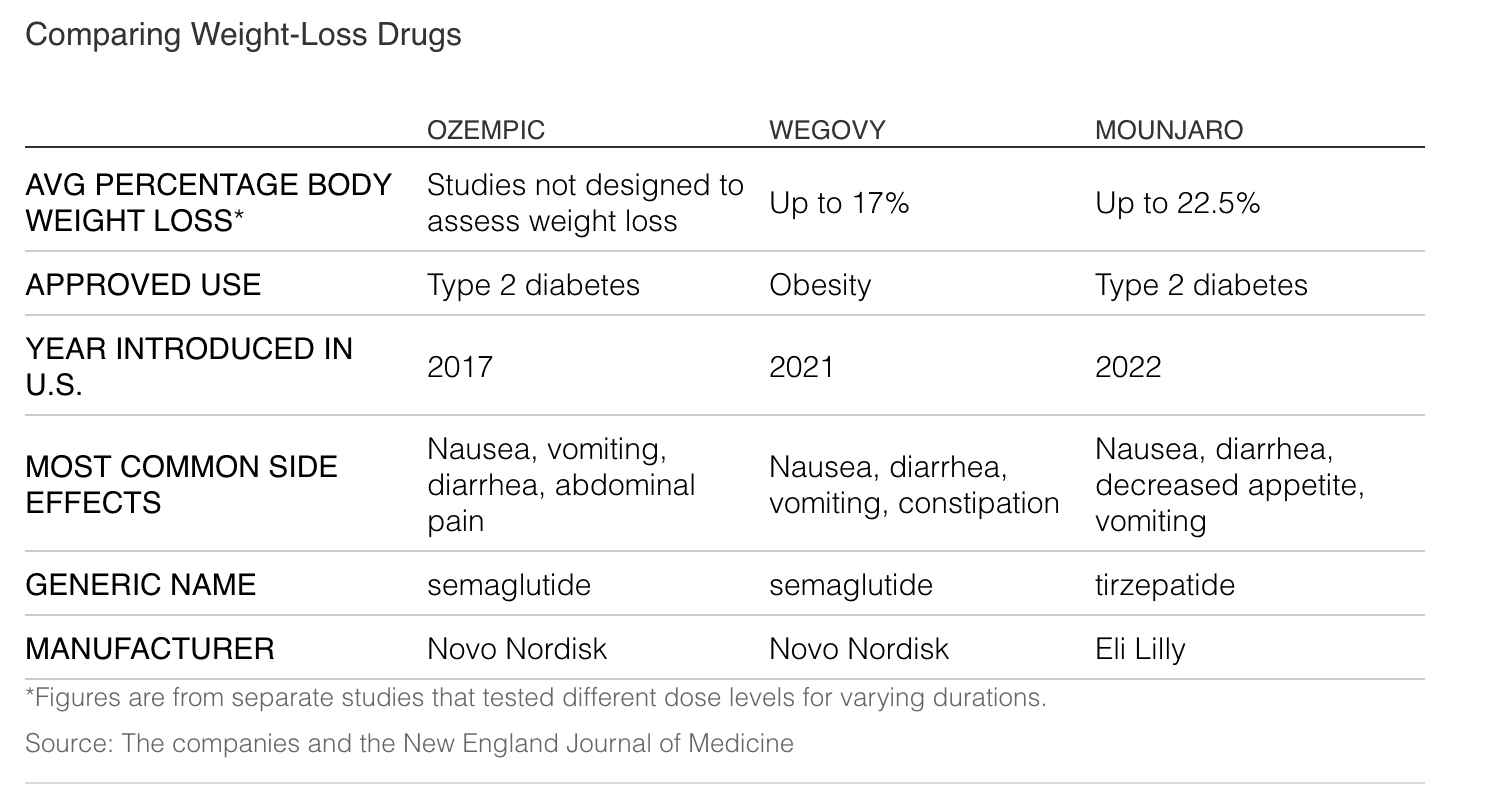





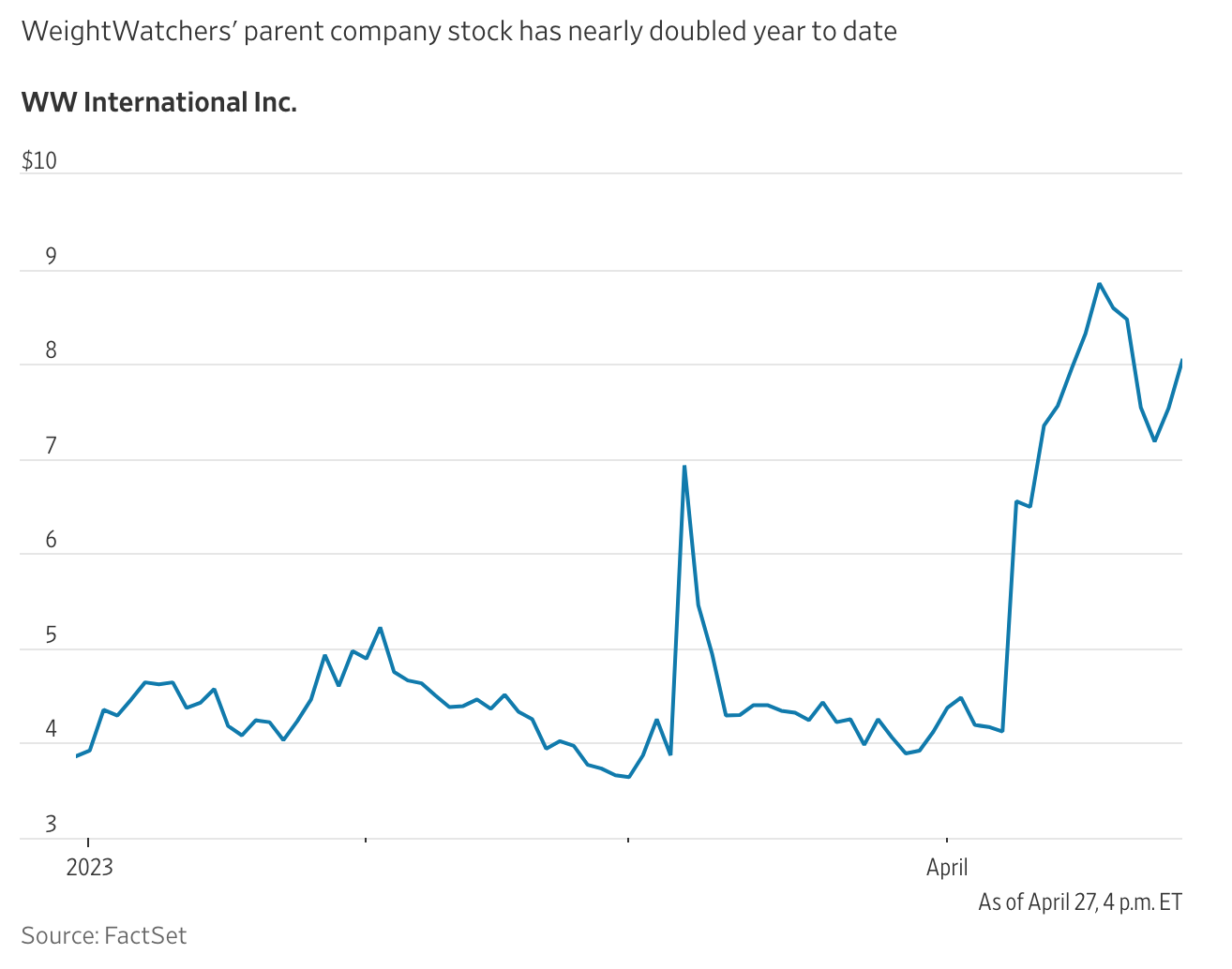

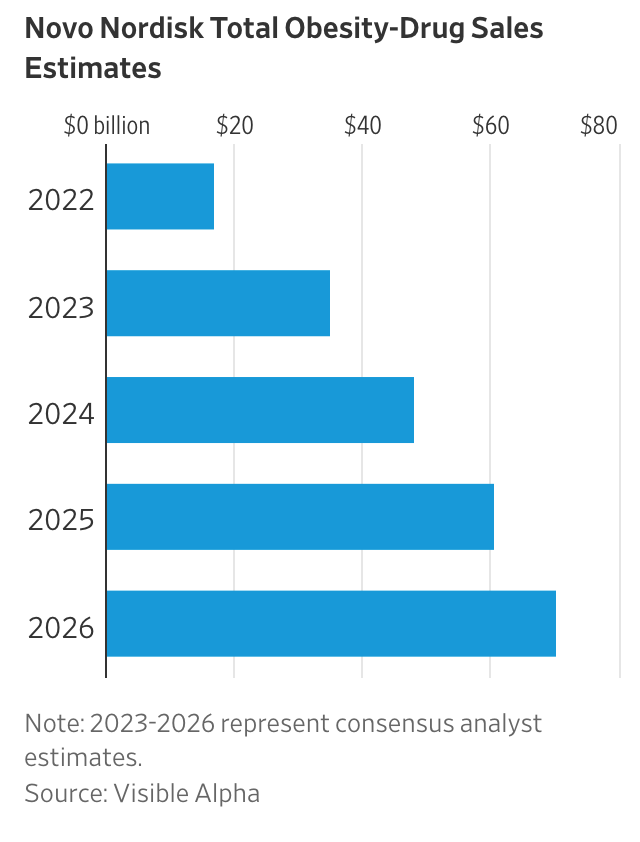



Leave a Reply
You must be logged in to post a comment.