Bitcoin Community Leaders Join Longevity Movement
Crypto leaders are obsessed with life extension. Here’s why. Bitcoin Community Leaders Join Longevity Movement

Related:
The Next Fountain-of-Youth Craze? Peptide Injections
Bitcoiners Are Now Into Fasting. Read This Article To Find Out Why
The Definitive Guide On Fasting, Autophagy, And Caloric Restriction
Fasting 101 Rebooting The Body’s Hard Drive Rejuvenating, Life-Extending And Removes Deadly Toxins
What Is Dopamine Fasting? Meet The Dangerous Fad Among Silicon Valley’s Tech Geniuses
Ultimate Resource For News, Breakthroughs And Innovations In Healthcare
Crispr, Eugenics And “Three Generations Of Imbeciles Is Enough.”
Ethereum co-founder Vitalik Buterin is on a mission to make humans immortal. Buterin, 27, proposes the idea that “aging is an engineering problem.” (Start At: 2:28:50)
“I have been gratified since the beginning of blockchain to see the enormous fanbase that I and the longevity movement have in there.”
Miller, alongside his engineering hall-of-fame accomplishments, is a senior research fellow at the Foresight Institute, a not-for-profit founded in 1986 with the aim of “advancing technology for the long-term benefit of life.”
“I’m very much involved in this new world of crypto commerce, often referred to as the blockchain sector,” he says. “I’m very hopeful about that as creating an ecosystem in which secure software will dominate because insecure software results in massive losses quickly, with no recourse.”
Srinivasan’s Twitter bio describes his vision as: “Immutable money, infinite frontier, eternal life. #Bitcoin.” Srinivasan states that “the ultimate purpose of technology is to eliminate mortality” and “life extension is the most important thing we can invent.”
Blockchain communities are clearly excited about longevity. But what does cryptocurrency have to do with life extension, and where might this future be headed?
If we’re being more open minded about accepting new weird ideas, can I suggest anti-aging research? Aging is a humanitarian disaster that kills as many people as WW2 every two years and even before killing debilitates people and burdens social systems and families. Let’s end it.
— vitalik.eth (@VitalikButerin) March 30, 2020
Crypto People Are Funding Longevity Research
Crypto philanthropists are donating significant wealth to this area, which is typically difficult to garner mainstream support for. They may be the only people on the planet optimistic enough to fund tech that currently only exists in sci-fi novels.
According to Buterin, longevity is a battle worth fighting for. Buterin donated $25 million in SHIB cryptocurrency tokens to the Future of Life Institute in June 2021 and has donated over $350,000 to the SENS Research Foundation to “reimagine ageing”.
He discussed the topic in recent podcast interviews with the likes of Lex Fridman and Tim Ferriss saying that “life extension is definitely really important to me.”
“I think I hope to see the concept of seeing your parents and grandparents die just slowly disappear from the public consciousness as a thing that happens over the course of half a century.”
Buterin has emphasized his adherence to the moral philosophy of effective altruism. This value, known to trans-humanists as the moral urgency of saving lives, is perhaps what motivated his donations of dog coins to both COVID-19 relief in India and life-extension.
“Just even the process of aging turning into something that just becomes reversible and it being a regular thing for people to live one and a half, two centuries and then go even further from there,” Buterin states.
Pinned in Buterin’s Twitter is an essay called “The Fable of the Dragon Tyrant” by professor Nick Bostrom, director of the Future of Humanity Institute.
It argues that allowing death from old age is unethical. If you view aging as a disease, the urgency to support the trans-humanist project also makes sense.
“While we still lack effective and acceptable means for slowing the aging process, we can identify research directions that might lead to the development of such means in the foreseeable future,” states Bostrom.
The key to freeing humanity from the dragon tyrant of aging, is funding. The new riches from crypto are key.
Bitcoin.com founder Roger Ver has already signed up to be cryogenically frozen. “Rather than investing in cryptocurrency stuff, I want to focus on the extreme life extension technologies, because if you die, you can’t enjoy your life anymore,” Ver told Cointelegraph.
He’s so confident in the tech, he even considered being cryogenically frozen as a legitimate alternative to going to prison in 2002.
Roger Ver once considered killing himself and going into cryonic suspension to avoid prison.
Now he plans to spend the next phase of his career — and his fortune — making cryonics a reality. https://t.co/IZ1Ham7NEa
— Cointelegraph (@Cointelegraph) April 28, 2021
DAOs are also taking part in this life extension renewal. “There is a strong overlap of crypto people and longevity people,” Vincent Weisser, core team member at VitaDAO tells Cointelegraph. VitaDAO exceeded its token raise funding target of $490,000 in June 2021.
Now, they are working with popular blockchain crowd-funding platform Gitcoin to include a future funding category for longevity and life extension.
Trans-humanist philanthropy and funding at scale holds the potential to significantly impact longevity research and the trans-humanist project.
What Ts Trans-humanism?
Trans humanism is a loosely defined movement that promotes the use of technology to enhance the human condition. This includes information technology, genetic engineering, and artificial intelligence for radical extension of human lifespan, augmentation of physical and intellectual capacities, space colonization, and super-intelligent machines.
The goal is not just life extension, but “more,” to the point of becoming superhuman. Although the trans-humanist pursuit of post-humanity is often thought of as medical, the gambit of trans-humanist technologies includes economic and social institutional design and cultural development.
Like crypto communities, trans-humanism is grounded in a vision of evolution and individual freedom of choice. In practice, this leads to a sense of personal responsibility for contributing to solutions, such as biohacking or making provisions for being cryogenically frozen and one day hopefully reanimated.
The goal of the trans-humanist project for society is one based on freedom in determining social arrangements, enabled by self-generating systems and spontaneous order. This description of perpetual, open systems is similar to blockchain.
Not everyone thinks eternal life, or the philosophy underpinning it, is a good idea. Political economist Francis Fukuyama calls trans-humanism “the most dangerous idea in the world” and argues it is “a strange libertarian movement” whose “crusaders” want “nothing less than to liberate the human race from its biological constraints.”
He lists the risks of the fraught nature of humankind to want to live forever, the effects on equality between the “haves” and the “have nots,” and that the essence of humanity is mortality. Yet, trans-humanism has a long history in crypto communities.
More Human Than Human
Trans-humanist values are reflected in the ideological underpinnings of blockchain communities around anarchy and autonomy, self-improvement, and a long-term mindset.
Trans-humanist ideas have long existed in the technology communities that pioneered the core tenets of public blockchains. For example, cryptography pioneer Ralph Merkle (inventor of public key distribution and Merkle trees) considered himself a trans-humanist, publishing on such matters as “The Molecular Repair of the Brain.”
Furthermore, there was substantial cross-pollination of ideas between the Cypherpunks mailing list, which discussed ideas on privacy and digital cash throughout the 1990s and 2000s in the lead-up to the invention of Bitcoin in 2008, and the Extropian mailing list.
Extropy is “the extent of a system’s intelligence, information, order, vitality, and capacity for improvement.” According to 1998’s “Principles of Extropy” published by president of the Extropy Institute, Max Moore, extropians are “those who seek to increase extropy.”
The core principles, refined in “The Extropist Manifesto” in 2010, are “endless eXtension,” meaning perpetual growth and progress in all aspects of human endeavor; transcending the restrictions of authoritarianism, surveillance, or social control; overcoming property rights, including IP and money, by sharing knowledge, culture, and resources; intelligence, including independent thinking and personal responsibility; and smart machines, specifically the attainment of “Friendly Artificial Intelligence” that exceeds human ability, through funding and favorable legislation.
Extropians advocate and explore the philosophies of trans-humanism (technological enhancement), extropy (improving the human condition), and the future. Numerous prominent cypherpunks also subscribed to the Extropian mailing list, including co-founders of the cypherpunk movement Timothy C. May and Eric Hughes.
Here’s my conversation with @VitalikButerin, his second time on the podcast. We talk about @Ethereum, @Bitcoin, @Dogecoin, @elonmusk, @ShibainuCoin, @IOHK_Charles, @Cardano, @Chainlink, @0xPolygon, life, immortality, AI, and even speak Russian at the end. https://t.co/Y3cjGuctvM pic.twitter.com/Pu7uwwF6X4
— Lex Fridman (@lexfridman) June 3, 2021
Another active member of the extropians was Hal Finney. Finney was co-developer of the first anonymous remailer, the first person to transact Bitcoin with Satoshi and the first maintainer of the Bitcoin codebase. He was cryogenically frozen when he passed away in the hope of living in the future alongside his wife, Fran, who noted that “Hal liked the present. But he looked towards the future.”
For this community, technologies like digital cash offered a way of long-term thinking about the future of humanity, trans-humanism, and solutions and preventions for cryogenics, outer space, and catastrophic environmental or societal collapse.
The cypherpunks’ interest in extropianism, and vice versa, was concerned with building infrastructure today that would sustain the future of human evolution. In some ways, this makes sense.
In order for one’s cryogenic suspended animation to be paid for, maintained and reversed to wake them up in the far-flung future where science advances to the point where this aspiration is realized, there needs to be an incentive.
In 1994, Wired magazine reported over 27 frozen people (technically 17 frozen heads and 10 entire bodies) at the Alcor Life Extension Foundation, the same company that Roger Ver has signed up with.
“Immortality is mathematical, not mystical,” stated Mike Perry, their overseer. The hope is that Bitcoin will be a resilient long-term incentive for someone to wake up Hal, Fran and other friends. Herein lies the need for long-term blockchain infrastructure, to last as a secure monetary reward until the century when “unfreezing” is possible.
Among the principles of extropianism set out by Moore is “intelligent technology,” meaning technologies that bring beneficial results, including “genetic engineering, life-extending bio-sciences, intelligence intensifiers, smarter interfaces to swifter computers, neural-computer integration, worldwide data networks, virtual reality, intelligent agents, swift electronic communications, artificial intelligence, neuroscience, neural networks, artificial life, off-planet migration, and molecular nanotechnology.”
“Expect to see more life extension, brain-machine interfaces, limb regeneration, curing deafness, bionic sight and more incredible feats” in the next decade, states Srinivasan. Trans-humanists predict an inevitable “singularity,” when technology becomes intelligent, uncontrollable and irreversible, to occur around 2035. Blockchain is part of this technology stack.
Where Trans-Humanism And Crypto Overlap
The lofty, futuristic ideas of trans-humanists depend on resilience and digital infrastructure. This is especially true for the goal of friendly artificial intelligence, which is seen as an enabling condition for rapid development across all other core principles of the project.
Thanks to blockchain technology, and blockchain philanthropy, we are beginning to have the resources to do it.
An immutable “worldwide computer” enables a decentralized autonomous organization to allow our uploaded brain image to automatically coordinate with friendly artificial intelligence in a decentralized, freedom-loving way.
Blockchain’s immutability makes it the perfect long-term infrastructure. Cypherpunks were paranoid about Orwellian dystopias in which authorities would rewrite history to match state propaganda.
The architectural and political decentralization of public blockchains means that no one can tamper with, control or delete the record of history. This makes it the perfect record-keeping infrastructure if we are going to live forever.
This is essential when it comes to your brain image or your Bitcoin balance. In order to document who owns what coins when you are cryogenically frozen and woken up in the next century, you need resilient, long-term, tamper-proof blockchains. The values of independence and immutability are essential to both crypto enthusiasts and trans-humanists. Buterin states:
“It’s great that we have people trying to upload or improve brain scanning. It’s also great that we have people including cryonics, so we could just go to sleep in the freezer and eventually, hopefully, sometime in the future […] anyone who gets cryogenically frozen will be able to wake up.”
Future Making
The combination of trans-humanist philosophy, blockchain technology, community obsession and money enable whole new possibilities.
The trans-humanist-blockchain vision is that we will all be connected, humans and machine intelligence, through decentralized, automatically executing smart contracts and marketplaces.
Blockchains provide a platform infrastructure to enable a host of technologically advanced human-machine futures. One example is a decentralized marketplace for AI, such as SingularityNET by artificial intelligence researcher, trans-humanist, and CEO Ben Goertzel. Here, intelligent computational agents buy, sell and barter over work for digital tokens via a blockchain.
Lifespan News – Vitalik Donates for Longevity https://t.co/vY1Gaz4kp6 pic.twitter.com/X4AUpJmWwx
— Dogelon Warriors (@DogelonWarriors) July 26, 2021
In The Trans-humanism Handbook, Melanie Swan predicts that “crypto cloudminds,” in which mind node peers interact through multicurrency pay channels of digital denominations, will algorithmically enforce good behavior between humans and machines through the privacy and transparency of blockchains.
According to Srinivasan, this could also lead to “cloud cities,” which allow their members to negotiate with other jurisdictions and crowdfund territories in the physical world.
Trans-humanism, like human beings, is only in its early stages of development.
Trans-humanism, with its focus on superhumans and longevity instead of an afterlife, can be viewed as something akin to a religious impulse.
Although many trans-humanists take their worldview to be in opposition to religious outlooks on life, trans-humanism may become the religion of blockchainers. Yet, this doctrine does not come without a clear burden of responsibility.
The Engineer’s Responsibility
While some fear trans-humanism, a core tenet is to ensure that technology produces positive outcomes for humanity. Trans-humanist advocate that the choice to improve human capacities lies with the individual.
Part of the longevity research agenda is figuring out how to measure the risks of friendly artificial intelligence and make it truly friendly to avoid a catastrophe.
Trans-humanist want to avoid “X risk,” which is existential risk to humanity of a hypothetical, global, catastrophic future event that could damage human well-being or destroy human civilization.
This is why colonizing outer space is so logical, as Elon and other crypto enthusiasts are pursuing. The “Extropian Principles, v. 3.0” by Max Moore from 1998 emphasizes this, stating that “migration into space will immensely enlarge the energy and resources accessible to our civilization.”
Of course, smart machines will also help us explore space because they can handle more gravitational force than humans as they enter the orbit of other planets.
To a trans-humanist, the goal of technology is to amplify our abilities and extend human freedoms. “How could we ensure humanity lives forever and life spreads throughout the universe?” asks Weisser from VitaDAO. “It’s all about probabilities and increasing the probability that humanity will survive,” he says.
A long-term mindset treats aging as an engineering problem. Now, it remains to be seen if the intersection of blockchain philanthropy, VitaDAO’s research collective, and other decentralized, trans-humanist pursuits will be cautiously and collectively propelled forwards with the kind of “long-termism” that will benefit humanity. As Buterin states:
“I hope you guys can […] come to my thousandth birthday party.”
Updated: 9-30-2021
$1B Science Fund Seeks Blockchain Projects To Expand Human Lifespan
Longevity Science Foundation accepts crypto donations and aims to use technology to advance healthy human longevity.
Scientists are continuously pursuing ways to lengthen the human lifespan, and blockchain might have been a missing part of the puzzle.
The Longevity Science Foundation, a Swiss entity launched by a consortium of biotech founders, clinicians and leading longevity research institutions, aims to spend more than $1 billion over the next 10 years to find tech-based means to achieve a 120-year human lifespan.
The foundation seeks to fund research, institutions and projects that use blockchain and other next-gen technologies to find new horizons in four critical areas of the field; namely, therapeutics, personalized medicine, artificial intelligence (AI) and predictive diagnostics.
The announcement states that such projects can make a significant difference in people’s lives, even within a five-year timeframe.
Applying theoretical longevity concepts to real-world use is a primary goal for the group. The foundation aims to transform scientific findings and technological advances into clinical treatments and solutions through donations.
“By identifying and funding the most promising and cutting-edge advances, the Foundation seeks to address one of the most pressing issues in the science and applicability of longevity medicine radical inequality in accessing and understanding longevity-focused treatment,” the announcement reads.
The foundation’s contributors, who can also make donations with cryptocurrencies, will get voting rights to have a say in deciding which projects receive funding. A visionary board will pre-select and evaluate potential candidates “for their technical soundness and potential impact on human longevity.”
Joining the Visionary Board of the Foundation is Dr. Alex Zhavoronkov, known for his work on longevity tech. Dr. Zhavoronkov is also an advisor to blockchain medical data marketplace Longenesis, which launched a joint project with the Bitfury Group to establish a blockchain-based consent management system for the healthcare industry.
“The Foundation has created a unique and transparent mechanism for boosting early longevity research worldwide and ensuring mass public participation in decision making,” Dr. Zhavoronkov said.
As for how technology can help healthcare research, LongeVC managing partner Garri Zmudze told Cointelegraph that if paired with AI, blockchain can unlock hundreds of terabytes of unstructured hospital data for further analysis.
Updated: 10-29-2021
The Future Of Longevity Lies With Digital Currency Visionaries
Healthcare researchers in collaboration with crypto innovators might prove that a longer and healthier lifespan is an attainable reality.
The promise of a longer and healthier life has dominated both cultural and medical discourse for centuries. From the first accounts of Nicolas Flamel’s philosopher’s stone granting immortality to the latest superfood that promised to reverse aging, we are captivated by the idea of a longer and more vibrant life.
Encouragingly, the longevity sector has proven that a longer and healthier lifespan isn’t just a magazine headline — it is an attainable reality. Humans will not only live longer but will have more active, healthy and engaged years on this Earth.
Research units from all over the world are making discoveries every day. Medical advancements, fueled by intensive research and experimentation, are changing how scientists, doctors and even cultural commentators think about how long and how well we can live. We are, indeed, now in the era of longevity.
What is driving these incredible breakthroughs? Pure creativity and innovation. The advent of artificial intelligence, genetic engineering and a renewed interest in public health stemming from the COVID-19 pandemic are catalyzing discoveries at an unprecedented rate.
To maximize the impact of these researchers, the future of innovation will need to be collaborative and cooperative. It will take a village to make longevity understandable and accessible to medical professionals and average individuals alike.
Where The Future Lies
The best route forward, in my opinion, is to solicit support from true visionaries. Take, for example, the company Longenesis, which I highlighted in my previous article for Cointelegraph.
Its “blockchain toolkit” applies the most compelling features of distributed ledger technology (DLT) to protect sensitive medical data, ensure secure data sharing between numerous partners and provide a pathway for patients to become stakeholders in medical research by contributing their data and participating in clinical trials.
Longenesis could accomplish so much relatively quickly because it had money and support from the highly futuristic and accepting blockchain community.
Backed by funding from its joint venture partners, Longenesis achieved its mission of creating a game-changing and blockchain-powered approach to unlocking healthcare data.
This is an increasingly common trend: the Iryo Network relied on support from blockchain professionals to create a token that empowers patients to stake their data and receive payment from research institutions in exchange for data access.
The Texas-based EHRData is working toward a similar goal of decentralized data storage and management by creating an electronic health record built on blockchain that patients can control, share and submit to researchers for staked rewards.
These applications were all made possible thanks to blockchain visionaries willing to fund the future of healthcare and longevity.
Early-stage funding can power startups, but it can also support even more foundational innovations by researchers. Genuinely cutting-edge innovation at the laboratory/research stage often requires significant funding for state-of-the-art equipment, materials, operating administration costs, etc., just to get started.
This is a challenge for researchers from smaller institutions, as funding often goes to projects that have access to these resources and, as a result, have already advanced in their discoveries.
For these cases, foundational grants can be instrumental in supporting the high upfront fees associated with researching something intended to change the world.
The longevity sector is rich with organizations looking to find and fund the next big idea, but very few organizations are willing to move beyond traditional funding models to make that happen.
It is difficult to conceive that an industry with innovation embedded in its very core is lagging in terms of new funding sources, but that is a possible reality facing groundbreaking researchers and institutions.
The Time For Futurists And Visionaries
Luckily, the innovation taking place in the digital currencies sector is spilling over into the biotech and longevity spaces. The community of visionaries who transformed the way money and investing works is here to change how humanity experiences life itself.
Back in 2018, Vitalik Buterin, the co-founder of Ethereum, donated $2.4 million in Ether (ETH) to the SENS Research Foundation, a California-based group working to research, develop and promote comprehensive solutions for aging-related diseases.
Buterin has also donated $25 million in Shiba Inu (SHIB) tokens to the Future of Life Institute to fund Ph.D. fellowships focused on ethical Artificial Intelligence applications and millions to the Methuselah Foundation, an organization researching tissue engineering to reverse aging processes.
The new Longevity Science Foundation will be accepting donations in crypto to contribute to its goal of expanding the human lifespan to 120+ years. The Foundation welcomes crypto visionaries, and all donors (crypto donors both included and encouraged), will receive voting rights in the funding selection process.
VitaDAO aims to extend the human lifespan openly and democratically by researching, financing and commercializing longevity therapeutics. Its decentralized structure is similar to that of many decentralized applications (DApps) and other decentralized autonomous organizations (DAOs), building a new and exciting governance structure.
Its genesis auction raised more than $5 million in funding via purchased VITA governance tokens, attracting approximately 400 new members committed to the DAO’s mission.
The crypto visionaries that have made these accomplishments possible are future-focused collaborators who want a front seat for the cutting-edge science of longevity. They are creating a village that will make longevity care possible and accessible to all, but there is a lot of work still to be done.
There is a significant overlap in the challenges of adopting longevity care and the resistance to blockchain technology and digital currency. By lengthening our lifespans, we create a future that requires the adoption of blockchain and digital currency to be successful.
Digital currencies and blockchain technology changed the way the entire world transacts and uses critical services. They are the foundational technologies for a more fair and efficient society. New applications for DLT are being developed every day, and new investors, adopters and appreciators are drawn to the industry because of its constantly evolving nature.
If we can apply that same entrepreneurial spirit and can-do attitude to the longevity sector, our lives will not only be longer but more enriched, meaningful and digitally enabled.
The longevity sector is working to make sure more people live long enough to enjoy a decentralized and transparent world. To do so, the industry desperately needs the brainpower and experience of the innovators who turned a single white paper into a global and unstoppable movement of transparency and equality. This feat was successful once, let’s make it happen again.
Updated: 2-2-2022
Centenarian-Sourced Probiotic Mixed With Dietary Fiber Has Anti-Aging Effects
A probiotic strain isolated from centenarians’ feces combined with dietary fiber showed beneficial effects on brain and gut function and has potential as an aging treatment for the elderly.
Highlights
* A Probiotic Isolated From Centenarians Called Ltl1361 Mixed With Dietary Fiber Alleviates Age-Related Cognitive Impairment And Protects The Brain Of Healthy Aging Mice.
* This Probiotic And Dietary Fiber Combination Also Improves Gut Function And Reduces Inflammation In The Blood And Colon.
* These Findings Suggest The Ltl1361 And Dietary Fiber Mixture As A Novel And Promising Anti-Aging Agent In Humans.
Accumulating evidence indicates that gut microbiota regulates host health. This microorganism population can be manipulated in multiple ways: with probiotics to change the variety and with dietary nutrients or fiber to supply specific compounds.
While studies have focused on the anti-aging effects within a particular disease, the evidence is limited for the anti-aging effects of centenarian-sourced probiotics combined with a dietary fiber complex.
Researchers from Guangxi University in China reveal the anti-aging effects of an isolated probiotic strain from healthy centenarians’ feces mixed with dietary fiber.
With three months of treatment, this combination improved learning and memory ability, antioxidant capacity, and inflammation markers in aged mice.
Published in the journal Nutrients, the researchers suggest that probiotics and dietary fiber combination might be used as a novel and promising anti-aging synbiotic agent in humans.
Fighting Aging With Feces-Sourced Probiotics And Fiber
As aging is inevitable and its relative negative symptoms are complicated, we tend to pursue anti-aging treatments, ranging from diets to drugs. Accumulating evidence has indicated that prebiotics and probiotics could affect our health by regulating our gut microbiota.
Prebiotics — a form of dietary fiber that feeds the “friendly” bacteria in your gut — are resistant to digestion and absorption during passage through the stomach and small intestine but can be fermented in the large intestine by gut bacteria.
This dietary fiber can influence our gut bacteria to produce beneficial metabolites, such as short-chain fatty acids (SCFAs). But data indicate that daily dietary fiber intake for the elderly is roughly 40% below the recommended adequate intake.
For example, concentrations of SCFAs, which may promote weight loss and provide various health benefits, are less optimal in the elderly.
Live microorganisms called probiotics — like the ones advertised in cultured dairy products (e.g., yogurt and kefir) — administered in adequate amounts exert health benefits to the host.
Probiotic strains derived from the elderly exhibit excellent antioxidant, cholesterol-lowering, and immune-regulating activities.
Lactobacillus is the largest probiotic group with a high possibility of developing functional food. However, individual bacterial strains always exhibit unique bioactivities that require experimental confirmation.
Several Lactobacillus strains have been reported to have anti-aging effects due to their radical-scavenging activity and oxidation stress-attenuating ability.
Centenarian-Sourced Probiotic And Dietary Fiber Improve Aging In Mice
Previously, the Guangxi University research team isolated a strain of Lactobacillus — which they named LTL1361 — from the feces of healthy centenarians living in Bama, China, and demonstrated its potential probiotic properties in preliminary research in cultured cells.
In this study, Ren and colleagues examined the anti-aging effects of the LTL1361 strain and dietary fiber on learning and memory ability, antioxidant capacity, inflammation markers, and SCFAs in natural aging mice.
Using the Morris water maze test of spatial memory, the Guangxi University researchers evaluated the effects of dietary fiber and LTL1361 on cognitive abilities in aged mice. Researchers used the Morris water maze to study spatial memory and learning.
In this test, animals are repeatedly placed in an opaque pool of water surrounded by environmental cues, where they are tracked as they learn to find a hidden escape platform. The researchers observed a reduction in the time it took aged mice treated with dietary fiber or LTL136 to find the platform.
The results revealed that the combination of dietary fiber and centenarian-sourced LTL1361 significantly improved learning and memory ability.
In addition to improving learning and memory, Ren and colleagues showed that the combination of dietary fiber and Lactobacillus LTL1361 protected intestinal wall function and areas of the brain associated with cognition (pyramidal neurons) and learning/memory (hippocampus).
Furthermore, LTL1361 and dietary fiber decreased oxidative stress and inflammation and increased the availability of SCFAs in the small intestine.
Can Probiotics And Fiber Improve Human Aging?
The combination of dietary fiber and Lactobacillus LTL1361, which is a synbiotic (mixture of probiotics and prebiotics) seems like a promising treatment for cognitive decline in the elderly.
Nevertheless, further studies are required to clarify the biological connection between our gut, the microorganisms living within our gut, and how they affect our brain during aging.
Most of the latest clinical research has focused on applying commercial probiotics (one strain or a cocktail) to healthy older people.
The results indicate that probiotic consumption may positively impact by increasing the levels of specific beneficial gut microbe populations or modifying subpopulations of Lactobacillus.
These studies show that probiotics can enhance the immune response and improve bowel movements, among other beneficial effects.
Other studies have shown that the health benefits of probiotics are related to their ability to revert age-related increases of opportunistic pathogens, such as Clostridium difficile, which are involved in antibiotic-associated diarrhea that impact nutrition and inflammatory status.
In the elderly, C. difficile-associated diarrhea was linked to a reduction in the number of Bifidobacteria. For this reason, therapies based on the use of probiotics to correct the microbiota imbalance seem promising.
However, current guidelines do not recommend their administration. Notwithstanding the promising results, other studies reported controversial ones, most of them having no significant effects.
Updated: 2-19-2022
The Crypto World Should Know About Longevity
Biotech researchers working closely with decentralized tech innovators indicates that there is an appetite for progress in the longevity sector.
As the first month of 2022 drew to a close, the internet was abuzz with trend predictions for the year ahead. Bloggers, Twitter influencers and established business moguls alike were, and still are, incredibly excited about the new opportunities afforded by an explosion of innovation in Web3.
However, something you may have missed in scrolling pages of celebrity-backed NFT drops or speculations about the metaverse was the launch of a new company called Altos Labs, reportedly backed by Amazon’s former CEO Jeffrey Bezos.
The language that followed the launch was largely typical of how many mainstream journalists talk about the longevity sector, words like mysterious and secret. I’ve written in earlier articles that there are a lot of misconceptions around the longevity sector, and this new round of headlines seems to confirm this as still accurate.
I believe there is a lot more to the longevity sector than secret deals featuring the planet’s wealthiest billionaires. The space is ripe with interested investors, groundbreaking discoveries and development opportunities.
The message is clear — longevity is here to stay. And for the crypto industry, or a world populated with trend-setting, tech-savvy individuals, the time is now to start getting up to speed on this incredible space.
Longevity Deals
While it was wrapped up in mysterious media discourse, the Altos Labs deal was indicative of the high-level interest in the diverse longevity sector. With a reported $3 billion backing on day one, the deal is believed to be the largest biotech deal to date and has attracted the talent to match.
The first executive team is composed of globally renowned scientists, CEOs and doctors eager to seek new heights in the science of anti-ageing.
The Altos announcement was reminiscent of Calico Labs, another longevity company announced in 2013 by Google co-founder Larry Page. While the Google spin-off has been subject to inquiries about its productivity, it successfully published its first preprint on cell reprogramming in May 2021.
It is not only tech CEOs making headlines about investment: Deals from the past two months alone include areas like stem cell therapy, liver regeneration, senescence targeting, reproductive longevity and more.
It’s clear that the planet’s top innovators are looking to live longer. It’s up to us to make sure that longevity technology is accessible to all when it reaches public use.
Longevity Discoveries
Insilico Medicine, a Hong Kong-based drug discovery company, announced a significant milestone at the end of 2021. Their first AI-discovered medicine entered human trials. The medication will be used to treat idiopathic pulmonary fibrosis, a chronic lung disease, and has shown promising early results in first studies. This marks the first AI-discovered drug to reach this stage and is an exciting indication of new inventions to come.
AI is a central area of research in the longevity space, with countless applications in drug discovery and beyond.
Other new and exciting areas include cell regeneration, plant-powered supplements and progress in wearable technology that helps monitor ageing-related diseases. Charitable organizations like VitaDAO and the Longevity Science Foundation are working tirelessly to provide research funding to these early-stage ideas and help them reach public adoption.
Longevity Sector Development
On Deck, a company that offers acceleration programs for top talent across multiple sectors, launched its first Longevity Biotech Fellowship in June 2021. The inaugural cohort boasted participation from top longevity and biotech companies including turn.bio, Juvena Therapeutics, Retro Sciences and more.
Researchers from universities including Yale, University of Washington and ETH Zurich also contributed to the curated community.
On Deck is currently accepting applications for their second cohort, which will kick off later this month. The community claims to push the boundaries of traditional health and is growing the biotech ecosystem one member at a time.
Longevity is also reaching public awareness via international organizations. Martha Deevy, director of the Stanford Center on Longevity, contributed to a World Economic Forum article on the societal implications of living longer lives.
The idea of the multi-generational workplace, for example, has been expanded to include five or six different generations in one office space as a result of employees living longer and retiring later.
Deevy noted that while old age is often seen as a time of dependence and vulnerability, this narrative is changing. New societal approaches should emphasize a lifelong balance between education, work and rest. Deevy’s article said it best: We need to pay attention to the longevity sector because the effects of living longer lives are already showing up in surprising areas of our everyday lives.
Longevity And Crypto
It is clear that the longevity sector has been busy with no signs of slowing down. A recent poll showed that 70% of respondents believed the human lifespan should continue to increase if the necessary medical and technological advancements were in place to support it. Similarly, 68% of respondents reported they would be willing to take anti-ageing drugs if proven effective and free from side effects.
There is an appetite for progress in longevity, and the above examples show that we are on the right path. Still, more can be done — stay tuned for more news about longevity nonfungible tokens (NFTs), crypto investment into this space and other updates.
Updated: 5-7-2022
Researchers Say They’ve Reversed Ageing In Human Cells By 30 Years
Researchers have reversed ageing in human skin cells by 30 years, according to a new study.
Scientists say they have developed a method to time jump human skin cells by three decades – longer than previous reprogramming methods, rewinding the ageing clock without the cells losing their function.
Researchers describe being able to partly restore the function of older cells, as well as renew the biological age.
According to the study, in experiments that simulated a skin wound, the partially rejuvenated cells showed signs of behaving more like youthful cells.
Experts suggest that, while in their early stages, the findings could eventually revolutionise regenerative medicine, especially if it can be replicated in other cell types.
Professor Wolf Reik, a group leader in the Epigenetics research programme who has recently moved to lead the Altos Labs Cambridge Institute, said: “This work has very exciting implications.
“Eventually, we may be able to identify genes that rejuvenate without reprogramming, and specifically target those to reduce the effects of ageing.”
“This approach holds promise for valuable discoveries that could open up an amazing therapeutic horizon.”
Dr Diljeet Gill, a postdoctoral researcher in Prof Reik’s lab at the Babraham Institute who conducted the work as a PhD student, said: “Our results represent a big step forward in our understanding of cell reprogramming.
“We have proved that cells can be rejuvenated without losing their function and that rejuvenation looks to restore some function to old cells.
“The fact that we also saw a reverse of ageing indicators in genes associated with diseases is particularly promising for the future of this work.”
As people age, their cells’ ability to function declines and the genome – DNA blueprint – accumulates marks of ageing.
Regenerative biology aims to repair or replace cells, including old ones.
One of the most important tools in regenerative biology is our ability to create ‘induced’ stem cells.
However, this process essentially wipes the cells of their function and gives them the potential to become any cell type.
The new method, based on the Nobel Prize-winning technique scientists use to make stem cells, overcomes the problem of completely erasing cell identity by stopping reprogramming part of the way through the process.
This allowed researchers to find the precise balance between reprogramming cells, making them biologically younger, while still being able to regain their specialised cell function.
They say the potential applications of this technique are dependent on the cells not only appearing younger but functioning like young cells too.
Experts suggest that, in the future, the research may also open up other therapeutic possibilities.
They observed that their method also had an effect on other genes linked to age-related diseases and symptoms, like the APBA2 gene associated with Alzheimer’s disease, and the MAF gene with a role in the development of cataracts.
The research is published in the journal eLife.
Scientists have been able to find a way to reverse the ageing process in human skin. Through a breakthrough research, a team of Cambridge scientists claims to have made the skin cells of a 53-year-old woman 30 years younger.
This is longer reversal of the ageing clock than any other previous studies without damaging the cells. A study detailing the method has been published in eLife magazine.
The team told the BBC that it can do the same thing with other tissues of the body. Their ultimate aim is to develop treatments for age-related diseases, like diabetes, heart disease and neurological ailment.
“This work has very exciting implications. Eventually, we may be able to identify genes that rejuvenate without reprogramming, and specifically target those to reduce the effects of ageing,” Professor Wolf Reik, a group leader in the Epigenetics research programme, told Sky News.
The findings are still in early stages and if more research is done, the method could revolutionise regenerative medicines, said the scientists.
It is built on the technology used to clone Dolly the sheep more than 25 years ago.
The researchers at Roslin Institute in Scotland cloned Dolly by developing a method that turned the mammary gland cell taken from a sheep into an embryo. The gland was taken from six-year-old Finn Dorset sheep and an egg cell taken from a Scottish Blackface sheep.
Dolly was born on July 5, 1996.
The technique was aimed to create human embryonic stem cells, which could be grown into specific tissues, like muscle, cartilage and nerve cells. These tissues could be used to replace old body parts.
It is the gradual decline in the ability of the cells to function optimally, which leads to tissue dysfunction and disease. Regenerative biology aims to repair these old cells.
The team in Cambridge used the Maturation Phase Transient Reprogramming (MPTR) method, which overcomes the problem of erasing cell identity, allowing the researchers to find the balance of making cells younger while still retaining their specialized cell function.
Live Longer: ‘Amazing’ Technique ‘Reverses’ Ageing By 30 Years And Could Treat Alzheimer’s
A REVOLUTIONARY technique has allowed scientists to de-age individual skin cells by 30 cells while retaining their specialised functions – and it could pave the way to new treatments for age-related conditions like Alzheimer’s disease.
As one gets older, time takes its toll and we find ourselves perhaps less able to do all the things we used to. The same kind of thing happens, albeit on a smaller scale, to our cells.
Scientists working in the field of regenerative medicine work to reverse the ravages of time, and other forms of damage, by re-growing, repairing and replacing cells and tissues.
One of their key tools lies in the ability to create so-called induced stem cells, which theoretically have the ability to become any other cell type, by taking regular cells and removing some of the marks that make them specialised.
Unfortunately, we are presently not able at present to reliably recreate all the conditions that turn stem cells into all the particular cell types — meaning that we can effectively make cells young, but in the process, they can lose their identity and function.
In their new study, epigeneticist Dr Diljeet Gill of the Babraham Institute in Cambridge and his colleagues addressed this problem by halting the cellular reprogramming halfway through — effectively making the cells biologically younger, while not actually erasing their specialised cell function.
Their method builds on the 2012 Nobel Prize-winning technique developed by Professor Shinya Yamanaka of Japan’s Kyoto University for turning mature cells with a specific function into stem cells that can become any other cell type.
The cell reprogramming process he developed takes around 50 days to run its full course, and makes use of four key molecules which have been dubbed “the Yamanaka factors”.
What Dr Gill and his team have done, instead, is to expose skin cells to the Yamanaka factors for just 13 days — which strips the cells of all their age-related changes and causes them to temporarily lose their identity.
When these partly-reprogrammed cells were given time to grow under normal conditions, however, the team found that the specific functions that make them skin cells returned.
Under a microscope, the cells were observed to produce collagen, the protein that makes skin resilient and strong and helps heal wounds, while genomic analysis confirmed that the cells had regained all the markers that are characteristic of skin cells.
The researchers were also able to confirm that the cells — which came from three donors aged 38, 53 and 53 respectively — had lost the hallmarks of ageing.
Dr Gill said: “Our understanding of ageing on a molecular level has progressed over the last decade, giving rise to techniques that allow researchers to measure age-related biological changes in human cells.
“We were able to apply this to our experiment to determine the extent of reprogramming our new method achieved.”
In fact, after checking both the cells’ “epigenetic clock” — which works by assessing the presence of chemical tags throughout the genome that indicate age — and all the gene readouts produced by the cell, the “transcriptome”, the researchers found that their process had effectively de-aged the skin cells be around 30 years.
The key to this research is that the cells do not just look three decades younger, but are able to act like it as well.
According to the team, the rejuvenated skin cells were able to produce collagen faster than cells of the same actual age that had not undergone the procedure.
Experiments involving creating artificial cuts in a layer of cells in a petri dish also showed that the rejuvenated skin cells moved faster to fill the gap.
The researchers said that this suggests their findings could be developed to create cells that are more efficient at healing wounds.
The team also reported that their “maturation phase transient reprogramming” method also appeared to have a potentially useful effect on genes related to age-related diseases.
For example, both the APBA2 gene — which is associated with Alzheimer’s disease — and the MAF gene which has been linked to the development of cataracts were found to show changes towards more youthful levels of transcription after the reprogramming process.
Dr Gill said: “We have proved that cells can be rejuvenated without losing their function and that rejuvenation looks to restore some function to old cells.”
“The fact that we also saw a reverse of ageing indicators in genes associated with diseases is particularly promising for the future of this work.”
The team said that, while the technique is in its early stages, it has the potential to “revolutionize” regenerative medicine.
Paper co-author and epigeneticist Professor Wolf Reik, formerly also of the Babraham Institute, added: “This work has very exciting implications.
Updated: 5-11-2022
Senolytics Improve Resistance Training In Old Mice
A senolytic combination only improved muscles with resistance training, not without it.
A paper published in GeroScience has reported that older mice taking the well-known senolytic combination of dasatinib and quercetin (D+Q) are able to build muscle more like young mice.
In this paper, the researchers cite their prior research showing that the SASP, in conjunction with its known inflammatory effects, harms the ability of muscle progenitor cells to proliferate, thus depleting the ability of muscle to regenerate.
That paper showed that removing senescent cells through the senolytic combination of dasatinib and quercetin (D+Q) allowed for muscle regeneration in old mice, although it did not benefit old mice.
In this new research, the research sought to determine whether this also applied to muscle hypertrophy: that is, whether senolytics can help older organisms to build muscle through resistance training.
Resistance Training For Mice
Unfortunately, it isn’t possible to get mice to the gym. Therefore, the researchers used an established technique of removing “synergistic” muscle tissue (in this case, of the soleus and the gastrocnemius) in order to spur the development of the targeted muscle (in this case, the plantaris) [2]. Sham surgeries, in which no tissue was actually removed, were performed on a control group.
The plantaris muscles of both young (5 to 6 months) and old (23 to 24 months) mice increased slightly compared to their respective control groups, although young mice had significantly more muscle mass both before and afterwards, and while older mice stopped growing plantaris muscle tissue after a week, younger mice continued to grow it for two weeks.
As expected, senescent cells, which increase in muscle tissue after exercise, were found in substantially greater numbers in the older mice, especially after 14 days. While their numbers varied wildly from mouse to mouse, older mice had substantially and significantly more senescent cells than younger animals did, according to tests for the known senescent biomarkers p21 and SA-ß-gal.
The Effects Of Senolytics
The effects of D+Q on senescent cells were significant, in line with previous murine studies. Cells expressing SA-ß-gal were decreased to a third of their previous level, while cells expressing p21 were approximately halved.
The researchers’ main hypothesis, that D+Q would increase muscle mass upon resistance training, was shown to be correct: older mice given this senolytic combination and the surgery had greater plantaris muscle mass and superior fiber characteristics to the older mice given only the surgery.
However, this comes with an important caveat. Older mice that received D+Q but only received the sham surgery, which did not impart resistance effects on their plantaris muscles, actually had muscles that were weaker or equal to the mice that did not receive D+Q at all.
In other words, in the absence of resistance training, senolytics were not shown to be of any benefit and may have even have caused harm below the level of statistical significance.
Senescent Cells In Human Volunteers
Fortunately, it is possible to get people to the gym. In a cohort of human volunteers between 20 and 39 years old, nearly no senescent cells were found in muscle tissue; however, after resistance exercise, p21 and SA-ß-gal tests found the presence of these senescent cell biomarkers, although their numbers varied wildly as they did in mice. This data suggests that the results found in mice may apply to human beings, although this was not a human trial.
Conclusion
The finding that senolytics may only have value in building muscle when combined with resistance exercise is a very important one that will certainly guide future trial design. If the results found in mice are recapitulated in human beings, a senolytic and exercise combination may be prescribed in the near future in order to give older people back some of their mobility and fight back against frailty.
Literature
[1] Dungan, C. M., Murach, K. A., Zdunek, C. J., Tang, Z. J., VonLehmden, G. L., Brightwell, C. R., … & Peterson, C. A. (2022). Deletion of SA ß-Gal+ cells using senolytics improves muscle regeneration in old mice. Aging Cell, e13528.
[2] Kirby, T. J., McCarthy, J. J., Peterson, C. A., & Fry, C. S. (2016). Synergist ablation as a rodent model to study satellite cell dynamics in adult skeletal muscle. In Skeletal Muscle Regeneration in the Mouse (pp. 43-52). Springer, New York, NY.
Updated: 8-12-2022
Announcing The Longevity Prize
This series of prizes honors and accelerates rejuvenation research.
The Longevity Prize is a series of prizes designed to honor the researchers who are helping to build a future in which age-related diseases are a thing of the past. This new initiative aims to accelerate progress in the rejuvenation biotechnology field and encourage innovation.
While great strides in our understanding of aging have been made in the last decade, there is still a lot we do not know and much work to be done. There has been some amazing progress made, but solving aging is the most challenging endeavor that humankind has ever faced.
There are more and more rejuvenation companies entering the arena to take on this challenge, but the clock is ticking for us all.
The Longevity Prize aims to provide researchers with an incentive to develop novel approaches to bring aging under medical control, thus providing longer, healthier lives for all.
Longevity Prize Is Doing Something Different
Unlike traditional funding and prize models where researchers are given a fixed amount of money to focus on a specific goal, the Longevity Prize aims to do something different.
By creating a series of prizes, the goal is to create a rising tide of research proposals, experiments, and collaborations.
Because the Longevity Prize is free from the traditional systems of funding, this means that more ambitious projects have the potential to receive attention.
Risk aversion is a huge problem in normal funding, so this initiative helps to address that by supporting undervalued or overlooked projects.
The first round of prizes totals $180k and was fundraised through Gitcoin. Community donations were matched by VitaDAO, Vitalik Buterin, and Stefan George.
The first prize to be announced is the Hypothesis Prize.
While great strides in our understanding of aging have been made in the last decade, there is still a lot we do not know and much work to be done. There has been some amazing progress made, but solving aging is the most challenging endeavor that humankind has ever faced.
There are more and more rejuvenation companies entering the arena to take on this challenge, but the clock is ticking for us all.
The Longevity Prize aims to provide researchers with an incentive to develop novel approaches to bring aging under medical control, thus providing longer, healthier lives for all.
Over a century of all the world’s biological knowledge is available to anyone taking the time to read the literature. There are cases where key discoveries are made in the past, but forgotten for long periods of time – only to be rediscovered.
The hypothesis prize aims to resurface such discoveries and research areas, focusing our attention on the most promising directions.
Normally, researchers can only start experiments when they successfully get the funds to begin. The first round aims to combat this problem by giving out prizes based on hypothesis generation that will then help to shape the second and subsequent prize rounds that follow.
After more than a century of research, the literature is filled with hidden gems and forgotten knowledge. Most of this knowledge is available to anyone to read, and there are plenty of cases where discoveries are forgotten for decades only to be found again years later.
Longevity Prize Would Love To Hear From You
Do you have a suggestion for an underappreciated area of aging research that deserves some attention? Perhaps you have read the scientific literature and can explain why a particular area of research needs more focus?
You can submit a proposal (1-3 pages max) for consideration and could win a prize of up to 20k. Finalists will be invited to present their proposal to the judges. Excellent proposals will be moved to the next phase, where they will be eligible for follow-on funding.
Lifespan.io is proud to be part of this awesome initiative alongside VitaDAO, Foresight Institute, and the Methuselah Foundation.
Updated: 8-29-2022
Scientists Move Closer To Unlocking The Secrets Of The Immortal Jellyfish, And Possibly Human Aging
Authors of a new study hope the genome mapping of Turritopsis dohrnii, known for its ability to rebirth itself, might lead to discoveries relevant to improving human healthspan.
A team of scientists in Spain has succeeded in mapping the genome of a jellyfish known for its ability to cheat death by rebirthing itself.
Dubbed the immortal jellyfish, the tiny sea creature can turn its biological clock backward and revert to a clump of juvenile cells—even after reproducing sexually.
Once back in its juvenile state, the jellyfish can also reproduce asexually by cloning polyps of itself—giving it two ways to propagate its genetic material.
In a study published Monday in the Proceedings of the National Academy of Sciences, the authors said they hoped their genome mapping might lead to discoveries relevant to human aging and efforts to improve the human healthspan.
Marine biologist Maria Pascual Torner, one of the lead authors and a postdoctoral researcher at the University of Oviedo in Spain, where the research was done, said the focus was to better understand the mechanisms of rejuvenation.
“It’s a mistake to think we will have immortality like this jellyfish, because we are not jellyfish,” she said. But it’s possible something in the immortal jellyfish’s evolutionary trick can be used to better understand the pathologies of aging, she said.
Many types of cnidarian, or jellyfish, have some age reversal capacity, but most lose that ability with sexual maturity, the study said.
Three types can rejuvenate after adulthood and of those three, only one, the Turritopsis dohrnii, keeps its capacity at 100%, according to the study.
One of the team’s biggest challenges was finding some of the needle-sized jellyfish in the ocean, which they finally did off the Italian coast, said Dr. Pascual Torner.
The scientists compared their genome mapping of T. dohrnii to that of a closely related species that isn’t known to have post-reproductive rejuvenation.
The results revealed key molecular mechanisms behind the rejuvenation of the immortal jellyfish, the study said. The research suggests T. dohrnii is potentially better at repairing and replicating its DNA and maintaining its stem cells.
Dr. Jan Karlseder, a molecular biologist and director of the Glenn Center for Biology of Aging Research at the Salk Institute, said the study contained an important message about extending the healthspan, or healthy years, of an organism.
“The most interesting thing is that it’s not a single molecular pathway…It is a combination of many of them,” he said. “If we want to look for an extension of healthspan, we cannot just focus on one pathway. That will not be sufficient. We need to look at many of them and how they synergize.”
Biological immortality doesn’t shield the jellyfish from predators such as sea snails, crustaceans and sea worms.
“The fate of everything in the ocean generally is to be eaten at some point,” said Monty Graham, a professor of integrative biology and director of the Florida Institute of Oceanography.
T. dohrnii usually hits its reset button when it’s looking to avoid death caused by environmental factors, he said.
When adult jellyfish, known as medusae, feel threatened by changing conditions, they roll back their genetic clock to become larvae again.
Reduced to a thin layer of cells and tissue, they drift through the ocean looking for something to attach to, such as a rock or blade of sea grass.
In the study, the scientists evaluated whether rejuvenated cells could differentiate into any cell type of that jellyfish, such as nerves and muscles.
The ability of an immature cell type to be pluripotent, meaning able to develop into any cell type the jellyfish needs, is at the core of rejuvenation, Dr. Graham said.
In humans, stem cells are considered pluripotent, however, much work still has to be done before any connection could possibly be made between the immortal jellyfish’s rejuvenation process and relevance to human aging, he said.
Once a larva finds a place, it metamorphoses into a polyp that clones itself, creating a colony of polyps. When conditions are right, the colony blooms, and generates new jellyfish that are genetic replicas of their parent.
When those jellyfish become medusae, they only get one sexual reproductive event, said Dr. Pascual Torner, who co-wrote the study with Dr. Dido Carrero, who holds a Ph.D in molecular oncology and biomedicine.
What isn’t known is the maximum number of times, if there is a maximum, that a medusa can regenerate, Dr. Pascual Torner said.
“But whenever a medusa rejuvenates, it creates a polyp,” she said. Researchers have been unable to re-create the rebirthing process in a lab, but in the wild a polyp normally generates new jellyfish once a year, she said.
Although adult jellyfish only get one sexual reproductive event in a lifetime, it’s critical to the survival of the species, said Dr. Graham.
The nature of evolution means that time catches up with all things eventually, Dr. Graham said, even the immortal jellyfish.
“At some point…the organism is going to have to give up to a future generation in order for the species to be able to deal with long term variations in the environment,” he said. “That’s the way it has to work or everything goes extinct.”
Updated: 9-24-2022
How Crypto Is Playing A Role In Increasing Healthy Human Lifespans
How can you add years to your life, and life to your years? Longevity science can help — and this is a concept that’s of particular interest to crypto pioneers.
It’s a question that’s infatuated scientists for decades: how can we prolong life expectancy — giving humans everywhere more years of good health?
This field is known as longevity science, and within this industry, experts argue care which regards ageing as a normal but treatable ailment are rare — and of the approaches available, they can only be accessed by those who are highly educated and privileged.
Just some of the key tenets that govern this approach to medicine involve therapeutics, personalized medicine, predictive diagnostics and artificial intelligence.
The goal is to eliminate a “one size fits all” attitude toward treatment, and ensure that therapies are customized to an individual’s unique medical profile.
This can matter in many different ways — to the best method for tackling cancer, to the food we eat and our risk of heart disease.
And while predictive diagnostics offers an existing way of unlocking better patient outcomes, this often hinges upon using large amounts of anonymized data to determine what’s happened in the past, and how greater levels of success are achieved in the future.
Bizarrely, there are parallels between cryptocurrencies and longevity science. You could argue that this approach to medicine is currently where digital assets were back in 2013 — a time when crypto discussion was confined to online message boards, niche group chats and convoluted whitepapers.
Longevity researchers are excitedly sharing their findings with one another — and collaboration is taking place across sectors. Experts are keen to ensure that anyone with an interest in this nascent field can get involved and contribute.
Educating The Masses
As in the crypto industry, a big challenge that longevity science faces is education — and simply explaining this concept to the public. This is a journey that takes time, effort, money and patience.
Because of this, a dedicated event has been established so this cutting-edge concept can be discussed in an open forum. The Longevity Investors Conference is set to take place in Switzerland from Sept. 28-30. It’s being sponsored by Credit Suisse, and tickets can be paid for in cryptocurrency.
It’s being organized by Marc P. Bernegger. He’s a founding partner of Maximon — a Swiss company that invests and builds in longevity-focused companies. Bernegger explored Bitcoin in 2012 and told Cointelegraph:
“There is room for everyone. We can all travel the same path but take different approaches. It’s still the same narrative.”
Just some of the items on the agenda include exploring the scientific meaning of longevity — and how this will affect individuals around the world in the long run.
Discussions will also be held on how to cultivate investment in this fledgling space, and according to Bernegger, this is a field that’s of great interest to crypto enthusiasts.
The conference aims to build bridges, and highlight how scientists play a vital role in ensuring that we can all benefit from longer lifespans and a healthy retirement.
While there are business opportunities to be found, investors face a challenge because they’re not from a scientific background.
Likewise, bright minds often need an entrepreneurial perspective in order to bring their genius concepts to market.
Bernegger added: “There are a number of different perspectives — the entrepreneurs, the scientists, investors who bring money. They need a combination of everything. This sector appreciates new players. The more money there is, the more smart and serious people you have, the better. The industry is still finding itself.
It is accessible now, and people are happy to help.”
Why Crypto Is A Good Match
It’s the science element that’s attracting early adopters of cryptocurrency to this space. The reason is simple: because many of these enthusiasts are forward looking, open minded and technology driven.
Describing the initial days of crypto, Bernegger explained: “They were all in for the technology. It was not just speculative. They saw the potential of a peer-to-peer solution, and now they see the potential with regard to ageing.”
Indeed, blockchain technology also has the potential to enhance the quest to achieve longevity. Decentralized autonomous organizations (DAOs) have already been established that are funding research to support and commercialize therapeutics.
This approach also ensures that donors can vote on the future direction of research projects.
Even though the bear market has cast a long shadow over the crypto sector, many of those in this industry are firmly in the “BUIDL” phase. They’re using this opportunity to innovate, cultivate new products, and develop the trends that will drive the next bull run.
Longevity science can be one of them — and according to Bernegger, pioneers know that paying close attention to health is far more important than the value of any token.
We already know that the rate of ageing can be controlled, to some extent, by genetic pathways and biochemical processes.
But in the coming decades, there are still so many questions to be answered — and dots to be connected — in the quest to improve our quality of life, and ensure that anyone can access it.
The Longevity Investors Conference says attendance will be strictly limited to 100 hand-picked delegates, and they’ll be able to benefit from the insights of over 30 outstanding speakers.
It’s a compelling opportunity to get to know the industry inside and out, all while establishing meaningful contacts with the best people in the field.
It’s going to take place in Gstaad, one of the most exclusive Swiss mountain resorts, in a “one-of-a-kind setting” within a plush, five-star hotel, and world-class speakers flying in to attend and present.
This includes members of the Longevity Science Foundation Visonary Board. This nonprofit recently entered into a partnership with The Giving Block — tapping into a vital stream of crypto philanthropy.
If you want to know how to add years to your life, and life to your years, this could be the most important conference you ever attend.
Updated: 11-11-2022
What Can Blockchain Do For Increasing Human Longevity?
The emerging longevity sector is attracting many crypto natives, who can both benefit from it and contribute to it with blockchain tools.
The nascent longevity industry focuses on the research and implementation of solutions and technologies to extend the lifespan of human beings — making people live healthier, longer lives.
Longevity is not yet considered an official medical term, and aging is not officially considered a disease but a natural occurrence in every living thing.
However, some biologists, researchers and practicing doctors believe this approach should change, and they are striving to discover the mechanisms of aging in humans.
In doing so, they are creating age clocks by defining biomarkers for measuring biological age, exploring the best lifestyle habits and natural supplements, and inventing new drugs that could stop us from getting older.
Longevity has been on the radar of crypto leaders for some time already, which is not a surprise given that the industry promises to improve humankind through innovation.
Indeed, one prominent event in the longevity industry, the Longevity Investors Conference, is organized by Marc P. Bernegger and Tobias Reichmuth, who were previously involved with the Crypto Finance Group.
Crypto Meets Longevity
At the most recent iteration of the Longevity Investors Conference in September 2022, speakers taking part in the “Crypto meets longevity” panel noted that both industries — crypto and longevity — are disruptive fields that challenge established norms.
“Everybody here in this room is, to a certain extent, a pioneer because we really come together in a phase where it’s similar to the internet, Bitcoin and other exciting industries,” said Bernegger, who is the founding partner at Maximon — an accelerator for longevity companies.
Aubrey de Grey, an English author and biomedical gerontologist, highlighted that the mindset of crypto and longevity innovators is very much alike, with both being “completely comfortable working in an area that is still very unorthodox and needs to be taken forward.”
Ryan Pyle, founding partner at Maine Investments — a Zug-based company managing digital assets — said that the longevity industry reminds him of what crypto was like in 2013 and that while no one knows precisely where the industry will be in five to 10 years, the potential is very promising:
“So, I see the ecosystem being very exciting, and I think what Maximon is trying to do at this longevity conference is really early stage — like probably two or three years, maybe, too early, which is great. That’s where you want to be, right? You’d rather be too early than too late.
A lot of crypto people have invested in this space just because they’re totally accepting of this level of risk.”
Bernegger also shared that because the longevity industry is at such an early stage, it’s currently a highly attractive period for investors to enter and that “similar to Bitcoin back then, it is less about the price but also about the underlying technology and potential.”
How Blockchain Can Apply To Longevity
On the application of blockchain technology itself, Bernegger is less optimistic, stating, “I think it’s difficult, then, to combine [blockchain] technology with another exciting field like longevity. […]
From my side, personally, I think the biggest synergy is less in combining two technologies and really more the money.” On the potential in the investing and funding side of crypto, Bernegger added:
“I personally rather would focus on the funding side and less on the technology side, without neglecting that there are few exciting projects in the DeSci [decentralized science] space which definitely have huge potential. But I think it will take years to really see tangible products solving a real-world problem there.”
Bernegger also mentioned the compliance aspects of both industries, stating that regulators might be a burden to research and adoption not only due to their role to protect end customers but “to another extent also to protect the status quo.”
The longevity industry is still in a very nascent stage and has not seen many blockchain-related use cases yet. Nevertheless, knowing what kind of problems have already been solved with the help of blockchain technology allows some professionals to see potential applications.
Claire Cui, a self-described longevity enthusiast and blockchain adviser, mentioned decentralized data as one potential use case:
“What crypto has figured out is basically [the potential of] blockchain technology to cover some of the issues that people have today, like data privacy, data ownership. So, that’s where people are very curious about. And in health, it’s even more sensitive. Nobody wants to have his DNA in health records leaked somewhere because somebody hacks it.”
Data ownership also allows users to earn from sharing it with companies that use it for scientific research. Thus, regular users become part of the revenue chain.
Another application of blockchain in the longevity industry is decentralized autonomous organizations, or DAOs, and decentralized intellectual property.
One existing example is a platform called VitaDAO, a DAO for community-governed and decentralized drug development that collectively funds and digitizes research in the form of nonfungible tokens representing IP. Christian Angermayer, founder of Apeiron Investment Group, said:
“I think one interesting part of blockchain technology could be that we sort of make sure that people can collaborate better but then also that literally everybody collaborates and gets a fair share in whatever comes out of it, both reputation-wise and, especially, also financially.”
The longevity industry seems to be an exciting area in which the crypto community can explore investment and use cases, particularly because, by its nature, it is reminiscent of blockchain’s early days.
At the same time, many existing areas of blockchain adoption can be applied to the longevity industry to solve some of the problems it’s facing.
Updated: 1-8-2023
Want To Avoid Death? Maybe Cryonics Isn’t Crazy
Despite the mockery from scientists, it’s hard to ignore the sliver of possibility that cryogenic freezing could extend life.
Emil Kendziorra has his death all planned out.
In the event that his life is cut short, a team of medical professionals will supply the man’s recently-deceased body with oxygen until it can be transported to a suspension facility.
There, the blood and water in his body will be replaced by a chemical mixture, a kind of human antifreeze, to prevent ice crystals from forming when his body is placed in arctic cold liquid nitrogen. Known as vitrification, this will put his cells into a state of suspended animation.
The goal is that many decades from now, the medical community will be able cure whatever ailment ended Kendziorra’s life and resurrect him from his suspension. It’s possible this won’t work. But, for now, he is sticking with the plan, and the process is not that expensive, nor outlandish.
The field of cryogenics has been gaining ground over the last few decades, albeit very slowly and amid endless ridicule by scientists.
To date, about 500 people have been put in cryogenic stasis after legal death, with the majority of them in the US. But a few thousand more, including Kendziorra, are on waiting lists, wearing bracelets or necklaces with instructions for emergency responders.
Kendziorra, 36, runs Berlin-based Tomorrow Biostasis GmbH, one of the first cryonics businesses in Europe to join a market dominated by American firms organizations like The Alcor Life Extension Foundation and The Cryonics Institute. The former cancer doctor has several hundred people on his firm’s waiting list.
They skew to their late 30s, male and tend to work in technology. Patients can choose to have their entire body preserved and held upside down 1 in a four-person dewar, a thermos-like aluminum vat filled with liquid nitrogen, or just preserve their brain, which is cheaper.
Kendziorra says cryopreservation overall has become less expensive over the past few decades on an inflation-adjusted basis, a claim that he bases on historic prices published by his peers, who he says are making a collective effort to bring down costs.
That could be critical to shifting cryonics from a fringe pursuit to something a little more mainstream, especially since it is no longer just for billionaires like PayPal Inc. co-founder Peter Thiel (who has reportedly signed up with Alcor).
Kendziorra, for instance, has made cryonics just another monthly subscription by capitalizing on insurance, he told me during a Twitter Spaces discussion on cryonics last month.
His customers pay a 25-euro ($26.54) monthly fee to Tomorrow Biostasis, and they also make the company the beneficiary of a minimum 100,000-euro life insurance payout upon their legal death. Kendziorra says that covers the full cost of cryonics including the biggest outlay: maintenance over the next century or so.
All told, most of his customers are paying about 50 euros a month for both the company’s subscription fee and the life insurance policy for the option of a long sleep at death.
Of course, most companies don’t survive for more than a century, so Tomorrow Biostasis also partners with a non-profit group in Switzerland to carry out the storage of customers on its behalf.
Arizona-based Alcor, one of the biggest cryonics organizations with 1,400 people on its waitlist, is also structured as a non-profit foundation.
The domain itself is largely funded by wealthy individuals including CEOs of tech companies, angel investors and scientists, Kendziorra says, adding that for them to invest in his own firm, their primary motivation shouldn’t be “monetary” but rather to help further the field.
The mechanics all sound sensible, but that still leaves the question of whether cryonics will work, medically speaking. Doctors and scientists have used words like quackery, pseudoscience and outright fraud to describe the field.
Clive Cohen, a neuroscientist from Kings College London, has called it a “hopeless aspiration that reveals an appalling ignorance of biology.” The Association of Cryobiology has compared it to turning a hamburger back into a cow.
But it’s foolish to dismiss an option that looks at least potentially viable given the advances technology has made in the last few decades and the milestones science will likely continue to achieve amid the declining cost of cryonics.
The field overall deserves to be taken more seriously. For his part, Kendziorra would rather take the slim chance that he might one day be revived to extend his life: “I very much hope that I never need to but if I get diagnosed with an incurable disease, where the alternative is burial or cremation, then I’d much rather be cryopreserved.”
There are, of course, many complex issues still to solve, including whether to vitrify people’s bodies with chemical fixation, which causes less structural damage but makes biological revival more difficult, and whether to risk fracturing the body’s cells to the point where they can’t function again after thawing.
Kendziorra says that storing a body at -196° Celsius means that there is almost no degradation to a person’s body even over many decades, but little is known about how well the delicate synaptic circuitry of the brain is preserved, or whether it could ever be fixed if damaged.
And aside from curing whatever ailment killed one of his customers, scientists would also have to be able to reverse death itself.
Yet that seemingly fundamental problem also points to the most intriguing argument of cryonics — around defining death.
Consider that the lifesaving action we now called CPR, or cardiopulmonary resuscitation, only came to mainstream attention in the 1960s. Before then, it was reasonable to declare someone dead at the point of a cardiac arrest. Today it would be considered medical malpractice to not try and resuscitate someone whose heart has stopped beating.
(The current record for restarting a heart is after nearly nine hours, when an Italian mountain climber suffered a hypothermic cardiac arrest during a freezing thunderstorm on the Dolomites, according to Guinness World Records.)
The shift in how we legally define death, and the possibility that science will advance to the point where it can resurrect damaged tissue and brain synapses — just as well as we can currently thaw embryos after decades on ice — makes it hard to disregard what may be possible in the future.
Cryonics may serve to mostly put a pause on life, the aluminium dewars acting like an ambulance to a better-quality hospital in the future. It is not a futile quest for immortality as many critics have argued, but rather an effort to extend life a little more. That is not so outrageous.
Success is admittedly a tenuous bet. The field’s last big breakthrough was made more than a decade ago, in figuring out how to cool a body to freezing temperatures without forming ice crystals. The next milestone will be reviving a mammal from cryogenic temperatures, according to Kendziorra, and no one is close to achieving that.
But when the cost of possibly extending life could come down to that of a monthly Netflix subscription, many more people may decide it’s worth a try.
Updated: 1-23-2023
How Sea Spiders And Other Creatures Could Unlock The Secrets Of Tissue Regeneration
“Scientists hope these animals will suggest ways to better repair limbs and tissue in people”.
Many animals can grow back lost claws, limbs or tentacles after shedding them when threatened or attacked by predators. Scientists are studying regrowth in these species, aiming to use those lessons to heal the human body.
Relatives of scorpions and spiders, ocean-dwelling sea spiders show a remarkable ability to regrow body parts. The animals regrow limbs when they molt, like the sea spider Endeis spinosa, which has been seen growing back three small legs.
Scientists reported Monday that one sea spider species, Pycnogonum litorale, can regrow body segments, an anus and reproductive organs. This species, glowing blue under an ultraviolet light, can regenerate after nearly half its body is sliced away.
Regrowth is well-studied in the axolotl, a species of Mexican salamander, which can start growing back an amputated limb within hours of it being cut away.
The axolotl can regrow multiple organs, including lungs, heart and brain. When regenerative cells from a donor salamander were grafted into the arm of another, they grew into an extra limb. Scientists want to better understand the conditions under which limbs grow back.
Some jellyfish are able to regress, transforming into a clump of juvenile cells when the environment turns hostile. Scientists last year identified some of the genes that allow the species Turritopsis dohrnii to return to its larval state.
Flatworm species have the rare ability to regrow their heads. Slices of the flatworm Dugesia japonica grew two heads–one at each end–when placed in a solution of the drug Praziquantel. The drug is used to treat parasitic infections.
Stem cells called neoblasts let the flatworm grow back lost organs, including its reproductive system. Scientists are studying the flatworm’s yolk producing organs (glowing pink) and yolk cells (glowing green) to understand how the creatures bring back these complex organs.
Zebrafish can regrow injured fins, tails and even heart muscle, with key genes guiding the architecture of the new part. Here, in a week-old fin grown after an amputation, genes directing growth at the front of the fin glow red and those directing the back glow green.
Updated: 1-25-2023
How To Be 18 Years Old Again For Only $2 Million A Year
Middle-aged tech centimillionaire Bryan Johnson and his team of 30 doctors say they have a plan to reboot his body.
Novak Djokovic, age 35, sometimes hangs out in a pressurized egg to enrich his blood with oxygen and gives pep talks to glasses of water, hoping to purify them with positive thinking before he drinks them.
Tom Brady, 45, evangelizes supposedly age-defying supplements, hydration powders and pliability spheres. LeBron James, 38, is said to spend $1.5 million a year on his body to keep Father Time at bay.
While most of their contemporaries have retired, all three of these elite athletes remain marvels of fitness. But in the field of modern health science, they’re amateurs compared to Bryan Johnson.
Johnson, 45, is an ultrawealthy software entrepreneur who has more than 30 doctors and health experts monitoring his every bodily function. The team, led by 29-year-old regenerative medicine physician Oliver Zolman, has committed to help reverse the aging process in every one of Johnson’s organs.
Zolman and Johnson obsessively read the scientific literature on aging and longevity and use Johnson as a guinea pig for the most promising treatments, tracking the results every way they know how.
Getting the program up and running required an investment of several million dollars, including the costs of a medical suite at Johnson’s home in Venice, California.
This year, he’s on track to spend at least $2 million on his body. He wants to have the brain, heart, lungs, liver, kidneys, tendons, teeth, skin, hair, bladder, penis and rectum of an 18-year-old.
“The body delivers a certain configuration at age 18,” he says. “This really is an impassioned approach to achieve age 18 everywhere.” Johnson is well aware that this can sound like derangement and that his methods might strike some as biotech-infused snake oil, but he doesn’t much care. “This is expected and fine,” he says of the criticism he’s received.
Johnson, Zolman and the team are more than a year into their experiments, which they collectively call Project Blueprint. This includes strict guidelines for Johnson’s diet (1,977 vegan calories a day), exercise (an hour a day, high-intensity three times a week) and sleep (at the same time every night, after two hours wearing glasses that block blue light).
In the interest of fine-tuning this program, Johnson constantly monitors his vital signs. Each month, he also endures dozens of medical procedures, some quite extreme and painful, then measures their results with additional blood tests, MRIs, ultrasounds and colonoscopies.
“I treat athletes and Hollywood celebrities, and no one is pushing the envelope as much as Bryan,” says Jeff Toll, an internist on the team. All the work, the doctors say, has started to pay off: Johnson’s body is, as they measure it, getting medically younger.
There are some obvious signs that Johnson is at least healthier than most 45-year-olds. The dude is way beyond ripped. His body fat hovers between 5% and 6%, which leaves his muscles and veins on full display.
But it’s what has happened inside his body that most excites his doctors. They say his tests show that he’s reduced his overall biological age by at least five years.
Their results suggest he has the heart of a 37-year-old, the skin of a 28-year-old and the lung capacity and fitness of an 18-year-old. “All of the markers we are tracking have been improving remarkably,” says Toll.
Zolman, who got his medical degree from King’s College London, is more measured. He stresses that his work with Johnson is just beginning and that they have hundreds of procedures left to explore, including a range of experimental gene therapies.
“We have not achieved any remarkable results,” he says. “In Bryan, we have achieved small, reasonable results, and it’s to be expected.”
“What I do may sound extreme, but I’m trying to prove that self-harm and decay are not inevitable”
In Johnson’s mind, however, success is already at hand. While he isn’t the first software developer to grow fixated on living a healthier life, he’s chasing something close to the ne plus ultra version of what tech-industry types call the quantified-self movement.
Over the past decade or so, Silicon Valley’s idea of optimizing your innards has mostly taken the form of the occasional exercise or diet fad, from intermittent fasting to Soylent. Johnson’s pitch is that you have to count a lot more than your steps to get a clear picture of what’s best for your body.
“What I do may sound extreme, but I’m trying to prove that self-harm and decay are not inevitable,” he says. In conveying the lessons of his unorthodox lifestyle to the rest of us, he’s also counting on a strategy that’s well known in the software business: making it feel, as much as it can, like a game.
In his 30s, Johnson created a payment processing company called Braintree Payment Solutions LLC. It was a massive success, but the long hours and stress left him overweight and deeply depressed, bordering on suicidal.
He sold the business to EBay Inc. in 2013 for $800 million in cash, then began a long journey to sort himself out.
This included learning more about his biology, an obsession that made its way into his work. He founded a biotech-focused venture firm called OS Fund and then, in 2016, a company called Kernel, which makes helmets that analyze brain activity to learn more about the mind’s inner workings.
Researchers are currently using the helmets to try to quantify the impacts of meditation and hallucinogens and find ways to lessen chronic pain.
By this time, Johnson had begun tinkering with his body: altering his diet, taking loads of supplements, snorting the occasional vial of stem cells.
I wrote about Kernel (and these early health experiments) for Bloomberg Businessweek in 2021, and I’ve followed Johnson’s physical and mental transformations for a few years now.
Throughout, he’s insisted that people don’t have the most important information they need to live healthy lives—that seeing the data in black and white can help people break destructive habits.
“You can look at your body and your situation and turn it over to willpower,” he says. “And, like, good luck.” Forcing himself to abide by Blueprint has taken late-night binges (of pizza, booze, whatever) off the table, and all the testing and tweaking has given him confidence that he’s doing as right by his body as he can.
Zolman, a generation younger than Johnson, has experienced his own kind of medical wakeup call. In 2012, his first year of med school in London, Zolman hurt his back playing basketball.
The injury proved bad enough that for about a year he struggled to walk properly and sometimes had to use a wheelchair after visits to the hospital.
The doctors he met with couldn’t seem to fix things, so he started doing his own research and developing his own physical therapy program, including deep-tissue massages across his legs, glutes, lower back, abs and pelvis. “As soon as I did that,” he says, “boom, I could walk.”
This could sound like the start of any TikToking quack’s sales pitch for enlightenment juice or liver bedazzlers, but Zolman, who isn’t shy about listing his academic achievements, finished his medical degree with honors in 2019.
Soon after, he began studying any and all clinical research he thought might help him live healthier for longer.
Zolman is convinced that progress in the field of longevity requires a more concerted pursuit of medicines and therapies that seem in any way promising. Out on the edges of medical science, he argues, there must be better outcomes than the ones we grudgingly accept.
In 2021 he opened a firm, 20one Consulting Ltd., in Cambridge, England. “My goal is to prove through biostatistics a reduction of aging of 25% across all 78 organs by 2030,” he says. “It’s an extremely hard and crazy idea.”
For beginners, his treatment, offered on a sliding scale, focuses on the basics of improving diet and exercise. More expensive programs, which top out at $1,000 an hour for people in Johnson’s bracket, include lots of testing, therapies and health-aid devices. (He doesn’t charge if the patient doesn’t see results.) Johnson, though, is the only client going this hard.
In Cambridge, Zolman spends much of his time reading research papers and synthesizing their findings into something Johnson can try. “There is no person in the world who is 45 chronologically but 35 in every organ,” Zolman says.
“If we can eventually prove clinically and statistically that Bryan has made that change, then it will be such a large effect size that it will have to be causative of the intervention and beyond what’s genetically possible.”
To determine that kind of progress, Zolman says, he keeps track of 10 or more different measurements for each of a patient’s organs.
With the brain, for example, he uses a range of MRIs and ultrasounds to track blood flow, tissue volume, scarring, swelling, and plaque growth in the cerebrum, ventricles, midbrain, cerebellum, pituitary and brainstem, and supplements those measurements with cognitive ability tests and blood draws.
Configuring the tests, let alone performing them, can be an arduous exercise, because much of the required hardware is usually found only at research institutions.
While he’s effusive and optimistic about his program, Zolman also tries to strike the tone of a realist. He concedes that it will take years to know if he’s chasing the right things and just how well any of this works.
“I think what he’s doing is impressive, and he has personally challenged me to be better. What he does is also essentially a full-time job”
Each morning starting at 5 a.m., Johnson takes two dozen supplements and medicines. There’s lycopene for artery and skin health; metformin to prevent bowel polyps; turmeric, black pepper and ginger root for liver enzymes and to reduce inflammation; zinc to supplement his vegan diet; and a microdose of lithium for, he says, brain health.
Then there’s an hourlong workout, consisting of 25 different exercises, and a green juice packed with creatine, cocoa flavanols, collagen peptides and other goodies.
Throughout the day, he eats some solid-ish health food (we’ll get there), with the recipes tweaked based on the results of his latest tests.
After eating, Johnson brushes, Waterpiks and flosses before rinsing with tea-tree oil and applying an antioxidant gel. His doctors say he has the gum inflammation of a 17-year-old.
There’s a regimen and series of measurements for every last part of Johnson’s body. He’s taken 33,537 images of his bowels, discovered that his eyelashes are shorter than average and probed the thickness of his carotid artery.
He blasts his pelvic floor with electromagnetic pulses to improve muscle tone in hard-to-reach places and has a device that counts the number of his nighttime erections. Of late, he’s been presenting as a teenager in that regard, as well.
Daily, he measures his weight, body mass index and body fat, and he monitors his waking body temperature, blood glucose, heart-rate variations and oxygen levels while sleeping.
He’s also undergoing a fairly constant stream of blood, stool and urine tests as well as whole-body MRIs and ultrasounds, plus regular tests aimed more specifically at his kidneys, prostate, thyroid and nervous system.
To repair sun damage to his skin, Johnson applies seven daily creams and gets weekly acid peels and laser therapy, and he’s begun staying out of the sun.
To improve hearing in his left ear (which suffered from childhood hunting trips in Utah), he does sound therapy, which tests the limits of the frequencies he can hear and then produces inaudible sounds that stimulate the cells in his ear and brain.
(Clinical studies performed at Stanford University and elsewhere have concluded that this can help the average person improve their hearing by at least 10 decibels, a significant margin.)
He has, however, rejected many of the internet’s favorite health fads, including resveratrol, ice baths and high doses of testosterone.
Doctors on his team help with the scans and tests, read the results and offer advice on what’s safe and what might be dangerous. At one point, Johnson’s body fat had fallen to 3%, which threatened the healthy functioning of his heart.
His team recommended tweaks to his diet, including eating more throughout the day instead of consuming all of his calories at breakfast.
Johnson’s lifestyle isn’t for me. In September, shortly before I walked up to his door in Venice for dinner, he texted to warn me that he’d just had some fat injected into his face and seemed to be suffering from an allergic reaction to the excruciating procedure. As a result, he said, he might look a little weird.
He was not wrong.
When he opened the door, I could barely recognize him. His face was so puffed up it looked like he’d spent the afternoon chugging bee venom. Stranger still, his pale skin was glowing, absent of most of the flaws that accompany middle age. He could have been mistaken for a big, swollen porcelain doll.
The procedure, he said, wasn’t the usual Hollywood look-younger filler. It was the first in a series of injections to build a “fat scaffolding” in his face that would produce genuine, young-person fat cells. “Filler is just patching over something,” Johnson said.
“It will take a few months for the fat scaffold to build, but then, as I regenerate, it will actually create fat on its own. If I do an MRI or multispectral imaging, then hopefully it will show that I’m identical to an 18-year-old again.”
Usually, the early adopters of this procedure harvest the fat from other parts of a patient’s body, but Johnson, who doesn’t have the fat to spare, received his from an undisclosed donor.
This treatment struck me as largely cosmetic. (Johnson also dyes his hair.) And, Zolman notes, there’s little to no evidence that having a fatter, more youthful face or luscious red hair offer clinical benefits on their own.
“But, if you do this at the whole-body level, it becomes clinically relevant,” Zolman says.
“If you restore young fat-level distribution throughout the whole body, you’re going to have less toxic compounds being secreted and affecting the rest of the body and you’re going to have things like better heat control. If you had no fat, you’d be f—ing dead. If you had no skin, you’d be f—ing dead. These are not aesthetic organs.”
As we talked about Blueprint, Johnson prepared dinner for me, a sample of his typical fare. On the yum side, I was given something called nutty pudding, which consisted of almond milk, macadamia nuts, walnuts, flaxseed, half a Brazil nut, sunflower lecithin, cinnamon, cherries, blueberries, raspberries and pomegranate juice. Delicious.
On the yuck side, I also had to eat a mound of vegetables that had been pureed into a gray-brown goop. Once upon a time, it consisted of black lentils, broccoli, cauliflower, mushrooms, garlic, ginger root, lime, cumin, apple cider vinegar, hemp seeds and olive oil, all of which sound fine on their own. But when put together and blended, it felt and tasted like dirt paste.
For dessert, we had chocolate, but not just any chocolate. “With Blueprint, there are layers,” Johnson said. “You can say, ‘Chocolate is good for you.’ Then the next layer is ‘Dark chocolate is better for you.’
And then there is this Dutching process that people sometimes do to chocolate where they alkalize it to take away the bitterness, but it ruins much of the value. So, you want non-Dutched dark chocolate, and you want some that has been tested for heavy metals.
And then you want chocolate from the regions of the world that have the highest concentration of polyphenols, which is what you’re trying to get. Unless you’re looking at that fifth layer of polyphenol concentration, you’re really getting very little benefit.”
The lifestyle seems exhausting, but Johnson revels in it, as does his 17-year-old son, Talmage. As our dinner kicked off, Talmage came into the house after a workout. For a moment, he was taken aback at his dad’s swollen face.
Then he let out a chuckle, as if he’d seen this type of thing before. Talmage prepared his own specialized dinner alongside us and said he’s adopted some, but not all, of his dad’s practices. For one, he doesn’t do the vegetable sludge, preferring his veggies raw or sautéed.
After dinner, father and son watched as I inserted my arm into a cardiac health monitor on Johnson’s kitchen island. The machine buzzed and whirred for a few seconds, then reported back that my ticker—at least by one measure—wasn’t that far off Johnson’s. It felt like a small victory for paunchy 45-year-old Scotch lovers everywhere.
Johnson has heard his share of criticism from people who’ve accused him of having an eating or psychological disorder or of being a delusional health zealot going about life in the most boring, restrictive way possible.
The handful of doctors I’ve interviewed on Johnson’s team all say he’s breaking ground in the field of longevity and probably extending his life, but even they have questions about whether their conclusions will apply to the rest of us.
“I think what he’s doing is impressive, and he has personally challenged me to be better,” says Kristin Dittmar, an interventional oncologist. “What he does is also essentially a full-time job.” She also stresses that cancer, her specialty, has genetic components that no cutting-edge science, let alone juices or creams, can yet beat.
One way to pass his gains along to others, Johnson says, might be radical transparency. He has a website where he posts his entire course of treatment and all his test results.
And, now, he’s launching another site, Rejuvenation Olympics, that encourages fellow travelers to do the same. The idea is to move away from the latest fads in favor of more rigorous medical science and a dash of competition.
The more popular this type of lifestyle becomes, the cheaper and more readily available some of the procedures Johnson tries might be.
“If you say that you want to live forever or defeat aging, that’s bad—it’s a rich person thing,” Johnson says. “If it’s more akin to a professional sport, it’s entertainment. It has the virtues of establishing standards and protocols. It benefits everyone in a systemic way.”
For a hint at the size of the early adopter demographic, there’s the blood test offered by a company called TruDiagnostic LLC, which tries to assess a person’s rate of aging by looking at whether various genes are functioning at hundreds of thousands of points across their genetic code.
Of the roughly 20,000 people who’ve taken the test in the three years it’s been on the market, about 1,750 have repeated it over years to track their battles against aging. The company says that within that group, Johnson has reduced his biological age the most.
It’s easy to imagine how a coterie of Johnson wannabes experimenting with ever-riskier procedures could go horribly wrong. More likely, most people will find Johnson’s lifestyle impossible or absurd.
Some researchers and health aficionados who have run across Johnson’s program take particular exception to his promotion of supplements and vitamins that they view as largely useless.
Still, some of the most respected experts who study longevity and aging say the underlying idea of an open forum for the science of life extension is inevitable.
“The whole longevity field is transitioning into a much more rigorous, clinical place,” says George Church, the famed Harvard University geneticist, who has stakes in a number of biotech companies.
“I think what Bryan is doing is very well-intentioned and probably very important.” He adds: “I also don’t think a lot of this stuff will be all that expensive when the dust settles.”
While Johnson won’t discuss it yet, he’s about to undergo some far more experimental procedures, including gene therapies, according to several of his doctors and advisers.
For better or worse, he’s very much dedicating his body to science in the hopes of proving what’s possible for the rest of us. “That’s the beauty of this,” he says. “It’s a new frontier.”
Keith Comito On The Benefits Of Blockchain Tech And Decentralization In Longevity Research
The co-founder of Lifespan.io is harnessing the power of decentralization to support cutting-edge scientific and technological discoveries.
Lifespan.io President and co-founder Keith Comito was into longevity before longevity was cool.
“We started our nonprofit back in 2014,” Comito said. “Back then, few credible researchers would be caught dead saying they were working on aging. It was still kind of a scientific backwater. So, we wanted to emulate what the cancer research advocates did, starting in the ‘40s.”
He said those early advocates identified the most credible research at the time and used “old-school” fundraising approaches like telethons to crowdfund for their work. They built a grassroots movement and grew their scientific expertise.
Through their efforts, defeating cancer is now an international priority with communities of millions of people crusading in the fight against this devastating disease that is closely linked to aging.
Comito said Lifespan.io is taking a similar approach but for all aging-related diseases. They’ve built a large community of people by sharing verified news about breaking developments in longevity, crowdfunding research using blockchain technology, and creating some of “the most successful YouTube collaborations in the space.”
“In 2017, I helped write the scripts for a popular series of aging videos that were the highest-viewed videos the week they came out, with 14 million views within days. That was huge to hit those numbers six years ago, and the like ratio was overwhelmingly positive.”
This illustrated to the field, Keith said, that it was okay for longevity researchers to come out into the open and not be ashamed of their revolutionary and life-saving work.
Comito leverages various skills honed from a diverse background, including computer programming, biology, and mathematics.
He has invented several metaverse-adjacent technologies, developed software that has been adopted in widely-used services like HBO Now, MLB at Bat and Disney+, and even accidentally created a top-charting Chuck Norris Joke Generator as a joke back in the early days of mobile applications.
Today, he’s looking at ways to bring our world into the metaverse that not only excite us but also heal our minds and bodies as well. Keith sees the future of Web3 overlapping and supporting the future of longevity while tying in other hot sectors like gaming and VR.
Shaking Up Science
Comito said that one area he’s spent the last few years exploring is the pure fundraising capabilities of cryptocurrency, working with organizations such as Gitcoin and Angel Protocol to brainstorm new models of crypto philanthropy to fund scientific breakthroughs. Of course, a number of “crypto heavyweights are already supporting life extension research,” he says.
Vitalik Buterin, for example, has supported several longevity projects, donating crypto to causes like the SENS Research Foundation.
This connection goes all the way back to the birth of cryptocurrency – through the support of early cypherpunks like Hal Finney, says Comito, who theorizes the common thread is transcending boundaries, whether financial or biological, to lead humanity to a brighter future.
The potential of blockchain technology to drive longevity research goes far beyond raising money, particularly with respect to decentralized science, or “DeSci.” “There are promising potential treatments for Alzheimer’s which lack traditional profit motives, for example, ones that can be developed with blockchain-based crowdsourcing,” he said.
Therapies like flickering lights and sounds at certain frequencies could remediate dementia. Comito himself is working on such an approach with his colleagues at Lifespan.io – aiming to launch a first-in-kind decentralized clinical trial to test non-drug interventions for Alzheimer’s disease, with the blessing of government organizations like the National Institute of Health.
“There have been very public failures on the treatment of Alzheimer’s with traditional approaches,” Comito said. “Over a trillion dollars has been spent on research, with strikingly little progress.”
He said a non-drug solution discovered by a decentralized group of people on the internet would be a “complete earthquake.” Decentralized science, powered by groups like decentralized autonomous organizations (DAOs), can also overcome issues associated with traditional research institutes.
DAO governance promotes transparency and rewards productivity, aspects which can be instrumental in advancing longevity research.
“I think we tend to view organizations like the FDA and the NIH as being calcified and anti-crypto,” Comito said. “But my experience has been that leaders of such organizations actually want us to shake things up.
With the help of blockchain technology, we can achieve medical advances so powerful and undeniable that existing systems will have no choice but to change.”
Crypto Will Power Further Innovation In The Space
Comito acknowledges that crypto has some perception issues to overcome in the wake of the FTX collapse. However, he sees that as more of a product of the overall market. While it is true that it’s hard right now for charities to raise money, Comito is optimistic that we will see a future filled with exponential growth.
“Last year, a number of charities started dipping into crypto because the market was hot,” he said. “They’ve drunk the Kool-Aid, and now’s the time to build and find out who’s truly getting something interesting done.
When the market recovers, these projects will be in great shape. We’re building tools to do something amazing in the next several years.”
Organizations like Endaoment and The Giving Block are laying the foundation for a philanthropic shift. In the longevity space, VitaDAO and the Longevity Science Foundation continue to fund cutting-edge research in the quest to help extend our lifespans and enrich our health.
This could also look like crypto and blockchain technology eventually playing a role in streamlining data interactions which will have a role in facilitating longevity.
We are fast approaching a world where everyone can own their data, carrying it in their healthcare wallet. This can help eliminate bloated industry architecture that relies on paper records in so much of the world.
In turn, this can accelerate opportunities for people to participate in clinical trials, reduce costs, facilitate payments to participants, limit emission-heavy travel, achieve diversity goals, and lead to more standardized data.
“Concerns have to be managed, but that is something we can do together as a crowd, which we consider a DAO of humanity,” Comito said. “Blockchain technology is not going away, and will have a key part to play in a future where all people will have sovereignty – not only over their currency and data but over their healthy lifespans as well.”
Updated: 1-30-2023
VitaDAO Closes $4.1M Funding Round With Pfizer Ventures For Longevity Research
The decentralized autonomous organization told CoinDesk that crypto enthusiasts, including Vitalik Buterin, have long been interested in funding research to extend human life.
VitaDAO, a decentralized autonomous organization (DAO) focused on raising funds for research on extending human life, has closed a $4.1 million funding round with Pfizer Ventures and several others.
VitaDAO was started in 2021 by Tyler Golato of decentralized biotech protocol Molecule to fund longevity research. According to the organization, it boasts a community of over 9,000 “enthusiasts, researchers and contributors,” and deployed over $3.5 million to projects researching diseases of aging, cellular biology and repairing age-related damage to the body in 2022.
The latest funding round includes pharmaceutical giant Pfizer Ventures; venture fund Shine Capital; investment group L1 Digital; decentralized science organizations BeakerDAO and Spaceship DAO; and longevity enthusiasts including former Coinbase Chief Technology Officer Balaji Srinivasan and Retro Biosciences’ Joe Betts-LaCroix. Previous investors include Ethereum co-founder Vitalik Buterin.
According to VitaDAO, Pfizer is the first pharmaceutical company to vote on DAO proposals and participate in the incubation and commercialization of VitaDAO projects.
Proceeds from the latest round will go toward funding longevity research projects and building out VitaDAO’s first biotech startup, slated to be announced in the first quarter of 2023.
In addition, the group plans to invest further in the “commercialization and licensing” of its non-fungible token (NFT) intellectual property (IP) through its partnership with Molecule.
VitaDAO’s deal flow steward Laurence Ion said that several projects funded by VitaDAO have moved to the next stages of development, including clinical trials with Mantis Photonics, which seeks to diagnose Alzheimer’s Disease through retinal imaging.
“There’s a move away in medicine to solve the symptoms of aging,” said Todd White, operations steward at VitaDAO. “There’s a lot of discussion around ‘is aging a disease?’ A lot of people are of the mindset now that aging and a lot of the diseases of aging … can actually be resolved.”
The group is governed by its native VITA token and holders can vote on what projects to support, while a group of “stewards” helps to “facilitate” the execution of those proposals, said White.
Ion told CoinDesk that with its democratic governance structure, the group aims to move beyond physical locations and is thinking about “a potential future sovereignty network.”
“I think we need a new paradigm,” he said. “We want to be a community as big as some countries and as resourceful as some countries.”
Crypto enthusiasts have long been into longevity science and delaying human aging. Coinbase CEO Brian Armstrong funded a company called NewLimit in December 2021 to “cure aging,” while former Ripple chief technology officer and current Stellar co-founder Jed McCaleb founded the Astera Institute to fund longevity studies.
White said the overlap between the crypto industry and the longevity research field is no coincidence. “If you think about how crypto started, it was about breaking down barriers and fixing a system that they perceived as broken. Well, the same applies in longevity.”
“There’s a lot more innovation and a lot more enthusiasm to try new things coming from the crypto community,” he added.
Updated: 3-2-2023
The Next Fountain-of-Youth Craze? Peptide Injections
As influencers share stories of physiological transformations, more people are seeking out experimental treatments: ‘If I can be a superhero and function at my absolute optimum, why wouldn’t I?’
After decades of turning to diet pills, steroids and plastic surgery to alter their bodies inside and out, people are increasingly open to an alternative method: injecting themselves with peptides at home.
Proponents say that peptides—a broad category of substances including FDA-approved drugs, supplements and experimental treatments—can help them build lean muscle, shed weight, increase energy and get a dewy glow.
Though the term has appeared on a range of consumer products for years, injectable peptides are getting more attention as celebrity doctors and influencers share stories of physiological transformations that go beyond diet and exercise.
These substances are being prescribed to patients by doctors as well as paramedical providers such as nurses and naturopaths, even though several in-demand peptides are not approved by regulators.
The lack of oversight has raised concerns about the purity of ingredients, improper dosing and unknown side effects. But advocates say they’re comfortable with any risks.
“I remember filling up my first syringe, and I was thinking, ‘Oh, my God, I can’t believe I’m gonna do this,’” said actor Brian Austin Green. Despite his fear of needles, he started taking peptide injections in 2021, when he was a contestant on “Dancing With the Stars,” under the guidance of William Seeds, an orthopedic surgeon.
Soon, he was taking three injections every morning for several months. He said that even with the show’s physical demands and grueling rehearsal schedule, “I so didn’t feel my age.”
This year, Mr. Green turns 50. He is in talks with Telegenixx, a telehealth startup where Dr. Seeds is a board member and co-founder, to start peptide injections again and promote the treatments on social media.
The startup’s chief executive declined to disclose which peptides it offers.
“People are more interested now in, What can I do to protect myself as I am aging, against disease, against stress?’” Dr. Seeds said.
Mr. Green, a father of five, said it’s important to him to be an active dad. “I don’t necessarily want to feel like I’m aging,” he said.
For years, peptides such as BPC-157, CJC-1295 and ipamorelin have been popular among bodybuilders and athletes who are seeking to speed up healing or build muscle.
The substances are being procured from compounders, a patchwork industry made up of state-licensed and FDA-registered providers, whose quality standards vary. Regulators have cracked down on their sales, and the World Anti-Doping Agency has expressly banned their use.
That hasn’t crimped broader interest. Doctors and medical spas nationwide are listing “peptide therapy” among their services and supplying injections to patients who are paying hundreds of dollars a month to take them at home.
Some consumers are going directly to suppliers of ingredients intended only for research in order to obtain the substances cheaply and without medical consultation, providers said.
In scientific terms, a peptide is a short chain of amino acids. There are thousands in the human body, and lab-produced versions that mimic them—insulin has often been cited as the first—marked great advancements in modern medicine.
Peptide-based drugs have been approved for treating diseases such as multiple sclerosis, Type 2 diabetes and obesity. But as the term “peptides” has entered the consumer vernacular, some providers have come to market them not as drugs but as rejuvenation treatments.
“We’re in this age of hyperfocus on wellness, antiaging, preventive health,” said Abraham Malkin, co-founder of two companies that offer at-home medical care. Both of them have six peptide injections on the menu, some of which Dr. Malkin takes himself. Only two of them have FDA approval, but Dr. Malkin’s website lists them for purposes outside those uses.
“We are very clear with our patients that peptides are generally not FDA-approved at this time, however we do talk to our patients to understand their health needs and enable them to purchase peptides that we feel can allow them to meet their health goals,” Dr. Malkin said.
Bec Donlan, a 36-year-old fitness coach and wellness consultant in Los Angeles, was introduced to peptide injections several years ago, as she sought to address autoimmune symptoms she believes were related to her breast implants.
“I’ve gotten back to normal health, but now I’m like ‘Great, so what else can I do?’” Ms. Donlan said. She is following a peptide regimen that she says has reduced inflammation, boosted her immune system and increased her metabolism.
Sometimes she posts about her regimen on Instagram, where she has more than 59,000 followers.
“If I can be a superhero and function at my absolute optimum, why wouldn’t I?” Ms. Donlan said.
Jamie Sherrill, a registered nurse who specializes in aesthetics, shared a TikTok last year in which she described “peptide therapy” as the future, holding a syringe in one hand and a vial in the other. She is known on the platform as Nurse Jamie.
Ms. Sherrill said her medical spa in Los Angeles County has a naturopath doctor who writes prescriptions for peptide injections. Speaking of the laser treatments and fillers she offers clients, she said, “I just think it all works so much better if you are taking a peptide.”
Some clients remain squeamish about at-home injections, Ms. Sherrill said. But she has seen interest rise over the past two years, especially as Type 2 diabetes drug Ozempic, which is taken by injection, has gained traction among people looking to lose some weight.
In addition to injections, some peptides are sold and prescribed as nasal sprays or capsules.
“I’ll have influencers or models come in and say, What can I use in addition to my Botox, microneedling or whatever else they are having done,” said Neil Paulvin, a regenerative-medicine doctor in New York City.
Tyler Jean, a 30-year-old naturopathic doctor in Los Angeles, started injecting the peptide BPC-157 daily—and sometimes twice a day—last fall alongside regular exercise and a healthy diet.
“I wanted to see how I could leverage some of these resources to better support my body’s ability to slow aging, optimize metabolism, maintain lean muscle,” said Dr. Jean, who is a patient of Dr. Paulvin. (Ms. Donlan, the fitness coach, is also one of Dr. Paulvin’s patients.)
Dr. Jean said that he stocks up on injections in New York, where they’re easier for him to get. After recently running out, he began taking BPC-157 in pill form.
Regulators have taken action against providers who sell unapproved peptides, including Tailor Made Compounding, which formerly supplied to patients of Dr. Paulvin.
In 2020, the Kentucky-based compounder pleaded guilty to a federal charge for distributing unapproved drugs, including peptides BPC-157, ipamorelin and melanotan II, also known as the Barbie peptide for its purported weight loss and tanning effects. The company was ordered to forfeit $1.7 million and placed on a three-year probation.
Now under new ownership, the company does not dispense the unapproved drugs referenced in the settlement, said Blake McLeod, the chief operating officer. Dr. Paulvin said he now works with multiple other compounders.
Generally speaking, pharmacies are not permitted to compound drug products with unapproved substances, the FDA said. A representative for the agency added that “compounded drugs should only be used to meet the needs of patients whose medical needs cannot be met by an FDA-approved drug.”
Unlike supplements that come in the form of capsules and powders, injections are subject to the agency’s premarket approval process.
While many of the peptides aren’t new, they have found new resonance in an era where influential podcasters are promoting nonconventional treatments to their audiences.
In March 2020, Upgrade Labs CEO and well-known biohacker Dave Asprey self-injected various peptides on his podcast, which included a video component. In an emailed statement, Mr. Asprey said, “You are in charge of your biology. Not your doctor. Not your government. You.”
Joe Rogan, who apologized in 2022 after critics said his popular podcast spread misinformation about Covid-19 vaccines, discussed his use of peptides on the show “Flagrant” the same year. Mr. Rogan did not respond to requests for comment.
Jordan Green, a biomedical engineering professor at Johns Hopkins University, said peptide-based drugs can sometimes be safer than other drugs meant to treat the same conditions. In 2011, he co-founded a startup that researches and develops peptide-based treatments for eye diseases.
“But, as with any drug, it needs to be carefully studied before the government and then physicians feel comfortable prescribing it to patients,” he said. Some of the results patients are describing could simply be placebo effects, he added.
Musician George Clanton, 35, learned about the at-home injections from watching the reality show “90 Day Fiancé,” on which a cast member was self-injecting a peptide called sermorelin.
“I looked it up and found a whole subculture of thousands of people discussing it every day on Reddit,” Mr. Clanton said. He ordered a few peptides online from a site that sells the substances for research purposes and indicates that they are not for human consumption. “I realize it’s stupid,” he said.
For several months before going on tour, he self-injected.
“It wasn’t like I was getting jacked,” he said, but “slowly, I just started to realize, I was waking up in the morning, ready to talk and think, instead of having to lay in the bed for an hour, and then have coffee and then mope around.”
Aside from some bruising and soreness, he hasn’t seen any downsides.
“People in my line of work, you know, sometimes there’s people injecting black-tar heroin into their bloodstream,” he said. “So I feel like this is so much less risky.”
Updated: 4-4-2023
Longevity Seekers Embraced This Drug. But Does It Actually Fight Aging?
An antiaging crowd latched onto the diabetes drug metformin hoping it would extend their lifespan. Studies give conflicting results.
Some health-obsessed Americans once thought they’d found a fountain of youth in a decades-old diabetes drug. Now, some are having second thoughts.
Metformin, which lowers blood-sugar levels in people with Type 2 diabetes, has been used as a diabetes medication in Europe since the 1950s and was FDA-approved in the U.S. in the 1990s.
Researchers have been investigating it for another possible use—to fight aging—since at least the 1980s, after observing that some people taking metformin appeared to experience other benefits.
The medication gained traction as a potential longevity drug in certain health-enthusiast circles after a 2014 study found that people with Type 2 diabetes taking metformin as their only treatment lived longer than those without diabetes.
Spurred by a selection of research that has suggested metformin could improve longevity, a growing community of self-described biohackers latched onto it, taking it even though they don’t have diabetes.
A tech-heavy crowd seeking to engineer improvements in their health through gadgets, data analysis and medications, they trade tips in forums on sites like Facebook, Reddit and chat app Discord.
As a treatment for people with Type 2 diabetes, metformin is considered safe. Doctors say off-label use doesn’t appear to have big risks, although some research has indicated the drug may be associated with an increased risk of Parkinson’s disease and may make it harder to build muscle mass through exercise.
However, the science behind its effectiveness as a longevity improver in healthy people is mixed, and some recent studies have been discouraging, giving some advocates pause.
Brad Stanfield, a primary-care physician in New Zealand, and a prominent figure in the longevity community whose YouTube channel on aging research has amassed more than 160,000 followers, made waves when he announced last year that he had stopped taking metformin.
Previously, he had touted its potential benefits to his followers and had taken the drug himself since 2019. He says he changed his mind after reviewing newer studies, including a 21-year randomized controlled trial that found metformin didn’t reduce cancer, cardiovascular disease or mortality rates in adults who were nondiabetic but at high risk for Type 2 diabetes.
“There have always been people spouting that they’ve found the fountain of youth,” Dr. Stanfield says. “It’s alluring, and you want to believe it.”
A study published last year that attempted to replicate the 2014 study linking metformin with longer lives found the drug wasn’t associated with a lifespan equal to or better than that of the general population.
Other studies of people with diabetes and in animals have shown conflicting results on metformin’s potential to increase longevity and delay age-related disease.
James D. Miller, a professor of economics in Northampton, Mass., says he started taking the drug about 10 years ago after reading about the 2014 study linking metformin with longer lives.
But he halved his daily dose after watching Dr. Stanfield’s videos on why he changed his mind about metformin.
“It is deeply annoying that I don’t quite know what to do,” says Dr. Miller, 56 years old. “I don’t know what’s optimal.”
Metformin still generates plenty of interest in the longevity world. Many people in this community work in technology, engineering and academia, and are naturally data-focused.
They enjoy experimenting with health trends, from daily cold showers to intermittent fasting, by analyzing research before it reaches the mainstream. They help each other with wellness routines and discuss new studies.
“Any tips to handle insomnia during fasting?” one Facebook user asked in an antiaging group called “End Aging Now” in January. “What protocols does everyone follow for hot immersion?” another inquired in a separate group in March. “What is the current thinking on metformin?” another commenter wrote elsewhere.
Adam Bataineh, a physician and founder of Numenor Health, a U.K.-based telehealth company focused on longevity that launched in October 2022, says roughly half of his patients ask about metformin.
His client base consists of people he calls “optimizers,” most often men in their late 40s to 50s who are starting to feel the effects of aging. Dr. Bataineh says he doesn’t take metformin himself.
Lee Spaziano, 49, an electrical engineer in Orlando, Fla., says he started taking metformin in 2021 after reading about the drug in a book by Harvard longevity researcher David Sinclair.
He was so convinced, he says, that he bought a second copy, a bookmark, and a bow and dropped it off at his doctor’s office with the sections on metformin marked. He got a prescription.
Recently, research suggesting that metformin might reduce the benefits of exercise has given him pause. Mr. Spaziano still takes the drug, but tries to compensate by lifting weights several times a week and getting plenty of protein.
Dr. Sinclair, professor of genetics and co-director of the Paul F. Glenn Center for Biology of Aging Research at Harvard Medical School, says he still takes metformin as prescribed by his doctor and monitors his blood work.
He says he doesn’t have Type 2 diabetes but his family has a history of the condition.
He believes the potential health benefits beyond treating or preventing diabetes outweigh the risks, but cautions against a blanket endorsement.
“What I do doesn’t mean others should simply do the same thing or that I encourage taking of medicines, unless a doctor approves it,” he says.
Adrian Massey, a 43-year-old UX designer for a tech company, was inspired to take metformin for longevity after reading research and observing that relatives who took it for diabetes appeared younger than their age, he says.
He struggled to get his primary-care doctor to supply the drug and opted to get it through a telehealth platform.
A bout of nausea in the first few weeks of taking the drug also did little to discourage him, but the muscle cramps that followed for the next month or so proved too much to ignore. He discontinued the drug.
“I was like, ‘This is too painful,’” he says.
Updated: 4-21-2023
Mind Aging Clock Reports Redefine Wellness And Resilience
Simple, highly scalable, cloud tech can provide accurate and comprehensive assessment of psychological age.
Deep Longevity has today launched its Mind Age clock on the SenoClock platform. Deep Longevity is hoping MindAge will be a breakthrough in the field of aging research, providing individuals with an accurate and comprehensive assessment of their psychological age.
Deep Longevity defines mind age as “the mental state most typical for people of a certain age group” – chronological age, as our readers will know, is simply your age in calendar years.
A higher mind age can be indicative of being in a poor psychological state – this can manifest as stress, an inability to form stable relationships or low self-esteem.
A lower mind age when compared with chronological age, on the other hand, can indicate a tendency to explore new ideas, meet new people and create a better future for oneself.
By baselining MindAge, Deep Longevity has created a quantifiable framework for improving mental health and wellness.
Longevity.Technology: The Mind Age clock uses machine learning algorithms to predict an individual’s psychological age based on data collected from a simple psychological test.
The resulting aging report aims to provide valuable insights into motivation, personal growth, environmental mastery, purpose in life, &c, and could revolutionize the mental health industry and the way the way individuals assess their psychological age and wellbeing.
Hosted on the SenoClock platform, MindAge aims to help individuals gain personalized guidance on how to improve their confidence, productivity, happiness and longevity.
With tech available over an API, the Mind Age clock incorporates advanced machine learning algorithms that processes responses to a predetermined set of survey questions to generate an individual’s psychological profile with high precision and accuracy; it then recommends a set of actions to improve resilience and wellbeing.
MindAge reports are organized in sections, each one covering an aspect of mental health and offering ways to improve a user’s psychological well-being:
Deep Longevity is aware that the mental health crisis is a burgeoning problem across the world – a situation only worsened by the pandemic and its aftermath. The company hopes MindAge will be able to change that reality for the better.
Deepankar Nayak, the CEO of Deep Longevity, said: “MindAge reports represent a major breakthrough in our understanding of the human mind and offer unique insights and motivations to improve wellbeing and resilience.
By taking a holistic approach to mental health and providing specific, actionable results, they have the potential to transform the way mental health consultations are conducted, and ultimately improve the lives of millions of people around the world.”
The MindAge algorithm creates a psychological profile, and the report generated uses the popular Ryff Scale to depict wellbeing on six distinct parameters.
This allows users to see how individual facets of their personality affect their psychological wellbeing and to what degree, illustrating areas of life in which they are doing well, as well as where they need to improve.
Fedor Galkin, the Director of Research at Deep Longevity, says: “By dwelling into the specific effect of each parameter, these reports provide a quick and simple analysis of the current mental state of the user. It will not only help a mental health professional understand their patient better but also offer personalized care based on the predictions.”
The MindAge report is able to generate personalized recommendations and simple steps for improving the mental health of individuals, and it this level of personalization was a key parameter in MindAge’s design.
Deep Longevity is well aware mental health care can not afford to take a ‘one-size-fits-all’ approach; no two people dealing with mental health will have the same experience or issue, and one person’s path to mental wellbeing have a different start and end point from someone else’s.
Indeed, this need for an individual approach is one of the reasons mental healthcare can be so expensive.
Under the hood, MindAge profiles people into different psychological types. The algorithm projects the test results on a 2D heat map, which allows the AI to pinpoint exactly where a person stands on their path to genuine happiness and longevity and where they should be heading.
A self-organizing map (SOM) then generates the optimal self-improvement route unique to the individual with a set of recommendations.
With a psychological age and wellbeing report being generated with minutes, Deep Longevity hopes the Mind Age clock will enable people to quickly start taking positive steps to improve their situations and change the way the mental health industry operates for the better.
Updated: 4-5-2023
Pfizer-Backed VitaDAO Votes On Creating For-Profit Company To Fund Longevity Research
The DAOs native token, VITA, rose 3% on news the community was considering the proposal.
Members of Pfizer-backed VitaDAO are voting on whether to create a for-profit company to secure and distribute funding for longevity research, the collective tweeted on Wednesday.
The company, to be called VitaTech, would leverage public funding to license longevity technologies developed in U.S. universities and support their continued development.
The funds would come from research foundations and government grants such as the Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR).
“To make up for [VitaDAO’s funding] shortfall, we are currently discussing a proposal to establish a for-profit US-based company that will bridge the gap between research and commercialization,” the collective tweeted.
The company will operate as a subsidiary or affiliate of VitaDAO. It also aims to serve as a launchpad for startups, helping research grant holders commercialize their tech, according to the proposal.
The proposal will enter a 10-week execution phase between April 3 and June 12, 2023, if approved.
VitaDAO raised $4.1 million in a funding round led by Pfizer Ventures with participation from several other investors in January. Pfizer is the first pharmaceutical company to weigh in on decentralized autonomous organization proposals and participate in the VitaDAO project’s incubation and commercialization.
VITA inched up 3% in the past 24 hours, according to CoinDesk data.
Updated: 4-26-2023
What AI Technology Is Doing For Longevity Now
Most of these applications involve analyzing large datasets.
In March 2023, MIT Technology Review revealed that Sam Altman, the CEO of OpenAI (ChatGPT), was the mystery investor behind the $180 million investment into stealth startup Retro Biosciences, a biotech company with the ambition of “adding 10 years to the human lifespan.”
This investment marks the latest tech entrepreneur expressing their interest in longevity science and a new connection with innovative AI technology.
According to February 2023 reports, AI is continuing to gain traction in healthcare applications. Currently, the market is estimated at $14.6 billion (USD) with a compound annual growth rate (CAGR) of 47.6%, with solutions spread across various healthcare fields, such as patient data and risk analysis, precision medicine, cybersecurity, lifestyle management, and drug discovery.
AI Is Currently Being Used In Longevity And Healthcare
The increasing convergence of AI technology and longevity science is sparking advancements in the sector, with established businesses, start-ups, and researchers utilizing the technology.
Most recently, scientists explored how ChatGPT, an AI-based language model, was able to predict Alzheimer’s in 80% of cases when analyzing speech. However, it is not the only implementation.
Due to its capabilities of analyzing a vast range of data, AI is proving instrumental in the discovery and development of new compounds.
One such application is Insilico Medicine’s ChatPandaGPT integration, which allows researchers to ‘talk’ to its PandaOmics target discovery platform, thus analyzing and navigating large datasets in order to discover new biomarkers and therapeutic targets.
Atificial intelligence models are currently being used to conduct genomic analysis and identify specific genes associated with healthy human lifespan.
One such project is Calico Labs’ collaboration with the well-known platform AncestryDNA, which analyzes a vast range of data to establish hereditary factors in longevity.
Personalized Medicine
By using AI technology, researchers may be better able to detect iomarkers for disease early, facilitating prompt interventions.
BioAge Labs’ partnership with Age Labs AS seeks to analyze samples and health records from the Nord-Trøndelag Health Study (HUNT) biobank to develop novel therapeutics.
The precise impact of diet and exercise on the individual level is still not completely understood, and AI may change that by analyzing a wider range of data.
Nutrino’s personalized AI platform is a predictive glycemic response algorithm that can help optimize eating habits and potentially reduce diabetes.
On a similar note, pecision medicine allows for tailored medical solutions. AI can be employed to analyze relevant data and help design and deploy these strategies.
For example, Deep 6 AI’s clinical trial matching system connects participants, patients, and researchers for clinical trials in order to broaden databases.
Occasionally, AI has been known to spot what a human cannot due to its ability to analyze data more closely and at greater volumes.
Zebra Medical Computer Vision AI medical imaging tool can be used to analyze data, including medical imaging, to diagnose diseases, such as bone, liver, lung, and cardiovascular illnesses.
This start-up was recently purchased by Nanox for a rumored sum of $200 million.
Using AI In Longevity Research
Recent advancements in the sector and widespread application across various industries have shown the technology to be effective.
Although each AI implementation is different, in longevity, it is finding its application due to its capabilities for analyzing and working with the immense range of data in the healthcare sphere, allowing researchers to identify patterns, relationships, and evaluate factors in age-related diseases.
In turn, they are better able to develop potential solutions and test them, at least in the initial stages, to ensure their feasibility.
Despite the achievements, AI isn’t a flawless solution, and people who apply the technology in their work are advised to do so with caution to ensure that any solutions created operate efficiently and can be used responsibly. For example, biased data introduced into AI datasets can discriminate against or favor certain groups.
In addition, such models may contain incomplete or inaccurate data, making them ineffective.
For AI in longevity to work effectively, it must have access to expansive datasets. This creates an issue wherein sensitive data could be breached or exposed.
In addition, it raises concerns of consent, as people may be unaware of when and how their data is used. These risks could be minimized by the responsible application and security of AI.
Although this isn’t exclusive to AI, the risk of geographical and economic disparities in access, as with most state-of-the-art technology, means that access may be limited.
This raises ethical concerns about how datasets are used, whom they are used for, and who will benefit from the solutions. As artificial intelligence technology advances, more benefits and challenges to its usage will become apparent.
Summary
Recent implementations of artificial intelligence in the longevity sector have presented some impressive results, including the power to harness data in order to deliver tangible suggestions for therapeutics development and to analyze medical images.
As AI continues to advance, its potential appears to grow in tandem.
However, scientists should be careful to avoid data-related ethical concerns and pitfalls when onboarding this new technology.
Updated: 4-28-2023
FDA Approves First-Ever Fecal Transplant Pill To Restore Gut Bacteria
According to the findings, approximately 88 percent of patients who followed the pill regimen did not experience reinfection after eight weeks.
The Food and Drug Administration (FDA) has announced the approval of the first-ever pill for fecal transplants, marking a significant milestone. Developed using healthy bacteria found in human waste, the pill could be an effective tool to fight gut infections.
A fecal transplant is a procedure that relies on the stool of a healthy donor to restore gut health and prevent bacterial reinfections in the respective patient.
Seres Therapeutics, based in Massachusetts, developed the new pill after rigorously testing this version of the stool-based treatment.
The FDA approved the prescription of pills based on a study of nearly 180 patients. According to the findings, approximately 88 percent of patients who followed the pill regimen did not experience reinfection after eight weeks.
On the other hand, reinfection occurred in approximately 60 percent of those who received dummy pills, the Associated Press reported.
Pill For Repeat Infections
The pill was created for people at risk of reinfections caused by the bacteria Clostridium difficile (C. diff). This bacteria frequently causes symptoms such as severe nausea, stomach ache, and diarrhea.
The FDA has approved the pill for adults over the age of 18, and older. Patients who have previously received antibiotic treatment can use the capsules. The drug treatment, known as Vowst, consists of four daily pills taken for three days in a row.
According to a report, this bacteria’s recurrence poses a significant risk of death, causing between 15,000 and 30,000 deaths per year.
Previously, antibiotic treatment demonstrated that bacteria can be killed, but it also destroys other healthy gut bacteria. As a result, people could become more vulnerable to gut infections in the future.
What Is The Procedure Like?
According to the company, the stool is thoroughly tested for viruses, infections, and parasites. Individual donors are also screened for any health risks or medical conditions.
After the results are obtained and the stool sample is approved, the experts remove the waste to isolate the healthy bacteria while killing the unessential ones.
A single stool sample can result in the creation of thousands of pills. Overall, this makes the fecal transplant process more efficient than it was.
The FDA warned in its approval announcement that the drug “may carry a risk of transmitting infectious agents. It is also possible for Vowst to contain food allergens,” according to AP.
Individuals who are ineligible for this pill will continue to receive fecal transplants. Since 2013, OpenBiome, one of the largest stool banks in the United States, has supplied up to 65,000 stool samples.
According to experts, this new pill method makes fecal transplants easier as well as less invasive. Ferring Pharmaceuticals’ treatment was also approved by the FDA last year, but it must be administered via the rectum.
Updated: 6-12-2023
What If The Most Powerful Way To Live Longer Is Just Exercise?
People are chasing longevity with ice baths and supplements. But the benefits of exercise have years of science behind them.
Longevity researchers have spent decades hunting for a magic pill to slow the aging process. But the best solution—at least for now—may be the simplest one: Move more.
No single thing—whether it’s regular cold plunges or off-label drugs and supplements like metformin, rapamycin or taurine—has a track record that can match exercise’s in terms of protecting against age-related diseases and helping people get more from their later years, a vast body of research shows.
The muscle and bone growth stimulated by exercise can help older adults maintain their independence, lessen fatigue and protect against bad injuries from falls, the leading cause of injury-related death among those over 65.
Regular exercise can reduce the risk of developing certain age-related diseases, including Alzheimer’s, cancer, diabetes and cardiovascular disease.
“It’s really remarkable how many of these different hallmarks of aging exercise can target,” says Nathan LeBrasseur, an exercise and aging researcher and director of the Robert and Arlene Kogod Center on Aging at the Mayo Clinic.
LeBrasseur co-wrote a study in 2021 that showed a structured exercise program reduced a key marker of aging, cell senescence. Senescent cells stop dividing as they age and contribute to multiple age-related diseases.
How Much Exercise?
Any amount of physical activity can help extend a person’s life, research suggests, especially for people who currently are doing very little. Federal guidelines recommend that adults get at least 150 minutes of moderate-intensity exercise a week.
A team of researchers who analyzed data on more than 650,000 adults over about a decade found that, compared with those who were inactive, those who got about half the government’s recommended physical activity added an average of 1.8 years to their lives.
Those who exercised for roughly five to eight hours weekly gained an average of 4.2 years.
“When you think about that, in terms of how many years you’re gaining per how many minutes of activity, it’s a very sizable yield,” says Steven C. Moore, the study’s lead author and senior investigator at the National Cancer Institute.
Lifespan benefits persisted across age groups, and for people who were classified as overweight.
The study, published in 2012 in the journal PLOS Medicine, found that those who were active and moderately obese gained about three years of life expectancy after age 40 compared with those who were normal weight but inactive.
More recent research examining physical activity and death rates has backed up those findings.
Exercise helps fight aging in a number of ways, including by improving immune function, reducing inflammation and increasing insulin sensitivity, research has found.
Beyond The Fairway
Tennis lovers, rejoice! Racket sports, including tennis, squash or racquetball, as well as running and walking were found to have the biggest benefits in a separate study Moore co-wrote on types of physical activity and mortality risk.
Exercise can help your memory and learning ability, too. Moderate-intensity exercise is linked to an increase in cerebral blood flow and brain glucose metabolism, which are connected to cognitive functions, says University of Wisconsin neuroscientist Ozioma Okonkwo, who co-wrote two studies on the subject.
“[Exercise] is one of the few things that the scientific literature is just unequivocal about,” says Dr. Christin Glorioso, a neuroscientist and co-founder of longevity biotech startup NeuroAge Therapeutics.
Glorioso, whose company is working to identify a drug that can treat neurodegenerative disorders, runs, hikes and sometimes swims in the San Francisco Bay near her home.
The exercise is partly to try to prevent Alzheimer’s disease that runs in her family, she says.
Theory, Meet Practice
Many self-described biohackers—those drawn to experimental lifestyle changes and medications like metformin and rapamycin to try to engineer longer, healthier lives—count exercise as the most important tool in their arsenal.
Rich Porter, a 42-year-old tech entrepreneur, this past year took his workout routine from what he describes as nonexistent to an hour every day, alternating between cardio and strength training.
His interest in longevity began about a year ago when he started treating wrinkles with Botox injections.
“I thought, that’s really just a superficial solution to a superficial problem and there’s actually something larger that I need to be looking at,” Porter says.
“I’d like to be able to do the things I love as long as possible, and then hopefully just die.”
Porter motivated himself to work out by picturing the things he hopes to be able to do at age 90, such as hoisting a carry-on suitcase into an airplane’s overhead compartment.
Exercise is always important, but becomes especially important after middle age when muscle mass and basal metabolic rate, or the number of calories the body naturally burns at rest, start to decline, aging researchers say.
Doctors and scientists generally recommend following the federal guidelines for time and intensity of training, including a mix of endurance and strength-training exercises.
Strength training becomes especially important for people in older age, says Mayo Clinic’s LeBrasseur.
Alexander Boldizar, a 51-year-old writer in Vancouver, British Columbia, averages two workouts a day, often including an hour of “zone two,” or moderate, steady aerobic exercise, followed by an hour and a half of Brazilian jujitsu class.
It’s part of an extensive longevity regimen that includes off-label drugs like rapamycin and acarbose, a diabetes drug, regular sessions in his home sauna, and a smart bed that tracks body movement and heart rate.
“You’ll see a lot of people in the longevity groups that say, ‘Pills are easier,’ but I think that’s a mistake,” says Boldizar. “I think it’s really important to get the lifestyle stuff down first.”
Updated: 6-25-2023
Should You Follow Bryan Johnson’s Blueprint? Here’s How You Can Allegedly Slow Down The Aging Process
Ex-Silicon Valley entrepreneur Bryan Johnson is spending $2 million each year to “biohack” his body with the goal of essentially becoming 18 years old again, but his Blueprint method has been critiqued for being far too extreme. Recent anti-aging research suggests there are much simpler ways to turn back the clock, for much cheaper.
How healthy is your cellular routine? Are you maximizing the functions of your mitochondria (the powerhouse of the cell) with the right supplements? Biohacking, or health optimization, is beyond fascinating, if not a bit frightening, when it veers into transhumanist territory.
In all likelihood, this industry will become more popular with the recent rise of improved machine-learning software (artificial intelligence) coupled with the puzzling but technologically promising developments in quantum computing.
Fierce levels of competitive behavior are built into our natural disposition as human beings, which is why you’ve started to see anti-aging biohackers who were once jogging down their research paths move into a full sprint – like former Silicon Valley executive Bryan Johnson.
Recently, Johnson has made many headlines for turning his life into a questionable, extreme anti-aging experiment. He’s opened up his journey to see just how well humans can truly reverse their epigenetic age for the general public to ooh and ahh over.
Johnson, a 45-year-old man, claims both that his biological age is roughly five years younger and that every 12 months moving forward he only ages nine months.
How This Silicon Valley Man Became an “Otherworldly” Lifeform
Caroline Graham at the DailyMail said that there’s “something otherworldly” about Bryan Johnson. When Graham met him in person, she couldn’t find one wrinkle or blemish, but according to Graham, Johnson’s alien-like visage couldn’t just be chalked up to cosmetic work or plastic surgery.
Whether or not that was a dramatization on her part (I mean, I look at his glamor shots embedded in the article and see plenty of natural wrinkling for a 45-year-old man around his glabella, crows feet, marionette lines, and neck, as well as sun spots and freckles dotted along his chest), Johnson has allegedly reversed his aging process so successfully through his bespoke program Blueprint that he could very well be on track to reverse his biological age to his goal age of 18 years old.
Though Johnson has made international news about his elite “fountain of youth” routine, one of his doctors, Oliver Zolman, has actually gone on record to lower expectations. Zolman praised the “small, reasonable results” but said that his team of doctors had “not achieved any remarkable results,” as Johnson’s biological age had actually only been lowered by roughly five years.
However, Zolman reported that his heart functions more like a 37-year-old man’s would.
This former Silicon Valley executive is sticking to his script, however, and has been documenting each step of his Blueprint journey for people to glean insights from and perhaps begin their own adventure to cheat Father Time.
His journey has led him to become somewhat of a viral meme, thanks to his questionable representations of his public persona and (intentional or not) holier-than-thou attitude.
Let’s get down to business – what does Bryan Johnson’s Blueprint for anti-aging actually consist of? To start, Johnson is a fan of high-intensity interval training, which he completes three days a week for at least an hour a day.
He additionally uses electromagnetic pulses to target his “hard-to-reach” muscles like the pelvic floor, a mechanism usually used by patients with incontinence issues or those who have just given birth.
He wakes up at 5:00 a.m. and goes to bed at 8:30 p.m., two hours after popping on his blue-light blocking glasses.
Johnson ingests enough vitamins and supplements to theoretically keep GNC in business (80 pills popped daily) and eats a strict, 1,977 calorie vegan diet broken up into three meals a day: his “Green Giant” morning smoothie, his lunchtime “Super Veggie” bowl and “Nutty Pudding” smoothie bowl, and his produce-laden nighttime salad, if not a stuffed sweet potato.
In total, he eats over 70lbs of vegetables each month, but he maintains a macronutrient breakdown of 19% protein, 33% carbs, and 48% fat.
In this process, Johnson maintains the following four principles: that modernity’s “self-destructive behavior is kinda insane,” that the body should be “empowered” to “speak for itself,” that “aspirations we need are beyond our imaginations,” and that we must “look in the darkness to avoid being blinded by the light.”
I’ll give credit where credit is due – he’s entirely justified in saying that “no one wants to be sick, diseased and sad,” so if he’s happy to become a human guinea pig in the name of science to perfect “exceptional health and vibrancy,” then all the more power to him.
With Johnson’s Silicon Valley bucks, he can certainly afford a team of 30 doctors to manage and measure his progress. It takes nearly $2 million each year in upkeep, as he subjects himself to frequent blood testing, MRIs, ultrasounds, urine and stool tests, and daily checks on his weight, BMI, body fat, blood glucose and oxygen levels, and temperature.
If something falls out of whack, his team of doctors is ready to adjust accordingly – like when his body fat plummeted to heart-health threatening levels below 3%.
He doesn’t drink, doesn’t smoke, is attempting to reverse his male-pattern hair loss and sun damage to his skin, and honestly the list could go on and on about each specific detail of his routine, but you can read more in depth on his completely public website.
Johnson’s model can’t be applied to women since it’s narrowly tailored to suit male physiology, but he has enlisted a female guinea pig to build out a companion blueprint. Johnson alleges that he is “actively participating in life with a vibrancy that far exceeds any joy I have had before,” despite this rigid-beyond-belief routine, because he’s trained himself to not want to deviate from his program. It’s aspirational in some ways – but that’s simply it.
Aging is part and parcel of the human experience, and no matter how many dollars you throw behind your own personal Blueprint, there’s really no cheating death – especially since God plucks people from earth at some of the most unexpected times.
It’s a morbid thought, but Johnson could optimize his routine so perfectly to walk back the clock and still not become the longest-living man since accidents outside our control can happen at any given moment.
Living forever doesn’t appear to be his goal, however; instead, he hopes to benefit mankind by finding real solutions to any number of the medical problems that plague our world.
“It’s not because I want to live for eternity,” said Johnson in his interview with Graham. “It’s not because I’m some rich guy seeking immortality.”
I’d wager most of you aren’t seeking immortality either, but still wish you could optimize your vitality to a point where you’re not regularly chasing sick care and instead can sustain a fulfilling lifespan through real healthcare.
Lucky for you, there’s no need to emulate such a rigid lifestyle as Johnson’s just to lower your epigenetic age.
There Are Much Simpler, Scientifically-Backed Ways To “Reverse” Epigenetic Age
In March, a study published in the peer-reviewed research journal Aging suggested that women could potentially reverse their biological age through an 8-week, methylation-supportive diet and lifestyle program.
If logic tracks, you could adopt these behavioral changes for longer than eight weeks as a sort of maintenance stage for youthfulness while aging.
The results are promising: Six female participants (yes, that’s a pretty small sample size, and the researchers did address this as a significant limitation) between the ages of 46 and 65 had, on average, a 4.6-year decrease in their biological age after the final blood tests were evaluated.
The researchers found that one of the six women actually lowered her biological age by 11 years. Let’s take a look at the blueprint for lifestyle changes these women made over the 8-week period to “reverse” their aging.
Every day, participants had to eat at least one serving of egg (totaling 5-10 eggs per week), liver or a liver supplement (totaling three 3 oz servings per week), 2 cups of dark, leafy greens, 2 cups of cruciferous vegetables, 3 cups of colorful vegetables, two servings of a low-glycemic fruit of choice, 1/4 cup of sunflower seeds (or sunflower seed butter), 1/4 cup of pumpkin seeds (or pumpkin seed butter), and one or two medium beets.
For their fill of dark, leafy greens, participants were recommended kale, spinach, collards, mustard greens, Swiss chard, or dandelion. Cruciferous vegetables could include broccoli, cauliflower, Brussels sprouts, cabbage, bok choy, radish, turnip, watercress, rutabaga, kohlrabi, and a few carry overs from the previous category.
For colorful vegetables, participants could vary their pickings, but this category excluded sweetcorn and white potatoes.
Each day, they were recommended to consume 6 oz grass-fed, organic, hormone-free, and antibiotic-free animal protein.
The “healthy” oils they were suggested to switch between were coconut, olive, pumpkin seed oil, and flaxseed oil. Participants were supposed to avoid grains, dairy, legumes, beans, added sugars, candies, and plastic food containers.
They also were instructed each day to eat two servings of foods that specifically support DNA methylation (the process controlling gene expression). One serving of methylation adaptogens could be 1/2 cup of wild berries, 2 cups of green tea, 3 cups of oolong tea, 2 medium cloves of garlic, 1/2 tsp of rosemary, or 1/2 tsp of turmeric.
There were a few more daily lifestyle changes that the six women undertook such as sleeping for at least 7 hours, fasting for 12 hours upon eating their final meal of the day, exercising for at least 30 minutes, drinking at least 8 cups of water, taking two capsules of probiotics, taking two servings of “greens powder,” and practicing the breathing exercise Steps to Elicit the Relaxation Response two times.
Interestingly enough, the six women only adhered to the program 82% of the time on average, but the researchers said that was normal to still achieve positive results.
They concluded that there are “widely accessible, cost-effective dietary and lifestyle interventions that are designed to support DNA methylation and are widely considered to be safe.”
Anti-Aging Tech Trends Will Leave You Strapped for Cash
To be frank, the conclusions of this case – despite its small sample size – are really important because the anti-aging industry is bigger now than ever before. There are so many profiteers looking to pocket a pretty penny after stoking fears about biological aging.
For as much as people may joke around about women hitting “the wall” after 25 or 30, that sort of rhetoric coupled with predatory profiteers in the anti-aging industry has essentially created a toxic complex.
Young people will do whatever it takes to push back on their natural aging process, and older people will drop fat stacks of cash on invasive and non-invasive procedures alike just to look young again.
Recently, a 14-year-old shared her own beauty routine meant to “slow down the aging process,” which she allegedly began doing at the very young age of 12.
While some of her habits are really harmless – and just good practices to have for your health in general – like supplementing apple cider vinegar, drinking green tea, being mindful of sun exposure, and using skin-friendly satin pillowcases, some people have pointed out that other steps in her routine may actually pre-age her young skin.
For instance, the girl uses Korean face masks twice a day, applies retinol (a skincare active meant to reduce the appearance of wrinkles) twice a day, and even puts extra retinol in her body lotion.
The global anti-aging market is currently projected to reach $120 billion by 2030, but as I’ve written before many of the products that tout themselves as effective fountains of youth are actually ineffective scams that unfairly trick women (and some men of course too!) into a Sisyphean, consumerist mentality.
Add your next 1.6 fl oz tub of $95 anti-aging moisturizer or 1 oz tube of $92 high-dose wrinkle serum to your online cart, and watch your skincare budget go through the roof.
There is truth behind claims that certain active ingredients can improve skin appearance, but that doesn’t mean you need to spend hundreds of dollars each month just on topical skincare alone.
That doesn’t even take into account the more invasive procedures that are not only a part of Bryan Johnson’s current Blueprint method, but that are dominating industry demand as well.
For instance, Johnson is trialing Cutera’s Excel V and Laser Genesis aesthetic laser treatment, which uses two different lasers on two different wavelengths to revitalize the skin.
Some sources say that cost per treatment can vary from $250 to $2,000, while others say it’s more like $275 to $1,000, and while some people can get results after one or two sessions, others may need up to five sessions.
One woman blogged about her experience using these type of lasers through six treatments to treat her rosacea, and while her results looked pretty promising she also made sure to caveat that her results won’t last forever.
Johnson has also been trialing “Fotona Dynamis Pro,” which is called the 4D facelift. It’s a non-invasive alternative to the popular plastic surgery procedure that also could treat sun damage, age spots, spider veins, and active acne, as well as potentially provide that lift that your typical, invasive facelift can.
Some cosmetic professionals recommend three or four 30-minute sessions spaced two to three weeks apart, and if you want to maintain your results, you’re supposed to come back every four or five months. Allegedly, Kim K has even been known to do this procedure.
Additionally, Johnson does MicroBotox injections for pore shrinkage, which can last up to six months and could cost around $450 per session. Johnson has been undergoing non-bleeding microneedling with hyaluronic acid serum, which can cost around $240 per session but requires about three to six treatments for results.
He does Scupltra injections which – if the rule of thumb is to be believed for “one vial per decade of life” – then he would need four vials at $700 a pop.
He has also been receiving the “vampire facial” known as PRF that draws your own blood to rejuvenate your skin. PRF can take around three to five treatments for noticeable results, ranging from $500 to $2,000 per treatment.
Johnson is also trialing the fat-transfer procedure called Renuva, which costs $2,125 per session on average, and full body LED light therapy, using a bed that can cost as much as $50,000.
If you ask me, though, a humble non-expert who considers herself quite observant, many of the skin rejuvenation or resurfacing before-and-afters I see are just not dramatic enough to warrant the cost and consistent upkeep.
I may not want to undergo Botox myself because of its many, many flaws (and hey, I’m not even past “the wall” yet anyway), but I can at least see visible, “anti-aging” results when people do go and get it done.
Similarly, when people save up money to get a really well-done facelift later on in life, I can see the return-on-investment. But neither of those procedures reduce a person’s epigenetic age – that can only be achieved through smarter lifestyle choices.
So is the self-harm and decay that a man like Johnson wails against actually inevitable? Should we biohack our bodies through borderline transhumanist measures just to turn back the clock?
Personally, even if I could afford it, the upkeep needed is just too rigid to feel like I would actually be living.
It’s all well and good that he feels fulfilled by being this guinea pig for progress, but it seems like we’ve already got most of the tools in our toolbox to age with grace if we just stick to a solid, time-tested routine.
Furthermore, I can’t imagine the stress to his mind, body, and soul that could potentially age him more than he thinks. Stress has long been studied as a surefire way to increase your epigenetic age and – especially in the case of women who are more prone to neuroticism and adopting obsessive personality traits – it certainly doesn’t foster a safe environment for your body to function in its natural harmony.
Yes, we’re built to face a certain level of stress, but too much of it can impair our fertility and our regular hormonal cycles, disrupt our gut health and potentially cause debilitating digestive issues, and might cause us to gain weight, among a slew of other issues.
Closing Thoughts
There’s something innately beautiful about the aging process, and if I am to be completely honest from an outsider’s perspective, Bryan Johnson’s current results in this age-reduction quest don’t look too hot.
He has sallow skin not too dissimilar from being afflicted by jaundice, his face has that same uncanny valley tautness that plastic surgery addicts are infamous for, he never goes one waking moment without being hooked up to a machine or having some device analyze his everyday bodily functions, and at what cost?
He has forced himself into a state of perpetual patient-dom. Even if he figures out exactly which habits correlate to the largest reduction in biological age, his research can never account for the sheer diversity of the human race – from inexplicable conditions that certain ethnic groups are more prone to like Tay-Sachs disease among the Ashkenazi Jew populations or sickle cell disease among those with African heritage, seemingly random (or allegedly not) genetic mutations that cause cancers, autoimmune disorders like Lupus, or neurological disorders like autism.
No, I won’t turn my nose up at some reasonably-priced, quality skincare products or treatments, but in general I don’t think it’s worth the extra stress in life to hyperfixate on staying as young as possible for as long as possible through most biohacking trends.
If we really want to “hack” our biology, we’d be far better off highlighting the importance of studies like the one found in Aging, as well as assessing our hormones because they play such an integral part in our body.
Updated: 8-26-2023
How We Age—And How Scientists Are Working To Turn Back The Clock
Insights into the underlying mechanisms of aging are revolutionizing the hunt for longevity treatments.
Scientists obsessed with aging are sketching a road map of how our bodies change as we grow old in the hopes that it will lead to treatments that could help us live longer, healthier lives.
They call this road map the “hallmarks of aging”—a set of biological features and mechanisms linked to our inexorable march toward death. Over the past decade, the hallmarks have helped guide the development of drugs that clear away cells that have stopped dividing and gene therapies that appear to restore cells to a more youthful state.
Scientists in Europe codified nine hallmarks in a 2013 paper in the journal Cell that is widely cited in the aging field. They include: shortening of telomeres (DNA segments at the ends of chromosomes); cell senescence, when cells stop dividing; and breakdowns in how cells regulate nutrients.
The hallmarks appear to manifest with age and accelerate aging when enhanced. They are interconnected in ways researchers are trying to understand. Some believe this could unlock insights into why we age.
Scientists are getting closer to solving mysteries that have long vexed thinkers. Gilgamesh, the titular character of an epic poem etched some 4,000 years ago on clay tablets, was obsessed with overcoming mortality. Chinese Emperor Qin Shi-Huang, who died in 210 B.C., drank mercury hoping to cheat death.
“Aging has excited the imagination throughout the history of humankind,” said Carlos Lopez-Otin, a biochemist at the University of Oviedo in Spain who co-wrote the hallmarks paper, “But it’s only recently that it has been subjected to profound scientific scrutiny.”
One hallmark attracting attention is changes in the epigenome, which consists of chemical compounds and proteins that can attach to DNA and regulate whether genes are turned on or off. Some researchers think an accumulation of errors in the epigenome drives aging and that removing the errors by “reprogramming” cells could lengthen life.
Shinya Yamanaka, a Japanese stem-cell researcher, shared a Nobel Prize in 2012 for discovering proteins that reprogram a cell’s epigenome to its embryonic state.
Scientists have used the proteins to extend the lifespan of mice and reverse blindness in mice and monkeys. Biotechnology companies such as Altos Labs, which Yamanaka advises, Retro Biosciences and Calico Life Sciences, part of Google parent Alphabet, are probing whether cellular reprogramming could extend lifespans or improve health.
Dorian Therapeutics and Senolytic Therapeutics are developing drugs that eradicate or prevent the formation of senescent cells, another hallmark, to see if that slows aging and mitigates age-related diseases.
Other scientists are experimenting with drugs targeting a hallmark called nutrient-sensing pathways: sensors that cells use to recognize fuel sources such as sugars and proteins.
The sensors become less effective with age and their deterioration has been linked to myriad health problems including metabolic disorders and cancer.
Nutrient-sensing pathways are affected by restricted diets, which research shows can bolster longevity. Diets that significantly cut calories increase lifespans and improve health, studies have shown. One theory is that calorie restriction stresses cells and increases their resilience.
Drugs including rapamycin that appear to mimic the effects of calorie restriction have increased life expectancy in mice. Rapamycin, which blocks a type of nutrient sensor, is typically taken by organ-transplant recipients to suppress the immune response.
The Dog Aging Project, headquartered at the University of Washington, is testing whether rapamycin can extend lifespan in pet dogs. AgelessRx and researchers at Columbia University are among the groups helping to develop clinical trials to test rapamycin’s antiaging potential in people.
A popular theory that emerged in the past century held that telomeres, another hallmark, could offer a silver-bullet solution to aging. Researchers who discovered the molecular nature of telomeres and telomerase, an enzyme that can maintain or extend their lengths, won a Nobel Prize in 2009.
Telomeres shorten as people age, and shorter telomeres appear to be associated with disease, studies show. But it isn’t clear that extending telomeres would lengthen life. Activating telomerase can allow cancer cells to replicate unchecked.
People who have unusually long telomeres have an increased risk of developing tumors and a blood disorder, according to a study published in May in the New England Journal of Medicine.
Still, some researchers and companies including Telomere Therapeutics and Geron are manipulating telomeres in efforts to treat cancers and other age-related disorders.
“It’s like Dr. Jekyll and Mr. Hyde: We want to stop telomerase in cancer cells…but elongating telomeres safely could be useful for a variety of age-related conditions,” said Jerry Shay, a molecular biologist at the University of Texas Southwestern Medical Center in Dallas and co-founder of Telos Biotech, which is lengthening telomeres to try to improve immune function in cancer patients.
Researchers of aging said the hallmarks have helped shape a shared vision for the field. But while they describe some of what happens during aging, they don’t explain why these changes occur, said David Gems, a geneticist at University College London.
They fall short of what philosopher Thomas Kuhn defined in the 1960s as a paradigm: shared values and ideas that explain a scientific phenomenon. “You can’t have a field without a paradigm,” Gems said.
The group behind the original hallmarks of aging suggested three more earlier this year based on subsequent research: chronic inflammation; imbalance in the microbiome, the community of microorganisms that live inside people; and defects in autophagy, a cell’s ability to recycle damaged parts of itself.
More hallmarks will likely emerge and others will be discarded or combined as research advances, said Danish geneticist Lene Juel Rasmussen, who suggested a few others with colleagues last year.
“The hallmarks are dynamic,” Rasmussen said.
Nine Hallmarks of Aging:
Here Are Some Of The Key Biological Changes Identified By Researchers That Appear To Happen With Age
Genomic Instability: As DNA damage builds up over time, mutations accumulate in the genome
Telomere Attrition: Telomeres, protective structures at the ends of chromosomes, have been found to shorten
Epigenetic Alterations: Processes that regulate whether genes are turned on or off can change over time
Loss Of Proteostasis: Machinery in cells that controls protein synthesis, maintenance and cleanup becomes impaired
Deregulated Nutrient-Sensing: Sensors that cells use to regulate fuel sources such as glucose become less effective
Mitochondrial Dysfunction: Mitochondria, the power plants of cells, can become damaged and dysfunctional
Cellular Senescence: Cells that stop dividing but don’t die accumulate as people age
Stem-Cell Exhaustion: Stem cells, which can develop into many cell types and serve as bodily repair systems, lose their regenerative power and other functions
Altered Intercellular Communication: Cells can lose the ability to properly communicate with each other
Updated: 8-31-2023
Saudi Arabia Is Dangling Billions For Research On Aging. Scientists Are Lining Up To Take It
The kingdom’s plans to put more than $1 billion a year into global efforts on longevity stir anticipation—and hesitation around the country’s politics.
RIYADH, Saudi Arabia—Vast oil wealth has enabled Saudi Arabia to establish dominant roles in global sports, electric cars and tech startups. The kingdom’s next target may be its most ambitious yet: extending the human lifespan.
Crown Prince Mohammed bin Salman, the de facto Saudi ruler, has allocated more than a billion dollars a year to an effort called Hevolution Foundation to develop new treatments for aging.
That could dramatically expand the available global funding for research on longevity biology, which now comes mainly from the U.S. National Institute on Aging.
The prospect of a huge surge of funding into the area, whose budgets pale in comparison to research on diseases like cancer, is causing a stir among scientists who study aging.
“People in the field are kind of holding their breath to see how the money is going to be spent,” says Steven Austad, a researcher on aging at the University of Alabama at Birmingham and senior scientific director at the American Federation for Aging Research, or AFAR, a U.S. nonprofit that has received $7.76 million in funding from Hevolution.
The Saudi foundation’s chief executive, Dr. Mehmood Khan, says much of the initial grant money is likely to end up at universities and startups in the U.S., where scientists are trying to develop treatments that slow, prevent or even reverse the aging process for humans.
“It’s creating a more idealized funding situation,” says Martin Borch Jensen, chief scientific officer of San Francisco-based Gordian Biotechnology and president of Norn Group, a U.S. nonprofit with a grant program to which Hevolution has contributed $7 million.
For now, Hevolution’s biggest challenge is how to spend its money in a field that is still relatively small and in a world where many view anything Saudi Arabia does with suspicion.
Some institutions and individual researchers have been hesitant to establish ties with an absolute monarchy that brooks no dissent domestically and was spurned by the West after the killing and dismemberment of Saudi journalist Jamal Khashoggi in 2018.
Recent injections of Saudi cash into U.S. golf and global soccer have sparked accusations that the kingdom is deploying its wealth to paper over human-rights abuses and boost its global image.
Hevolution could face similar criticism but, still in its early stages, the project hasn’t attracted broad attention.
AFAR’s board had repeated discussions before accepting Saudi funding last year for 18 research projects in aging biology or geroscience but has since renewed its partnership for a second round of grants, according to officials there.
“In the beginning, people were skeptical, but I think a lot of that has kind of gone away because they see that we’re giving out the money,” says Stephanie Lederman, AFAR’s executive director. “We want to fund this science, it’s really important for the human race.”
Dr. Aditi Gurkar, an assistant professor of medicine at the Aging Institute, University of Pittsburgh, says she paused before applying for an AFAR grant funded by Hevolution but ultimately went ahead because of the Saudi organization’s collaborative and global approach to aging science.
In April, she received a $375,000 grant to study with a physicist how nanoparticles could help detect cell senescence, a process in which cells stop dividing. It’s an innovative idea that she says would be difficult to secure funding for from traditional sources in the U.S.
When Norn Group announced Hevolution funding, some people objected to the Saudi ties, Jensen says, but he pushed past it because the organizations share a common goal.
“Our focus is on achieving that mission and improving human health and flourishing, which we’re committed to whatever the state of public opinion,” he says.
There is also some concern that the Middle East’s turbulent politics or a sharp change in the Saudi crown prince’s personal predilections could cut off funding unexpectedly.
Hevolution—whose name is a combo of “health” and “evolution”—aims to have a global impact, as Saudi Arabia looks to expand its influence around the world under its 37-year-old leader.
Its mission could also resonate at home, where Mohammed has staked his credibility on boosting quality of life for a now overwhelmingly young population and building new industries away from oil.
The Saudi media ministry didn’t respond to requests for comment from the prince.
Established as a nonprofit in 2018 by Saudi royal decree, Hevolution doesn’t conduct its own research but instead aims to work with established institutions to pump money into underfunded areas of study.
Its chief scientific officer, Dr. Felipe Sierra, ran the aging biology division at the NIA for more than a decade, and its scientific advisory board includes Dr. Thomas Rando, who directs the stem-cell research center at University of California, Los Angeles.
“We’re sort of doing the nontraditional approach. Who else might be able to solve the problem?” says Khan, the chief executive. “One of our goals is to actually attract new scientists—in terms of entering science—and scientists from adjacent fields that may not have data but their technologies could be relevant to solving.”
Since starting operations in July 2022, the Saudi foundation has focused mostly on establishing itself, dispersing less than $20 million. Khan expects that to ramp up toward $1 billion within the next two to four years.
Initially, more of that money will go to research, but eventually the goal is for a roughly even split with investments into antiaging startups, he says.
Khan, who was chief scientific officer at PepsiCo, where he worked for more than a decade, says he wants to expand the field globally and pursue as many promising opportunities as possible.
“We’re not saying epigenetic reprogramming is our first priority or autophagy is our first priority or senescence is our first priority,” he says, referring to three cellular processes that many scientists link to aging. “We’re looking at all of them.”
He would like to see Hevolution help identify biomarkers to track aging and also fund early large-scale human trials for existing repurposed drugs to treat aging.
Those are two crucial areas that require substantial funding and are unlikely to generate quick profits.
Khan was born in Pakistan and grew up in England before moving to the U.S. He spent 1987 in Saudi Arabia and helped set up a medical residency program at the kingdom’s leading hospital, where Saudi rulers receive treatment.
He later worked at the Mayo Clinic in Rochester, Minn.
“There’s a lot of snake oil” in the field, says Khan, 65, who also ran Boston-based biotech startup Life Biosciences for two years before joining Hevolution. “There’s loads of claims, and one of the things we always have to ask ourselves is ‘What is this based on, and is it legitimate? Is it anchored in real science?’”
Hevolution recently hired two investment partners and plans to announce its first direct investment before the end of the year after reviewing over 100 potential opportunities.
It has registered a nonprofit entity in the U.S. and a limited liability corporation with an office planned for Boston and others to follow in Europe and Asia.
Updated: 11-10-2023
How Pets Help Us Lead Longer, Healthier Lives
Research shows that canine companions can help with stress, allergies and even cardiovascular disease, in part by giving us a reason to focus on the future.
Dogs seem to make us healthier than we would be without them. Social psychologist Bruce Headey conducted a survey of Australian dog-owners and found that they take fewer trips to the doctor and sleep better than non-dog-owners. They are also less likely to be on heart medications.
It’s not that dog-owners are naturally healthier; bringing a dog into your life somehow brings these benefits along. James Serpell, a professor of animal ethics at the University of Pennsylvania, conducted a study in the U.K. that followed pet owners through the 10 months after they adopted their pet.
Among dog and cat owners, there was a significant reduction in minor health problems such as headaches, difficulty sleeping, indigestion and sinus trouble in the first month, and these changes lasted for the study’s duration.
Their scores on measures of general health also improved, and dog-owners increased their physical activity considerably.
Some of the health benefits of living with a canine occur deep down inside us. No matter how tidy your home may be, there’s some dust in the air, on the floor and on surfaces.
In a 2018 study by biostatistician Alexandra Sitarik and colleagues, researchers collected dust samples from 54 family homes—half of which had a dog and half of which didn’t—when the dog was initially brought into the home and a year later.
By the one-year mark, the presence of a dog in the home was associated with “a higher percentage of variation in bacterial dust composition,” including traces of Moraxella, Porphyromonas, Capnocytophaga, Fusobacterium, Streptococcus, and Treponema bacteria.
This isn’t a bad thing. On the contrary, what’s come to be called the “Microbiota Hypothesis” suggests that dust from homes with dogs may influence the development and response of the human immune system by changing the composition of the gut microbiome in ways that reduce risk for allergies and asthma.
A considerable body of research has found that young children who grow up with dogs in their households are less likely to develop allergies, eczema or asthma, which often occur together as part of what’s called the allergic triad.
The theory, according to allergists, is that early exposure to dog dander could induce a high-dose tolerance to allergens. By stimulating their immune system not to react to dog dander and other microbes carried by canines, growing up with a dog helps kids develop a greater tolerance for certain germs and airborne allergens, thus preventing potential allergies from developing.
Scientists believe this may be why kids raised with dogs have fewer allergies than those from pet-free homes. A 2018 paper by Bill Hesselmar of the University of Gothenburg in Sweden and colleagues examined data from more than 1,000 children, age 7 to 8, and found that among those who grew up without pets, 49% went on to develop allergies.
The rate dropped to 43% for kids with one pet and 24% for those with three pets.
When it comes to pain, having a dog doesn’t make you impervious, but it can make the discomfort more bearable. This is partly because having a canine companion provides a continuous source of meaning, connection and support.
Research led by Mary Janevic of the University of Michigan School of Public Health examined how older adults with chronic pain felt their pets affected them.
Participants reported that their dogs motivated them to get up and get moving, which helped alleviate their pain. The pets distracted people from their pain and generally improved their moods.
As Janevic noted in an interview, “Engaging in pet care can give a sense of daily purpose and routine that keeps a person going, even when they are having a pain flare-up. In this way, pets can be thought of as a natural resource for chronic pain self-management.”
These findings were echoed in a 2020 study by April DuCasse and colleagues at Florida A&M University, which found that people with chronic pain reported that their pets improved their mood, sense of hope, activity levels, comfort and functionality.
Besides having a positive impact on their human’s quality of life, the research found that canine companions provided some of the participants with “a reason to live and focus on the future” and “support that mitigates their suffering and enables them to live a more meaningful life.”
Research also suggests that having a dog is associated with lower blood pressure and cholesterol levels, a reduced risk of Type 2 diabetes and reduced physiological responses to stress. These effects may partly explain why dog ownership is associated with a 31% decreased risk of death from cardiovascular disease.
Across various age groups, multiple studies have found that our heart rate, blood pressure and stress levels tend to be lower when there’s a dog around.
A 1995 study by health researcher Erika Friedmann and colleagues examined the impact of a dog’s presence when young people age 9 to 16 were asked to read aloud.
The kids’ heart rate and blood pressure readings were lower when the dog was present, which the researchers interpreted to mean that it made the environment feel less threatening and more friendly.
The presence of other people doesn’t always have the same beneficial effects as the presence of pooches. In a 2002 study by social psychologist Karen Allen and colleagues, adults were placed in mentally and physically stressful situations.
They were asked to perform mental arithmetic and endure a “cold pressor” test, in which their hand was immersed in ice water to induce a stress response.
The participants endured these tests alone, with their spouse or friend present, with their dog or cat present, or with both a human and an animal companion. In all of the scenarios, the people who had their pets present had lower baseline heart rates and blood-pressure levels.
During the stressful situations, their blood-pressure and heart-rate increases were smaller, and they recovered faster, when their pets were present.
Of course, having a dog isn’t enough to prevent people from developing cardiovascular diseases. But when serious problems do arise, dogs can help us recover better.
Research by Mary Herrald of New Mexico State University and colleagues found that among people who have heart attacks, those who have dogs are significantly more likely to complete cardiac rehab, which typically involves physical therapy and education about how to manage risk factors and avoid everyday sources of stress.
What’s more, research by epidemiologist Mwenya Mubanga and colleagues found that people who have dogs survive longer after having heart attacks or ischemic strokes.
Besides having specific effects on health, your relationship with your dog may alter your attitude toward health and life in general. If you want to be around as long as possible to take care of your beloved canine companion, you may feel inspired to improve your lifestyle and stress-management habits.
You may feel motivated to take better overall care of yourself, whether that means taking your medication as directed, exercising regularly or going to bed earlier. Even if you’re not inclined to do these things for your own well-being, you may do them for the sake of your beloved pooch.
Updated: 11-27-2023
How Your Dog or Cat Could Help Speed Up Your Health Tests
The Mayo Clinic, Quest Diagnostics and others are exploring AI tools that build on work with animal data to train diagnostic algorithms for humans.
Imagine getting medical-test results within minutes or seconds, before you leave the doctor’s office, for even the most complicated diagnoses.
Steps toward that goal are happening in an unlikely place: The veterinarian’s office.
Researchers are using results from your pets’ tests for things like hookworms and other parasites to train artificial intelligence tools that not only speed up Fido’s results, but are informing human diagnostics, too.
The Mayo Clinic this year began using diagnostic algorithms developed by a Salt Lake City-area startup called Techcyte that uses its work with animal data to inform its AI development.
Mayo’s large-scale laboratory, which processes samples for its own patients and other health systems, used to have a two-week backlog of parasite samples. Now it can finish the 200 that arrive each day in 24 hours.
Big Testing Companies Such As Quest Diagnostics will start using Techcyte algorithms next year for fecal testing for parasites. And Techcyte says it is trying to win clearance from U.S. health regulators to allow smaller sites such as doctor’s offices to use algorithms in tests that would give patients results before their appointment ends.
Techcyte is also working with Mayo to develop AI-based cancer-detection tests. The AI can give pathologists and physicians more data to determine whether a patient has cancer, including doctors outside academic medical centers.
“We can produce results faster, but also the algorithm is unfailing,” said Dr. Bobbi Pritt, interim chair of Mayo’s Department of Laboratory Medicine and Pathology. “It doesn’t suffer from human error, and so it doesn’t get distracted. It doesn’t get fatigued.”
Veterinarians are providing the groundwork as they increasingly use artificial intelligence in caring for dogs, cats and other pets or livestock. Among those selling AI-based diagnostic tests for animals are Antech and Idexx Laboratories.
Drugmaker Zoetis, which manufactures pharmaceuticals for farm animals and pets, worked with Techcyte to develop a diagnostic device called Vetscan Imagyst. Zoetis says more than 3,000 veterinary practices in the U.S. use the device today for AI-based fecal, blood, dermatologic and urine tests.
The Imagyst digitizes the process of studying samples with a microscope while giving reliable results that come in a matter of minutes instead of having to wait hours or several days from lab technicians.
“The pain in this market is the same whether you’re a vet, a human lab, or an environmental lab,” said Ben Cahoon, chief executive of Techcyte.
“Looking at a microscope is a very difficult and tedious job, meaning you need someone who’s an expert. They are looking at a microscope literally for eight hours a day.”
The Imagyst looks like a traditional microscope but works as a digital scanner. A vet tech prepares a sample on a glass slide—that step can still take 10 minutes—and puts it on the machine, which uploads the image to Techcyte’s cloud.
The algorithm goes to work reading the image and generates results in a few minutes.
The most popular test, using fecal matter, is among the most common tasks performed by veterinarians; experts recommend that pet owners get their pets checked for parasites at least once a year.
A blood smear test, which can count red and white blood cells, and platelets, gives results in less than four minutes. Vets normally send blood samples out to labs.
To design its fecal test, Zoetis collected roughly 50,000 samples from veterinarians and laboratories around the world, many containing more than 100 parasites like hookworms or tapeworms.
After its scientists marked the parasites, the images were fed to Techcyte’s algorithm. The findings were validated against the findings of human scientists.
The Imagyst accuracy was comparable to findings of clinical specialists, according to Zoetis-funded studies published in the journal Parasites & Vectors beginning in 2020.
Joseph D’Abbraccio, a veterinarian about a two-hour drive from New York City in the Hudson Valley, runs his Imagyst devices about 20 times a day.
D’Abbraccio says his staff read the results on computers while caring for animals instead of waiting around in the lab. The fast turnaround on test results means pets don’t need to be hospitalized meanwhile, which can cost hundreds of dollars.
“I get a faster clinical judgment on the patient, which then in the end decreases my client’s bills, because the animal’s not waiting here for days for tests. I get results before lunch.”
Animal health wasn’t part of Techcyte’s initial business plan when it started in 2013 and focused on human health. Cahoon joined as chief executive three years later, when the company still hadn’t launched a product.
Attending a medical conference, he learned the animal health industry faced a backlog of studying stool samples—a “fecal tsunami,” he says he heard.
He thought developing tests for animals and the environment would be faster because they have fewer regulatory barriers. At trade shows, Cahoon wore a full-size poop costume pitching the company. Zoetis started working with Techcyte in 2019, and two years later became an investor.
Meanwhile, Techcyte continued researching AI for people, building on its learnings from animal health. For example, the framework for training the animal AI tests—evaluating different values and settings—was used for human tests as well, since they’re both microscopy.
The testing company ARUP Laboratories in 2019 began using a Techcyte algorithm for a human fecal test. Techcyte rolled out bacteriology tests last year at health systems like Mayo.
Techcyte’s research was getting noticed, including by Mayo’s Pritt. A 2020 peer-reviewed study in the Journal of Clinical Microbiology by researchers at Techcyte and ARUP found that the company’s tools were 98.88% consistent with conventional microscopy and more sensitive than humans.
Pritt said the animal-health success added credibility to Techcyte. “Looking for parasites and stool samples is very similar for animals as it is for humans,” she said.
At Mayo’s laboratory, the AI helped to slightly increase the positivity rate of samples to 5%, Pritt said. The AI also sped up the review of negative samples to 30 seconds, instead of five minutes done by a staff member.
Because most slides come back negative, workers no longer spend most of their time screening slides without parasites.
Technologists at Mayo still confirm the positives identified by AI.
Mayo is also working with Techcyte to develop a digital pathology program and AI-based tests on tissue samples instead of just liquids and cells, including tests for cancer, said Maneesh Goyal, chief operating officer of Mayo Clinic Platform, which invests in startup firms including Techcyte.
Mayo is in the process of digitizing some 25 million slides of tissue samples collected from patients, and for all patients in the future. These images will train the algorithms, which may be ready for use in care next year, Goyal said.
Updated: 12-19-2023
The Most Secretive Longevity Lab Finally Opens Its Doors
Retro Biosciences, a startup with $180 million from Sam Altman, has a simple and audacious goal: Add 10 good years to your life. And until now, we haven’t had a glimpse of its best ideas.
Joe Betts-LaCroix didn’t have time to wait for architects, construction workers or really any of the normal things that go into building a new office, let alone a new laboratory. It was May 2021, and he wanted to do experiments … lots of them … right away.
And so he and a small team of people took over an abandoned retail building in Redwood City, California, and filled it with shipping containers, which they’d soon fill with mice.
They built the heating and air conditioning system for their lab pods by hand and did the same with the air-filtration system and their precisely tuned mouse vivarium. At the end of two months, Betts-LaCroix’s team had its first experiments up and running.
“I was told by a nearby developer that’s been building a similar-sized lab for more than a year that they’re going to spend $15 million on it,” Betts-LaCroix says. “I probably spent, I don’t know, $200,000. I’d just rather figure out how to do it and do it in a way that works.”
The company Betts-LaCroix started, alongside the scientists Matt Buckley and Sheng Ding, is called Retro Biosciences Inc. and has a pitch that’s as ambitious as Silicon Valley gets.
It wants to give every human 10 additional years of healthy, vigorous life. To pull this off, and pull it off quickly, Retro has eschewed a number of biotechnology startup traditions.
Most notably, instead of chasing a single super-promising compound or treatment, it’s decided to pursue five tracks of research at the same time. It’s a high-risk, costly strategy made possible only by the company’s unusual backing. Retro has raised $180 million from one investor—Sam Altman, OpenAI Inc.’s co-founder and recently ousted and de-ousted chief executive officer.
The customs of the biotech field dictate more pragmatism and polish than with other tech startups.
In most cases, companies charge after what they believe might be a singular breakthrough, and the end goal, after toiling for years and years and performing a costly clinical trial, is often to sell whatever results to a pharmaceutical or medical device giant.
Things such as building handcrafted container labs and hopping from one promising lead to the next are simply not done.
Once upon a time, Altman and Betts-LaCroix had, in fact, discussed starting a smaller longevity-technology-focused company around a single therapy, but the more they talked, the more they became excited by other things, too.
“Usually in this field you get to pick one idea and spend nine years on it, and then, at the end, maybe it works and maybe it doesn’t,” Betts-LaCroix says.
“Sam was willing to do something different and throw lots of money at a bunch of things in parallel.” Betts-LaCroix describes Altman’s support as “lucky,” “freaking awesome” and “cool,” which is what anyone given $180 million to pursue their hopes and dreams should say.
Retro has operated in secrecy for most of its two-year existence, and this marks the first time the company has talked about its work in detail and opened its office and laboratories for perusal.
The company of about 50 people has small teams shooting for breakthroughs in autophagy (the removal of damaged cells), the rejuvenation of blood plasma and three research programs tied to what the biotech industry calls partial cell reprogramming.
Cell reprogramming is a process, proven out in numerous animal experiments, in which the cells of an older creature can be treated with a combination of proteins or molecules and turned into much younger cells.
Based on Nobel Prize-winning science, it’s something Retro and a handful of other startups consider the most promising longevity technology to yet appear.
Silicon Valley, more than anywhere else, has for decades been hoping not only to retard aging but also to find an actual “cure” for death. This goal has made the region and its technophiles the butt of many jokes and the target of criticism.
Betts-LaCroix, well aware of the history and the teasing, has tried to tamp down some of the most extreme longevity rhetoric. “People don’t want to die,” he says.
“They will latch onto something if given hope, which is in some ways the force that I’m fighting against. The science of this realm is not the cure to your existential crisis or your desire to avoid death altogether. There are lots of things that are going to kill you.
But this type of technology could extend healthy human lifestyles by years, and it’s an incredible gift to humanity. It’s worth working on.”
Retro and its peers really do think this time is different. Many researchers in the field contend that the science behind cell reprogramming, in particular, has been solved and that therapies are now an engineering problem.
They see full-on age reversal as not only achievable but also perhaps imminent. That’s why the race is on—and why you might want to build a lab in a hurry.
Betts-LaCroix, who’s 61, can sometimes be found pacing around the Retro offices with a 22-pound sphere of tungsten in hand. It’s about the size of an orange and awkward to support.
He seems to take some pleasure in passing it to guests and watching to see if they’re up for the challenge or if their wrists and elbows will immediately buckle as the ball heads toward the ground.
In the latter case, Betts-LaCroix will comment on tungsten’s high density (19.25 g/cm³) and periodic table naming origins (W, from the ore, wolframite), while his guest blushes and makes internal pledges to go to the gym more. (Not that this happened to me.)
The detailed talk about tungsten or the physics of HVAC systems reflects Betts-LaCroix’s autodidact nature, shaped and encouraged during an unconventional childhood in Oregon.
He grew up in the heart of the 1960s counterculture movement with parents who largely let their son do as he pleased and explore what he wanted to explore.
When, as a 7-year-old, Betts-LaCroix took apart the family’s sewing machine and refrigerator, that was considered just fine. And if he ran late to school, his parents would send a note asking for him to be excused on account of alien abduction.
“My father taught me you can redesign anything from first principles,” Betts-LaCroix says. “I was in a subculture where we considered ourselves to be redefining society. We were starting over from a blank slate and could make our own rules.”
Outside of physics and math courses, Betts-LaCroix hated high school and rarely attended class. He graduated with a D average, then spent the next six years living in a shared house with “musicians, artists and weirdos.”
Betts-LaCroix set up an electronics lab in the basement and performed a constant stream of electromechanical experiments. He covered his expenses by doing electronics, hardware and software work for local businesses and occupied the rest of his time by consuming huge quantities of books.
Eventually a girlfriend of Betts-LaCroix’s got into Harvard College, and he decided to follow her.
He spent a semester at a local college, buckled down, got straight A’s and submitted what he describes as an unusual transfer application that impressed the admissions office.
Once at Harvard, Betts-LaCroix found that his relationship with school and academics changed.
“It was like, ‘My God. This is a secret treasure trove,’ ” he says. “I could hang out with these brilliant professors and talk to them and learn from them. I wondered where this had been all my life.”
At Harvard he obtained an undergraduate degree in environmental geoscience while doing biophysics research at the California Institute of Technology in his spare time.
Later he built robotics systems at the Massachusetts Institute of Technology, all the while publishing a stream of papers across these fields in scientific journals.
After moving to Silicon Valley, he co-founded OQO Inc. in 2000, which was a much celebrated though unsuccessful maker of tiny personal computers, and then worked at the much celebrated though unsuccessful gene-sequencing startup Halcyon Molecular Inc.
In more recent years he’s been a prolific angel investor in startups and a well-known figure in biohacking, longevity and quantified-self circles.
Beyond OQO and Halcyon he spent time working at a couple of other technology companies, in the venture capital realm and as the chief technology officer of Vium Inc., which tried to add automation and precision to the field of animal testing.
In the background, however, he hosted and attended longevity technology salons and events. These activities were more of a hobby, a side passion. That is, until a series of scientific breakthroughs proved too strong a call.
“We want to build the best way to test as many transcription factor combinations as possible and also to predict which ones are most worth going after”
In 2006 the Japanese surgeon-turned-researcher Shinya Yamanaka made a dazzling discovery. He took skin cells from an older mouse and turned them into stem cells (specifically, what are known as pluripotent stem cells).
In other words, he transformed a group of cells that the body had configured to perform specific functions and rewired them back to their original, blank state.
Researchers had long theorized that such a feat might be possible but assumed the techniques needed to perform the reversal would be complicated and require years, perhaps decades, of experimentation.
Yamanaka and his team stumbled upon a combination of four transcription factors, proteins that ferry instructions to DNA, that talked the older cells into activating genes that are usually turned on and buzzing only in the earliest moments of life.
Yamanaka received the Nobel Prize six years after publishing his paper on the science, and so-called Yamanaka factors are now famous throughout the biotech world.
We all start as blank canvases. Embryos initially have undefined stem cells that, as they divide and develop into fetuses, take on specialized roles as liver cells, neurons, heart muscle tissue and so on.
It’s an amazing process in that all the stem cells share the same DNA, but over time they start to emphasize different parts of their genetic code and form unique identities.
Observing how these transformations take place has been of major interest to scientists trying to discover the root cause of diseases and degeneration.
Before Yamanaka’s breakthrough, stem cells had to be harvested from embryos, and this process carried with it ethical controversies and biological limitations. Post-Yamanaka, researchers can more or less create stem cells at will.
Yamanaka’s finding was exciting enough that it paved the way for a new era in stem cell research. But it was work led by scientist Alex Ocampo and his colleagues then at the Salk Institute that redirected some of the attention around Yamanaka factors toward the longevity field.
Ocampo bred mice with the factors inserted into their genes and developed a means for turning the reprogramming mechanisms on for a couple of days at a time.
When this process was done just right, the mice showed broad rejuvenation among their organs and ultimately lived 30% longer than unmodified peers. A paper based on this research appeared in 2016 and soon led to a slew of startups trying to further the science and turn it into the basis of longevity therapies.
Retro is one of a handful of startups that have based much of their research and business prospects around these advances in cell reprogramming.
NewLimit Inc., a company co-founded by Coinbase Global Inc. CEO Brian Armstrong, has set out to do a massive survey of thousands of transcription factors in search of the best combination of proteins for stunting or reversing aging.
This project requires the company to compare and contrast blood cells gathered from young and old donors and to run a constant stream of DNA-sequencing tests and computational analysis as the cells are altered by various transcription factor recipes.
“We want to build the best way to test as many transcription factor combinations as possible and also to predict which ones are most worth going after,” says Jacob Kimmel, a co-founder and head of research at NewLimit.
Investors—including the venture capital firms Dimension, Founders Fund and Kleiner Perkins and individuals such as former Google CEO Eric Schmidt, tech investor Elad Gil and Y Combinator CEO Garry Tan—have agreed to put $40 million into NewLimit, and the company’s founders have pledged $110 million more.
That’s a significant amount of funding, but it’s well behind the $3 billion that Jeff Bezos, investor Yuri Milner and others are reported to have poured into Altos Labs Inc., another Silicon Valley cell-reprogramming startup.
Altos has recruited some of the top scientists from academic labs around the world, trying to turbocharge their research by giving them more funding and resources. The company declined to comment.
While the particular goals of these businesses vary, their overall mission is shared. They’re not looking to revert skin or liver or brain cells all the way back to their blank, stem cell state.
Rather, they’re trying to find ways to regress damaged or decaying cells to a healthier, younger condition. Many promising experiments have been done with animals and tissue samples where exactly this process has taken place.
Sometimes, however, the experiments run amok. Mice, for example, sometimes have their cells reprogrammed and then start growing tumors known as teratomas all over their bodies.
This, of course, adds major caution to any human experimentation. “There’s thinking that you can inject a virus that will go into someone’s tissues, put the genes for these transcription factors into the cells and express them for a while in hopefully a very controlled way,” Betts-LaCroix says.
“You have to stop before you go too far and make a pluripotent stem cell, because it will start trying to grow an entire person right there and form the tumors.”
Ocampo now has a lab in Switzerland that focuses on partial cell reprogramming. He remains encouraged by the technology, though with some caveats when it comes to the idea of developing safe longevity therapies for humans.
The genes linked to partial cell reprogramming are tied to higher incidences of cancer when they’re expressed at high levels.
In addition, the means of delivering potential therapies to a wide range of cell types remain limited. “You can reach places like the liver easily, but the brain and heart are more difficult,” Ocampo says.
It could take many years for these startups to create safe, approved treatments. Ocampo doubts a therapy delivered straight into the body based on the Yamanaka factors will arrive this decade.
“However, I think that other techniques and treatments using alternative factors or small molecules could be possible,” he says. “Overall, reprogramming is the technology with the strongest potential, and I don’t think anything else in the longevity field comes close.”
Retro’s headquarters are split into three main areas. There are offices that have been decorated true to Betts-LaCroix’s counterculture vibes. One small room, for instance, has jellyfish wallpaper on one wall and some sort of cotton-ball material on another that’s meant to evoke a cloud.
The bathrooms have LED lights that project star patterns and different colors.
A second, mid-building section is full of lab and DNA-sequencing equipment, much of which Betts-LaCroix got on the cheap from Curative, a onetime Covid-19 testing highflier that’s morphed into a health insurance company.
Most of the square footage is taken up by the mouse and tissue testing facilities. In its earliest days, Retro had two vivarium shipping containers full of mice and one container for conducting cleanroom-style tests.
Now it has more than a dozen containers and adds new ones as the operation expands. All the containers are interconnected via lab-grade HVAC and air-filtration systems that Betts‑LaCroix and his team built from scratch.
Many of the thousands of mice are immunocompromised as a result of the experiments taking place within their cells. So researchers must don protective gear and go through an air lock before entering the vivarium.
“One germ can ruin the mice’s whole day,” Betts-LaCroix says. The mice live in shoebox-size plastic containers full of little blocks of wood they can chew on and other material they can use to make nests.
Retro has tried to be very precise with lighting and temperature for the mice by placing sensors and control systems on each container instead of treating the conditions of the room as a whole, as is typical at other research centers.
“Nobody tracks that some of the mice in their experiment were near the top of a rack of cages versus near the bottom, where they might get less light,” Betts-LaCroix says. “And then they wonder why they can’t replicate the results of their experiment. It’s a huge problem. One of my life agendas is being on this warpath to improve animal experiments. These mice are making a sacrifice, and we should make it count.”
Retro has a variety of cell-reprogramming experiments going, including studies in mice and some with human tissue gathered from patients suffering from cancer. On the cancer front, one team has been working to reprogram T cells to better attack solid tumors.
It’s common for T cells to exhaust themselves and shut down as they try to combat the rapid growth of cancer cells. Although it’s early days, Retro says it may have found a way to flip some switches in the T cells to keep them fighting.
It’s been running experiments on tumor samples removed from sick individuals and pounding them with its reformatted T cells. Another project centers on reprogramming liver cells to return them to a younger state.
Retro’s most promising reprogramming effort may be the one run by Anastasia Shindyapina, a staff scientist at the company. She heads a program to rewire humans’ immune systems.
Shindyapina hails from Russia, which has a long-held cultural fascination with longevity technology, and met Betts-LaCroix at a Silicon Valley biotech gathering where he persuaded her to abandon academic work in favor of chasing Retro’s varied, high-risk bets.
“It was in the early, early days of Retro, and no one knew if this would turn out good or bad,” she says.
A rejuvenated immune system could have some of the broadest possible longevity effects, as numerous types of cells throughout the body would be able to block and hunt down disease better and repair the body more quickly, the way we do in the prime of our youth.
Over the past year, Shindyapina says, her team has produced ever more encouraging results. “When your immune system is screwed up, it affects every tissue,” she says. “You have to target the immune system first, and it will give rise to everything else.”
The human body has more than 1,500 transcription factors, which can be combined in an astronomical number of ways. Picking which factors to focus on requires machine-learning algorithms that can parse the data and hunt for patterns.
The labs at Retro, NewLimit and elsewhere are full of DNA analysis machines made by 10x Genomics Inc.
The company has pioneered gene-sequencing techniques that let scientists study changes taking place within individual cells. “It allows us to understand biology with far greater resolution,” says Buckley, one of the Retro co-founders.
“If you can rejuvenate a single cell, maybe you can rejuvenate some tissue and then maybe an organ and then maybe the entire organism.”
Buckley and Alex Trapp, a staff scientist at Retro, have been at the forefront of this computational biology revolution for years. Their work stretches across not only the cell reprogramming but also areas such as autophagy and blood plasma research.
In the autophagy arena, Retro has been trying to develop an injection or pill that could mimic the benefits of caloric restriction, which leads the body to cleanse itself of old, damaged cells.
And with the blood plasma work, Retro is playing off the years of research that show old animals benefit from being infused with the plasma of younger animals.
Although the studies here have been encouraging, the actual means of doing the transfusion is off-putting: In addition to being a time-consuming process resembling dialysis, it basically involves the old feeding off the young.
Here again, Retro looks to produce a therapy in a pill or injection that would trigger the effects of the plasma exchange without having to actually perform a physical swap.
At every turn, Retro’s future hinges on bets, crossed fingers and faith that this is the moment when biology and computers really will unlock the secrets of how the body works.
For encouragement, Trapp points to that miracle of longevity, the naked mole-rat. “They live for 40 years and never get cancer and never have any age-related pathologies,” he says.
“We can look at how these other species have evolved over millions of years to do particular things and harvest those insights.” He also points to the simple idea that human bodies, without question, know how to rejuvenate.
At conception, embryos combine and reset old and damaged genes into a healthier, cleaner state. “If this didn’t happen, people would get continuously older and carry on the damage from their cells,” Trapp says. “Obviously the body and evolution have figured out how to do this. Now we need to figure out how to do it artificially ourselves.”
The amount of money that Retro, NewLimit and Altos Labs have raised is both staggering and not. These companies are pursuing technology that could reshape human life.
In a world where Hollywood will place equal-size financial bets on several movies in the coming year, it’s actually almost shocking that more companies haven’t opted to chase what could be the best business of all time.
The hard truth, of course, is that many, many promising biotech companies have gone bankrupt on their journey to prove the merits and safety of their products. Five research areas mean at least five clinical trials, each requiring a lot of time and money.
“Sam will not be able to fund this all the way,” says Ding, the Retro co-founder and famed researcher in the cell-reprogramming field. “Getting to the final products will require more investors and going public at some point.
I’ve seen plenty of very fancy companies crash and burn along the way. The magical story can only be told for a limited number of years, frankly, before you see results.”
At the same time, Ding maintains that Retro’s funding and nimble operations give the company as good a chance as any to succeed, and he’s as confident as ever that a solution to aging is right at hand.
“Often in scientific discovery, you can go for years or even centuries looking for the answer to problems,” Ding says. “Here we know the answer.”
Silicon Valley’s Quest To Live Forever Has Many Warring Factions
Tech titans, venture capitalists, crypto enthusiasts and AI researchers have turned longevity research into something between the hottest science and a tragic comedy.
When a chipper person with a clipboard approaches you on the street in San Francisco, you can usually expect the inquiry to be about one of a few public-policy goals. Do you have time to talk about abortion rights? Housing? Online gambling ballot initiatives?
These days, though, you might also hear a question that’s both more grandiose and more personal, the kind of thing you wouldn’t expect to hear from someone who looks like they want your signature. The question: How long do you want to live?
One Saturday in August, Anastasia Egorova, the 37-year‑old chief executive officer of a longevity research nonprofit, organized two dozen volunteers in San Francisco and 10 other cities to collectively get answers from almost 200 passersby.
Many respondents pegged their desired life span at somewhere from 80 years (roughly the US average) to 120 years (close to the global record).
Egorova’s clipboard-toting volunteers in São Paulo and Toronto heard much the same that day, as did volunteers in Haifa, Israel, and Malmö, Sweden.
Few people rose to the challenge posed by the merch many of the clipboarders were wearing. All of the hats, T-shirts and stickers read, “Say Forever!”
Egorova and her fellow anti-death activists, a bunch of scientists and longevity enthusiasts who call themselves “Immortalists,” argue that a good and rational person should want to live as long as possible.
While they recognize the existence of the chronic pain and unspeakable traumas that make for strong counterfactuals, they’ve made a personal commitment to try to live forever, and they believe defeating aging should be a top priority for both research dollars and social activism.
They carry around posters with mantras such as “Death is unacceptable,” “Death is boring” and “Stay alive,” all of which made appearances this fall outside the historic Ferry Building overlooking San Francisco Bay.
One particularly devoted volunteer took the idea of permanence to heart and got the words “SAY FOREVER” tattooed across the front of his neck. “Dying is bad,” Egorova says. “This is something humanity doesn’t take seriously enough.”
The Immortalists may sound more like the Merry Pranksters than a serious lobby, but they embody a strain of tech industry culture that’s taken hold in the occasional gaps between conversations about artificial intelligence and crypto.
Different circles in the Bay Area and other tech hubs are looking for methods to cheat death, from popping supplements and pipetting solutions in a lab to trying to digitize their consciousness or freeze their brain to be revived later.
(They hope.) The underlying belief is that technology could someday transcend our ultimate biological destiny. Or as influential investor and provocateur Balaji Srinivasan’s X bio puts it: “Immutable money, infinite frontier, eternal life.”
What distinguishes this obsession from a typical midlife crisis is the scale of the money behind it. Jeff Bezos, Sam Altman, Larry Ellison, Larry Page and other tech titans have pledged hundreds of millions of dollars toward companies pursuing longer life.
Partly as a result, immortality-obsessed people in the industry have begun to view radical life extension as a near-term option. Some are, by day, investors or researchers in AI.
Others are cryptocurrency winners with fiat money to burn. Ethereum co-founder Vitalik Buterin has emerged as a flag-bearer for longevity advances—he’s donated millions of dollars to research organizations and posts things like: “Aging is a humanitarian disaster that kills as many people as WW2 every two years.”
Robert Nelsen, a hedge fund manager who has a stake in longevity-focused biotech Altos Labs Inc., takes almost a dozen drugs a day, including rapamycin, which has been shown to increase life span in mice.
Venture capitalists are pivoting to focus on longevity companies, and tech connectors are launching longevity-focused fellowships to entice newcomers to the field. Most of these crowds are united by techno-optimism and a belief that they’re privy to a hidden truth.
Contrarianism is the norm, says Amol Sarva, one of the investors eager to fund advances in the field. He says the industry is a mix of “graybeard wizards” proclaiming mystical-sounding futures, more conventional biotech experts focused on science, the “Peter Pan types” who project youth and the “cyborg types” talking about a future in which human consciousness transcends the fleshy body.
Sarva, who used to run the WeWork clone Knotel Inc., has raised a $100 million fund with a partner to invest in longevity-related companies, with checks already written to those using AI to better treat cancer and stem cells to create biology-powered computers.
He compares the energy of the longevity scene to that of the Homebrew Computer Club, a disco-era gathering that fueled the birth of the personal computer and Apple Inc.
Although it can be easy to reduce the various longevity efforts to one big blob of existential dread powered by money and fragile male egos, the community includes cliques with very different aims. Some are focused on extending the average human life by a modest range.
Others are more conservative, promising only to add more “health span,” meaning healthier years within a natural time frame.
And some go fully eternal, promising—with typical Silicon Valley restraint—to build their own sovereign-ish state where they plan to master and defeat death.
“We’ve accepted aging and death as inevitable, and now there’s a very good reason to believe they will be solved,” says Adam Gries, an investor focused on longevity. “The only question is when.”
To keep track of the different factions, we’ll give them some nicknames. In one corner are the Biotechs, the most buttoned-up of longevity researchers.
They’re developing drugs that they plan to submit for formal approval by the US Food and Drug Administration and that they aim to have available behind the pharmacy counter within the next decade.
Some of these medications are meant to promote autophagy—a cell’s ability to recycle accumulating molecular junk—or clearing out senescent cells, which have aged faster than other cells and can no longer replicate.
It’s early enough yet for these drugs that one of the most promising treatments targets dogs rather than people. “We started with big dogs because a Great Dane has an average life span of 6 to 9 years, and it’s starting to go gray even at age 3 or 4, so you can see it much faster,” says Celine Halioua, the CEO of Loyal.
Those accelerated dog years make it much more practical to run clinical trials on aging, as opposed to a 50-year trial for human longevity. Halioua’s idea is to give dogs more healthy years by reducing the release of growth hormones, which are correlated with faster aging in larger breeds.
She’s sensitive to scaremongering about zombie dogs and stresses that her goal is “healthy life span extension” for a cherished pet.
“We talk about Fluffy being able to chase the ball longer, being excited when you’re home and not being stiff when she gets out of bed,” Halioua says. Dogs are a “nonthreatening” way to introduce the public to the idea that the biology of aging is malleable, she says.
A 15-year-old dog doesn’t evoke all the fears of playing God that a 150-year-old human does.
Then there are the Wellness Obsessives. These are people fed up with the medical establishment’s focus on acute care instead of preventive care, and they’re keen to try supposedly proactive therapies that are often ignored or dismissed as snake oil.
(Think Braintree Payment Solutions LLC founder and unofficial Bloomberg Businessweek mascot Bryan Johnson, who’s spending millions of dollars a year to make his middle-aged body work more like a teenager’s.) While some of their experiments are pretty far out, the WOs are still considered relatively conservative in longevity circles.
They’re focused on zealously evaluating their bodily functions so they can make incremental improvements. Like the Biotechs, they tend to say some version of “health span” a lot.
“You are more in control of your health than you think,” says Martin Tobias, a venture capitalist and the former CEO of Upgrade Labs Inc., a chain of facilities specializing in gadgets such as an oxygen trainer and an AI-enhanced stationary bike.
“If you’re the CEO of your own health, you can get outcomes that are not usually delivered by the traditional medical system.”
Tobias, who’s about to turn 60, says he wants to make sure he’s around when his youngest daughter, now 8, is graduating from college. To get there, he’s trying some stuff that wouldn’t seem out of place at a Goop retreat.
His Seattle garage is packed with $500,000 in equipment, including two saunas, an infrared light bed, an electromuscular stimulation suit and a cryotherapy chamber.
He takes cold plunges, flies to Central America to get injections of stem cells and undergoes treatments to lengthen his telomeres, chromosomal proteins that shorten with age.
Tobias and others like him believe that in the pursuit of slowing their own clock, it’s worth burning money on a dozen experimental treatments even if most don’t work. Big names in this world include physician Peter Attia, geneticist David Sinclair and futurist Peter Diamandis.
It’s tough to know whether any of it’s working, given that the biomarkers thought to measure a person’s “biological age” are still poorly understood.
Still, Tobias is doing the work and analyzing the results, and he says he’s seeing improvements that are bringing him some peace, along with a sense of control. “My telomeres are measuring 38 years old,” he says.
Both the Biotechs and the Wellness Obsessives are at pains to emphasize that they still expect to die. For the Radicals, that’s loser talk. Patrick Linden, a philosopher who wrote a book called The Case Against Death, calls the acceptance of mortality a “fallacy” meant to “protect yourself from the tragedy of wanting something you can’t have.”
The Radicals’ cause began to reach the mainstream in the mid-2000s. In 2005, Nick Bostrom, an influential AI theorist, wrote a fable in which death takes the form of a tyrannical dragon that terrorizes a kingdom, demanding a constant stream of sacrifices, until a brave king spends vast sums over many years to develop a missile that kills it. (A video version of the fable, posted on YouTube far more recently, has about 10 million views.)
Around that time, British researcher Aubrey de Grey, whose foot-long beard gave him a bit of a Merlin vibe himself, became the public face of a fledgling longevity movement.
De Grey said in a BBC interview that the first person who’d live to 1,000 was probably already alive, then delivered a TED Talk about why humanity should devote resources to fighting aging.
He argued that once we’re able to repair aging damage at a faster rate than we age, we’ll reach “longevity escape velocity,” where age-related mortality keeps receding farther and farther away.
This possibility of living much longer—or forever—has inspired bioethicists and philosophers to ponder the prospect’s unexpected consequences. It’s an isolating field of study.
“If I go to the hairdresser and I tell them what I’m working on, the immediate reaction is, ‘That sounds horrible,’ ” says Raiany Romanni, a 29-year-old bioethicist exploring the stories we tell ourselves about death and meaning.
She says she sees acceptance of death not as a natural part of the grieving process but as naiveté. “I spend my professional life thinking that death has no purpose,” Romanni says. “That can be really harmful on a personal level. But if we keep telling ourselves comforting stories, it could be infinitely more harmful.”
In the meantime, some of the most hardcore Radicals are backing the old libertarian play of colonization—trying to move to one place en masse and make their ideology a matter of policy.
They gave it a dry run for two months this summer at Zuzalu, a Burning Man-esque temporary city in Montenegro. There, on the edge of the Mediterranean, Ethereum’s Buterin gathered hundreds of longevity true believers, crypto founders and AI researchers to discuss the practical realities of living forever.
What political structures would you need? How could you build regulation-light “innovation zones” to pursue experimental treatments?
If everyone started living dramatically longer, how quickly would populations grow or (maybe) shrink? Between cold plunges and sauna sweats, two venture capitalists unveiled their plans for a movement called Vitalism, which they said would coalesce around the belief that “death is humanity’s core problem” and that the species should do what it takes to “reach freedom from aging as soon as possible.”
Depending on your views, the rhetoric is either inspiring or ridiculous. But in Silicon Valley, where wealth and power are concentrated in the hands of a small crowd of billionaires, winning over a few people can sometimes be enough.
More traditional researchers are very aware that two-month bacchanals and multimillion-dollar treatment regimens can make their field sound like a distraction for the rich.
Loyal CEO Halioua, who researched the economics of gene therapy at Oxford University, says the broader industry is “sketchy,” and people who overhype the benefits of unproven treatments or talk about immortality are causing a “massive reputational issue.” To her, people like Johnson and Buterin help relegate longevity science to the fringes.
Some of the field’s leaders have been discredited in other ways, too, she says. In 2021 she and investor Laura Deming both alleged that de Grey, the onetime face of longevity, had sexually harassed them.
Deming, who began working in a longevity research lab at 12, wrote that when she was 17 and 18, she received emails from de Grey, a trusted mentor, calling her “hotter than hell” and lamenting that because of her age they couldn’t discuss his “adventurous love life.”
Halioua alleged that at a fundraising dinner for de Grey’s foundation, which funded some of her research, he “funneled me alcohol and hit on me the entire night” and said that “as a ‘glorious woman’ I had a responsibility to have sex with the donors in attendance so they would give money to him.”
The foundation’s investigation into de Grey’s conduct upheld both women’s principal claims and also found that he’d interfered with the investigation. He was fired and has now started another longevity organization.
Halioua says it’s disappointing that de Grey is still appearing at some longevity conferences, but she feels satisfied that a quick Google search can spare others from what she and Deming experienced.
In an interview, de Grey dismisses the allegations as overreactions to anodyne comments. He calls his firing “that whole shit show” and says it was a cover for his board to wrest power from him.
De Grey’s reputation has taken a hit, but the “Say Forever!” volunteers were still thrilled when he showed up at a recent gathering of theirs in San Francisco.
It was the Saturday before Halloween, and a dozen Immortalists had costumed themselves as vampires and shamans, wearing bird masks and faux-bearskin capes and carrying plastic skulls and rain sticks.
They paraded along the city’s Embarcadero waterfront, dancing and miming along to a heavy-metal-esque song with lyrics by Gries, the longevity investor. “I’m begging, wake up!” the singer growled in the recording. “Save our loved ones from brutal decline / Give the finger to old Father Time.”
A few curious strangers approached, but the attendees mostly took photos and videos for themselves—and for the benefit of their shared Instagram account.
Say Forever! co-founder Mikhail Batin, wearing a feathered mask and a pentagram-emblazoned robe, said he was betting on displays like these to grab the attention of artists, who in turn could have more mainstream persuasive power than the average peer-reviewed paper.
In the meantime, the various longevity factions are making progress in their own ways. In November, the FDA approved part of Loyal’s application for a drug meant to lengthen big dogs’ lives by blocking a specific type of growth hormone once they’ve reached their full size.
The drug could be available for your Great Dane or mastiff in 2026.
Meanwhile, particularly well-capitalized Wellness Obsessives are scoping out longevity tourism, flying overseas to get experimental treatments. Diamandis is charging $70,000 for a five-day tour of various private longevity clinics.
And a group that includes some Zuzalu organizers is planning to create a pop-up city this winter on the Caribbean island of Roatán.
Some Radicals are also hoping a mega-funder such as Elon Musk will sponsor their dreams over the next decades. Musk, for his part, recently said he thinks extreme longevity would cause the “asphyxiation of society.”
Elsewhere, he said, “I can’t think of a worse curse than living forever.” Says Linden, the philosopher: “We’re all hoping for Elon Musk, obviously, to wake up.”
Updated: 11-29-2023
The Clues To Longer Life That Are Coming From Dogs
Scientists are studying dogs to learn more about human longevity.
In the quest to help people live longer, scientists and companies are turning to dogs.
Humans have greater genetic similarities to dogs than other common subjects of aging research, like mice. Our species get many of the same age-related diseases, including cancer and osteoarthritis. And dogs tend to live alongside us, sharing our exposures.
It also helps that they age faster than we do, giving a quicker timeline to study longevity.
Behind the growing enthusiasm is a mix of scientists and entrepreneurs—building on the surging interest from people aiming to live longer. These groups say insights into dog longevity could provide lessons and perhaps eventually treatments that could help people, too.
Among some notable findings, scientists at the Dog Aging Project, a research effort based at the University of Washington and Texas A&M, found that physical activity was associated with better markers of cognitive aging in dogs.
Another found social companionship, such as living with other dogs, was associated with better health.
Business developments aren’t far behind. On Tuesday, a biotech startup that’s hoping to have the first FDA-approved treatment to extend healthy lifespan in dogs, took a step toward that goal.
In a letter viewed by The Wall Street Journal, the Food and Drug Administration affirmed that its drug had demonstrated “reasonable expectation of effectiveness.”
The company, called Loyal, still has to complete several more steps before it can market the drug, and it’s only aimed at canines.
“It’s ideal to study aging in dogs, and then the leap to be able to use that to transfer into human application really is the next step,” says Louise Grubb, chief executive of veterinary drug company TriviumVet, which is working toward gaining FDA approval of a separate drug targeting age-related diseases in pet cats and dogs.
What The Science Says About Dogs And Humans
No animal model is a perfect stand-in for humans. But dog research may offer us some clues that other animal models cannot.
Researchers at the Dog Aging Project are currently enrolling canine participants in a double-blind, placebo-controlled trial to study the effects of rapamycin, a drug originally approved to prevent organ rejection in human transplant patients. Some people use it off-label as part of a longevity regimen.
If any treatment is shown to extend healthy lifespan in dogs, we can have more confidence that it will have a similar effect in people, says the project’s co-director Matt Kaeberlein, a molecular biologist and longevity scientist.
The complexity of our environments could affect how drugs or behavioral changes have been shown to work in, for example, mice in controlled lab settings, he says.
From breed to breed, dogs are very genetically diverse. Some breeds are more likely to develop certain diseases than others, says Daniel Promislow, principal investigator of the Dog Aging Project and professor at the University of Washington.
This can help scientists identify subjects for research on the genes or environmental factors that might contribute to those diseases.
There are limitations, too.
Differences between our species, including diets and the prevalence of sterilization in dogs, could affect the way we age and how we measure it.
And most research in pet dogs is designed to give us hints, not provide cause and effect, says Dr. Manuel Moro, a scientist at the National Institute on Aging who works on developing animal models.
Growing Interest In Dogs
Between 2019 and 2021, global financing for pet longevity firms more than quadrupled, according to an analysis from longevity research and media company Longevity.Technology.
Driving the interest, in part, is the notion that getting an aging drug on the market for animals is likely to be cheaper and faster than doing so for people.
TriviumVet, the veterinary drug company, is working toward FDA approval of the formulation of rapamycin being studied by the Dog Aging Project.
The company is studying the drug’s effects on age-related illnesses, like heart disease, in pet cats and dogs, but the work has also caught the eye of some researchers studying aging in humans, says Grubb.
Garri Zmudze, a founding partner at longevity and biotech-focused venture firm LongeVC, has raised $20 million for a separate early-stage fund that will invest in the pet space, including longevity and therapeutics.
“It will take a lot of time to translate [animal data] on humans but I believe that we can start early,” says Zmudze.
Celine Halioua, chief executive of Loyal, the biotech startup working toward conditional approval of its lifespan drug, says there is a larger aim in addition to helping dogs live healthier for longer.
The company has set a possible precedent for other drugs to be approved for lifespan extension, potentially opening a door for other animal—or human—drug companies to follow.
“I think we can both take the opportunity to build better medicines for our dogs and also to better understand these really complex diseases,” says Halioua, whose own 85-pound Rottweiler mix, Della, is nearing the end of her projected lifespan.
The firm’s drug is an injectable that is designed to reduce levels of IGF-1, a hormone that drives cell growth, in large dogs. High blood levels of IGF-1 have been associated with shorter lifespans in some animal and human studies.
The company’s research has indicated that the drug can reduce those hormone levels, but it would still need a large clinical trial demonstrating it can extend dog lifespans in order to achieve full FDA approval.
It also needs the agency’s signoff on the drug’s safety and proper manufacturing before getting conditional approval and beginning to sell it, which Loyal hopes to do in 2026.
Still, the FDA nod this week is a promising next step for the field, dog aging researchers say, and will likely drive more interest from biotech and pharmaceutical companies.
“If it is proven that the drug is effective in dogs then there is a higher chance that it will work in the case of humans, too,” says Eniko Kubinyi, a biologist studying dog behavior and cognition with the Budapest-based Family Dog Project.
Updated: 3-30-2024
Your Dog Will Have An Anti-Aging Drug Before You Do
Clinical trials on humans could take decades — long enough for the study subjects to live out the rest of their lives. That’s where man’s best friend comes in.
Before we have an anti-aging drug for humans, we’re likely to have one for dogs. Multiple clinical trials are currently underway to test potential anti-aging compounds on dogs, since our best friends have become a popular animal model for human aging. Fido also represents a potentially huge market.
The science could benefit both species, but premature claims are already causing a credibility problem.
Recently, Harvard University biologist David Sinclair started marketing life-extension supplements for dogs, touting unpublished clinical trial data that others in the field found completely unconvincing.
Earlier this month Sinclair, who didn’t respond to requests for an interview, changed the wording of a press release, which originally promised the chewy treats would “reverse aging.” Now it says they reverse the effects of age-related decline.
Sinclair’s trial used dog owners’ subjective assessments of cognitive changes in their older pets — and other scientists say the trials don’t show a consistent enough effect even for this more modest claim.
(Sinclair became famous in the 1990s for some highly publicized papers tying aging to proteins called sirtuins — an idea that led to the now widely disputed belief that red wine has anti-aging properties.)
While the FDA is authorized to regulate veterinary drugs, it doesn’t approve supplements for pets or people, so these can be sold without going through tests for safety and efficacy.
Whether anti-aging supplements for dogs work or not, there’s likely to be demand, said Arthur Caplan, a professor of ethics at New York University. In the past, desperate dog owners have had their dead or dying dogs cloned — hoping the clone would be essentially a reincarnation of their dead pet.
Some researchers who study aging fear that the spectacle of a high-level professor hawking dog longevity supplements will further tarnish the reputation of a field already dragged down by self-proclaimed experts pushing fad diets and unproven anti-aging treatments for people.
There’s a lot to gain from a better scientific understanding of aging. Getting older is a risk factor for all the major killer diseases — heart disease, cancer and even severe Covid.
And in the US, the ranks of people over 70 will swell within the coming years, creating a vast increase in the number of people suffering from dementia or other age-related problems.
But scientists don’t yet agree on what causes aging or what approach would work best to slow it down. While wear and tear will eventually affect all living things, some organisms live many times longer than others, even among closely related species. Some researchers think an animal’s aging rate is controlled by certain genes.
Other experts cite the shrinking of the caps on the end of or chromosomes, called telomers.
Others blame the degeneration of the packaging around our DNA — so-called epigenetic markers, which can activate or suppress certain genes. Some scholars blame damage caused by chronic inflammation. Still others, the buildup of cellular waste products.
Some of these possible mechanisms of aging can be altered with drugs in a way that endows worms, fruit flies and mice with longer lives. Which drugs should be tried in humans?
Clinical trials to test their effects on longevity in people could take decades — long enough for the study subjects to live out the rest of their lives.
One way to identify the more promising candidates would be to see which ones also work in dogs. Dogs are long-lived enough to serve as a better model for human aging than mice, but short-lived enough that treatment can be tested in a few years.
Matt Kaeberlein, CEO of Optispan and an affiliate professor at the University of Washington, was among the most vocal critics of Sinclair’s dog longevity claims. He’s also in competition as co-director of The Dog Aging Project.
That project involves collecting data on thousands of dogs as well as conducting a dog clinical trial with a drug called Rapamycin. It’s currently approved for people who’ve had organ transplants.
At high doses, it causes mouth sores and other nasty side effects, he admits, but can extend the lives of mice and — at low doses — might do the same in dogs or humans.
A group of biohacker types is already taking Rapamycin off-label in the hope of life-extension, he said. He’s trying to get data from them, messy as it is, because there might be useable information there.
(Caplan, the NYU ethicist, says he thinks it’s unethical for doctors to prescribe this drug off-label for longevity.)
Kaeberlein said the biological data they’re collecting from all those thousands of dogs could lead to an explanation for the fact that big dogs don’t live as long as small ones.
“If you compare a Great Dane to a chihuahua on average, it’s at least a twofold difference in life expectancy,” he said.
But his project might be a victim of the field’s wider credibility problem. It had been funded by the National Institutes of Health, but he and his colleagues learned recently that a five-year grant established in 2018 and extended one year probably won’t be renewed.
He’s now working to get private money.
Charles Brenner, a biochemist at the City of Hope National Medical Center in Los Angeles, is another vocal critic of Sinclair’s claims regarding dogs and humans.
(Like most researchers on the forefront of aging, he has his own supplement ties as chief scientific advisor of a bioscience company called ChromaDex.)
Brenner is also skeptical of those who claim various treatments or drugs can reverse a person’s “biological age” as calculated through proxies measured in blood — including telomeres and epigenetic markers.
None of these measure aging as well as walking speed, he said.
When I asked him about the Rapamycin study in dogs, he said it’s “worth a try” because the trial measures actual lifespan rather than some proxy. But he isn’t betting on this particular drug.
He’s more optimistic about work done by a company called Loyal, profiled in 2021 by Bloomberg Businessweek.
Brenner says while Loyal has been secretive about the drug, he thinks what they’re now testing inhibits the production or action of growth hormone. Growth hormone, he said, is connected to the faster aging they see in larger dogs compared to smaller ones.
There’s promise in studying how and why animals age — not just dogs but clams that can live to 500, rockfish that survive until 200 and whales that reach 80.
Once scientists understand the mechanisms of aging, they’ll be much better able to find ways to help us — and our furry friends — live longer and healthier lives.
But first, they need investors and the public to take them seriously.
Updated: 4-15-2024
The Longevity Vacation: Poolside Lounging With An IV Drip
The latest trend in wellness travel is somewhere between a spa trip and a doctor’s appointment.
For some vacationers, the ideal getaway involves $1,200 ozone therapy or an $1,800 early-detection cancer test.
Call it the longevity vacation. People who are fixated on optimizing their personal health are pursuing travel activities that they hope will help them stay healthier for longer.
It is part of a broader interest in longevity that often extends beyond traditional medicine. These costly trips and treatments are rising in popularity as money pours into the global wellness travel market.
At high-end resorts, guests can now find biological age testing, poolside vitamin IV drips, and stem-cell therapy. Prices can range from hundreds of dollars for shots and drips to tens of thousands for more invasive procedures, which go well beyond standard wellness offerings like yoga, massages or facials.
Some longevity-inspired trips focus on treatments, while others focus more on social and lifestyle changes. This includes programs that promise to teach travelers the secrets of centenarians.
Mark Blaskovich, 66 years old, spent $4,500 on a five-night trip last year centered on lessons from the world’s “Blue Zones,” places including Sardinia, Italy, and Okinawa, Japan, where a high number of people live for at least 100 years. Blaskovich says he wanted to get on a healthier path as he started to feel the effects of aging.
He chose a retreat at Modern Elder Academy in Mexico, where he attended workshops detailing the power of supportive relationships, embracing a plant-based diet and incorporating natural movement into his daily life.
“I’ve been interested in longevity and trying to figure out how to live longer and live healthier,” says Blaskovich.
Vitamins And Ozone
When Christy Menzies noticed nurses behind a curtained-off area at the Four Seasons Resort Maui in Hawaii on a family vacation in 2022, she assumed it might be Covid-19 testing. They were actually injecting guests with vitamin B12.
Menzies, 40, who runs a travel agency, escaped to the longevity clinic between trips to the beach, pool and kids’ club, where she reclined in a leather chair, and received a 30-minute vitamin IV infusion.
“You’re making investments in your wellness, your health, your body,” says Menzies, who adds that she felt more energized afterward.
The resort has been expanding its offerings since opening a longevity center in 2021. A multiday treatment package including ozone therapy, stem-cell therapy and a “fountain of youth” infusion, costs $44,000.
Roughly half a dozen guests have shelled out for that package since it made its debut last year, according to Pat Makozak, the resort’s senior spa director. Guests can also opt for an early-detection cancer blood test for $1,800.
The ozone therapy, which involves withdrawing blood, dissolving ozone gas into it, and reintroducing it into the body through an IV, is particularly popular, says Makozak. The procedure is typically administered by a registered nurse, takes upward of an hour and costs $1,200.
Longevity vacationers are helping to fuel the global wellness tourism market, which is expected to surpass $1 trillion in 2024, up from $439 billion in 2012, according to the nonprofit Global Wellness Institute.
About 13% of U.S. travelers took part in spa or wellness activities while traveling in the past 12 months, according to a 2023 survey from market-research group Phocuswright.
Canyon Ranch, which has multiple wellness resorts across the country, earlier this year introduced a five-night “Longevity Life” program, starting at $6,750, that includes health-span coaching, bone-density scans and longevity-focused sessions on spirituality and nutrition.
The idea is that people will return for an evaluation regularly to monitor progress, says Mark Kovacs, the vice president of health and performance.
What Doctors Say
Doctors preach caution, noting many of these treatments are unlikely to have been approved by the Food and Drug Administration, producing a placebo effect at best and carrying the potential for harm at worst.
Procedures that involve puncturing the skin, such as ozone therapy or an IV drip, risk possible infection, contamination and drug interactions.
“Right now there isn’t a single proven treatment that would prolong the life of someone who’s already healthy,” says Dr. Mark Loafman, a family-medicine doctor in Chicago. “If it sounds too good to be true, it probably is.”
Some studies on certain noninvasive wellness treatments, like saunas or cold plunges, do suggest they may help people feel less stressed, or provide some temporary pain relief or sleep improvement.
Linda True, a policy analyst in San Francisco, spent a day at RAKxa, a wellness retreat on a visit to family in Thailand in February.
True, 46, declined the more medical-sounding offerings, like an IV drip, and opted for a traditional style of Thai massage that involved fire and is touted as a “detoxification therapy.”
“People want to spend money on things that they feel might be doing good,” says Dr. Tamsin Lewis, medical adviser at RoseBar Longevity at Six Senses Ibiza, a longevity club that opened last year, whose menu includes offerings such as cryotherapy, infrared sauna and a “Longevity Boost” IV.
RoseBar says there is good evidence that reducing stress contributes to longevity, and Lewis says she doesn’t offer false promises about treatments’ efficacy. Kovacs says Canyon Ranch uses the latest science and personal data to help make evidence-based recommendations.
Jaclyn Sienna India owns a membership-based, ultraluxury travel company that serves people whose net worth exceeds $100 million, many of whom give priority to longevity, she says. She has planned trips for clients to Blue Zones, where there are a large number of centenarians.
On one in February, her company arranged a $250,000 weeklong stay for a family of three to Okinawa that included daily meditation, therapeutic massages and cooking classes, she says.
India says keeping up with a longevity-focused lifestyle requires more than one treatment and is cost-prohibitive for most people.
Doctors say travelers may be more likely to glean health benefits from focusing on a common vacation goal: just relaxing.
Dr. Karen Studer, a physician and assistant professor of preventive medicine at Loma Linda University Health says lowering your stress levels is linked to myriad short- and long-term health benefits.
“It may be what you’re getting from these expensive treatments is just a natural effect of going on vacation, decreasing stress, eating better and exercising more.”
Updated: 3-12-2025
Bitcoiners Are Obsessed With Longevity And Biohacking: Here’s Why
The truth about the links between the crypto and longevity communities runs deeper and weirder than you might expect.
Brian Armstrong allocates 2% of Coinbase shares to longevity.
Back in 2022, Coinbase CEO Brian Armstrong announced plans to spend 2% of his stake in the listed exchange to fund longevity research.
He co-founded New Limit the following year, which aims to radically extend lifespan.
“Longevity feels like it’s tractable,” he said in January this year. “Someone could really make a breakthrough here, or many people, in the next ten years. It’s not like ‘oh we want to do time travel,’ pie in the sky, too far in the future.
This is something hard but tractable. And that’s in the sweet spot of something that’s worth working on. You want to be early but right.”
He also made a sizable donation to the Dog Aging Project, which is running clinical trials to see if Rapamycin can slow aging in dogs.
Co-director Matt Kaeberlein says he spoke with Armstrong many times in the lead-up to the donation.
“Brian is obviously a brilliant guy but also very thoughtful,” he says. New Limit is “focused on epigenetic reprogramming … so I know it’s something that he’s deeply interested in and has put a significant portion of his own resources towards.”
Kaeberlein is a highly respected longevity researcher who has published more than 200 scientific papers. He’s also the founder of health optimization tech company Optispan and the Healthy Aging and Longevity Research Institute at the University of Washington.
Over the past five years, he says he’s noticed more funds flowing into longevity research from the crypto community but notes tech billionaire Jeff Bezos also invested in regenerative medicine company Altos in 2021, and Google spent $1.5 billion setting up the California Life Company (Calico).
“I think there is a natural sort of connection between the tech community, crypto community and the longevity community,” he says, describing them all as “big thinkers, people who tend to think outside the box and maybe even rebel a little bit against traditional approaches.”
Leibowitz suggested that some tech people see aging as just another engineering problem that can be fixed. But while Kaeberlein says aging is probably “the unsolved problem” in human health — it’s a lot trickier than many expect.
“You do see people with an engineering mindset come into aging biology with that sort of perspective, and then they get into the science, and they realize it’s way more complicated than it seems like from the outside, and it’s tough to engineer your way around aging,” he says.
But who knows, maybe if we learn enough, we can get there.”
Bitcoin To The Rescue As Longevity Research Is Poorly Funded
Dr. Max Unfried, research fellow at the National University of Singapore’s Center for Healthy Longevity, tells Magazine Big Tech and crypto donations give the misleading impression that longevity research is well-funded.
But, in fact, most funding is allocated toward curing diseases rather than attempting to extend lifespan.
For example, in the US, the National Cancer Institute’s budget is around 20 times higher than the funding directed to Aging Biology research at the National Institute of Aging.
“There’s a perspective of aging (research) by a lot of people, (that) a lot of money should be there,” he says, adding that’s far from the reality. “And I think that’s why crypto comes in, where you actually can get some money from crypto for research that would not get money by traditional routes.”
Numerous DAOs have stepped in to help fund experimental research, often with the promise of tokenizing the resulting intellectual property.
Unfried is a former member of VitaDAO, which has 10,000 members and helps fund longevity research and tokenize treatments.
Bitcoiner’s Hacker Mindset Leads To Longevity Biohacking
Co-lead of the Web3 pod at VitaDAO, Alex Miloski believes the hacker mindset can lead some crypto people to become interested in accelerating biotech.
“The longevity space is right in the middle of that because it is actually trying to almost hack biology right? Because you are not supposed to not age. So, I think that hacker mindset might play a role,” he says.
“It’s an area that has been underfunded or under-appreciated, and it’s essentially trying to disrupt the entire healthcare system as well. If we solve aging, then the entire healthcare system will look a lot different.
So, I think there is some intersection between what crypto is trying to do for finance and what [we’re] trying to do for healthcare.”
One DAO that takes the longevity hacking approach literally is BiohackerDAO. It was founded early last year after a group of biohackers met at the Vitalia longevity pop-up city in Honduras.
Founding steward Mgoes says the community ranges in age from early 20s to late 60s, with many working in crypto, tech or health startups.
They’re “enthusiasts about Biohacking, cell enhancement and just kind of like spending time and money and resources in their free time to self-experiment and measure their own biomarkers — kind of like Bryan Johnson is doing, but at a less [high] spending scale.”
In November, the DAO raised $1.3 million on Juicebox, with members discussing and voting on what experiments and projects they wished to support.
Research on the site so far has looked at whether intranasal insulin can improve cognitive ability and how EMF radiation impacts sleep.
In December, the DAO launched Intra, which it calls the “world’s first tokenized biomarker data marketplace” to trade fitness tracker data.
Biohacking Your Way To Longevity Or Immortality
Mgoes says he’s thought a lot about the reasons for the crossover between crypto and longevity communities.
“Bitcoiners are very tech savvy, and longevity or biohacking is basically just technology applied to biology, to the human body.
It’s probably one of the biggest untapped, or undertapped, frontiers of technology that we have,” he says.
The biohacker believes crypto’s libertarian ethos extends to “self-autonomy over your own body so you can decide what substances you take” as well as rethinking problems from a fresh perspective.
“These people also have a mindset that often goes against established systems,” he says.
“Like, let’s forget about our conception of healthcare and also our conception of human limitations, such as our lifespans. And let’s just tackle this idea of mortality and decay of our bodies from first principles.”
Some of these ideas stray into the arena of “transhumanism,” which is a philosophical and intellectual movement that seeks to use tech to help humanity transcend its current biological limitations.
Mgoes says that “guiding, shaping, directing, even accelerating human evolution … Those are very key fields particularly in transhumanism, and that’s something that we, inside BiohackerDAO, vibe with very, very much.”
Updated: 7-11-2025
Step Aside Longevity Bros. It’s Time For The Longevity Ladies
A growing number of female scientists, entrepreneurs and influencers are edging into a space long dominated by men. Their study of hormones and ovaries could unlock the key to a longer life for everyone.
Jennifer Garrison, a 49-year-old neuroscientist, has been studying how aging changes the way the brain communicates with the rest of the body in a lab she runs in Novato, Calif.
But on an afternoon in March, she walked across a glitzy Los Angeles stage to the pulsing soundtrack of Kelly Clarkson singing, “What doesn’t kill you makes you stronger” and greeted an audience of mainly high net-worth men gathered to hear about tech and longevity.
“What if I told you that understanding female bodies could actually help every single person on the planet live healthier longer?” she asked.
Garrison then directed the audience to watch a movie of a 3-D structure on a large screen behind her. She asked everyone to guess what it was. A brain, a heart, a liver, a thyroid, a stomach, people called out. No, no, no, no and no.
“This is an ovary,” Garrison told them. She later added: “We call them the canary in the coal mine of aging.”
Jamie Justice, a gerontologist at Wake Forest University School of Medicine, says that an important predictor of longevity in both females and males is the age at which their mothers go through menopause, defined as a year without a menstrual cycle, which women experience on average at the age of 51.
If a mother goes through menopause late, she is more likely to live longer. So are her daughters, sons and brothers.
Scientists say that there are common genes involved, but they don’t yet know all of them.
“If we understand what is happening, we can unlock secrets about biological aging in women and in men,” said Justice.
Breaking Into A Boys Club
Women have long dominated the wellness industry, with a focus on keeping the skin dewy and the limbs slender and Pilates-toned. Women have also been major contributors to the science of aging.
The biologist Cynthia Kenyon helped launch the field with her 1993 discovery that a single gene mutation doubled the lifespan of worms.
Kenyon is now vice president of aging research at Calico Life Sciences, a company focused on the biology of aging.
Still, when it comes to longevity, particularly the high-profile, publicity-generating pursuit of lifespans extending to 100 and beyond, men have been the faces of the movement.
Software entrepreneur Bryan Johnson set up a Rejuvenation Olympics where people compete to see who has the slowest rate of aging, as measured by a blood test.
He was filmed receiving a transfusion of blood from his teenage son in the 2025 Netflix documentary about his quest for immortality, “Don’t Die: The Man Who Wants to Live Forever.”
Dr. Peter Attia recommends that people approach aging as a “Centenarian Decathlon,” training to enable abilities they want to keep past the age of 100, such as having sex or lifting a 20-pound suitcase into the overhead compartment of a plane.
His own list includes driving a race car close to the pace he does today and hiking with a 20-pound backpack for an hour.
David Sinclair, a Harvard geneticist and serial longevity entrepreneur, regularly appears at conferences and on podcasts claiming scientists will soon be able to reverse aging.
“So much of longevity has been this very individualistic focus on men,” said Dr. Sara Szal, a gynecologist, female hormone specialist and director of precision medicine at the Marcus Institute of Integrative Health in Villanova, Pa.
“The men got attention and funding. They made some progress. Women were left out of the longevity conversation.”
That is starting to change with the rise of an increasingly prominent group of female scientists, entrepreneurs and influencers. In a field dominated by “longevity bros,” they could be called the “longevity ladies.”
What these longevity ladies all address in varied ways is a mystery that continues to perplex the field. Women live longer than men, five years on average in the U.S.
The female longevity advantage is found in every country where mortality data are collected, cuts across socioeconomic groups and persists in hard times such as severe famines and epidemics, according to Dr. Dena Dubal, a neurologist at the University of California, San Francisco, who analyzed data from other researchers.
Yet women also spend more years of their lives than men do in poorer health—and not simply because they are living longer.
Women are at higher risk than men for hip fractures, Alzheimer’s disease, arthritis and a host of other problems common in old age.
Scientists aren’t yet sure why this health disparity exists. Some suggest it’s a consequence of the stress of caregiving, which falls disproportionately on women.
Others wonder if menopause is to blame, because when ovaries stop releasing essential hormones it can speed up biological aging in women.
The fact that so many of these questions remain unanswered may have something to do with the fact that women were historically excluded from most clinical trials at the National Institutes of Health, including trials on aging.
For example the NIH-sponsored Baltimore Longitudinal Study of Aging, which launched in 1958, didn’t enroll women until 20 years later.
Researchers typically worried about including women of childbearing age in trials owing to concerns that experiments could harm future children.
Because female hormones fluctuate throughout a menstrual cycle, scientists agreed it was simpler to design studies without taking them into account, even when the subjects were mice.
It wasn’t until 2016 that the NIH required researchers to use female mice, too.
Across medicine, “Researchers studied men, and doctors applied the results to women,” said Dr. Louise Aronson, a geriatrician and professor of medicine at the University of California, San Francisco. “It is better now for many things, but old habits die hard.”
Szal says that she tells female patients they can’t assume that what is popular with male longevity influencers will work for them. She noticed that low-carb diets caused menstrual irregularities and thyroid problems in some of her female patients.
She also isn’t sure if women benefit as much as men from regularly tracking VO2 max, a measure of how well the body uses oxygen during intense exercise.
“We don’t know how many of the statistics, interpretations and conclusions apply to women,” Szal said.
Kayla Barnes-Lentz, 34, a longevity influencer, began seeking Szal’s guidance after they both spoke at a female longevity conference in Los Angeles earlier this year.
Barnes-Lentz, who had her first date with her husband in a hyperbaric oxygen chamber at a longevity clinic she was running at the time, says Szal is helping her continue to adjust her nutrition, exercise and sleep depending on her menstrual cycle.
As for her husband, she said his longevity routines don’t change much because, “he has a 24-hour hormone cycle.”
‘Longevity Is About Community’
The rise of the longevity ladies can be attributed to the convergence of two major changes. One is a broader cultural embrace of the role lifestyle choices, such as social connections and a good diet, can play in both enriching a life and lengthening it.
These are areas where female voices have traditionally held sway.
Ella Davar, 37, a registered dietitian and nutritionist in Miami, launched in July 2024 the Global Longevity Association, a group that aims to promote healthy aging and a longevity lifestyle.
She first built a following by posting longevity foodie videos on YouTube and cooking healthy recipes on TV.
She self-published cookbooks of easy-to-make recipes full of antioxidants and probiotics to promote gut health and reduce inflammation, such as broccoli bone-broth soup, red cabbage salad with walnuts and blueberries and her personal favorite, sauerkraut.
Davar started hosting longevity lunches and dinners in the homes of friends last year, explaining the benefits of everything she served to guests seated around a large table.
As the popularity of these meals grew, so did their size. More recent events include a luncheon in April at Mar-a-Lago, where chefs served fish, olives and sauerkraut (President Trump was not there, she says).
At a luncheon at the White House Historical Association in May, where attendees snacked on crudités dipped in red-pepper hummus and avocado crostini, Davar urged guests to take turns sharing their own thoughts on longevity.
Many opened up with deeply personal stories about their own illnesses and aging relatives. Davar, who regularly tells people that no one should eat alone, told diners to start hosting their own longevity meals for friends and family. “Longevity is about community,” she said.
The other big driver in the rise of the longevity ladies is the emergence of scientific findings showing the role that sex differences play in the aging process—and a growing belief that research on women might improve how aging is understood and treated in men too.
Dubal, the neurologist at UCSF, says some clues about the female longevity advantage have come from studying the role of the X chromosome.
Males have only one X chromosome, but females have two, one from each parent, though one of them remains dormant for much of a female’s lifetime.
In studies of mice, Dubal and her colleagues found that the silent X “wakes up” and makes proteins as the female mice age. In one experiment, they genetically engineered the brains of old male mice to make more of a protein that the awakening X makes in females.
They found that learning and memory improved in the male mice.
She suspects that this late-in-life source of protein in females may contribute to longevity, though she still needs to prove it. “I am really optimistic that understanding the second X is going to be relevant for both males and females,” she said.
One of the challenges for developing antiaging therapies is that it takes a long time to assess whether something has worked or not in lengthening a human life.
This is why scientists are increasingly conducting trials on ovaries, which age 2½ times faster than other tissue in the body.
“Ovaries are a model of accelerated aging,” said Dr. Zev Williams, a fertility doctor and director of Columbia University Fertility Center.
Doctors at Columbia are testing low-dose rapamycin, a drug approved to prevent organ rejection in transplant patients, to see if it slows down ovarian aging in a trial of women ages 35 to 45.
Rapamycin has slowed down ovarian aging in female mice and extended the lifespans of mice of both sexes.
Williams, a co-investigator on the trial, says that if it works, rapamycin might help delay menopause and reduce heart disease, stroke and dementia risks, although larger studies are needed.
Jamie Justice, the gerontologist, was tapped two years ago to run XPrize Healthspan, a competition with a $101 million purse, for which teams of scientists are trying to develop interventions to reverse muscle, cognitive and immune losses by at least 10 years in people between 50 and 80 years old.
The teams have to run one-year trials with both male and female subjects. Winners will be announced in 2030.
Jennifer Garrison, who has been running a lab at the Buck Institute for Research on Aging, co-led a team last year that pitched the winning idea for a new XPrize competition.
The goal is for teams to develop a way to track changes in ovarian function throughout women’s lives, from pre-puberty to post-menopause. Garrison likes to call it “The Double X Prize.”
She explains that many women monitor their hormones when they want to get pregnant, but don’t realize ovaries are important to health at every age.
For women in their 20s, for example, evidence of irregularities could be a sign of polycystic ovary syndrome, a condition that puts them at higher risk for infertility and heart disease.
Garrison hopes the competition will lead to better ways to identify ovarian changes earlier to help head off related diseases.
Another main goal, she says, is to get people to stop thinking about ovaries as simply important for reproduction—or of concern only to women. “They are a proxy for what is driving aging in everyone,” Garrison said.
It has never been easy to raise money for companies or projects focused on female health and longevity, says Justice, but within 30 minutes of announcing the Double X idea, the foundation raised $1.2 million from people at the meeting for the prize.
“Many men there jumped on board and said this is the most important thing we can tackle,” Justice said. They plan to launch the competition in January with a purse of at least $50 million, large enough to attract serious competitors.
The prize will debut at time when funding for research in women’s health—already relatively modest—is under threat from further cuts.
“The research is vulnerable,” Garrison told the crowd at the March longevity and tech event.
“I think that we are at a critical turning point.”
Garrison wants to make it as clear as possible that researching female bodies promises to benefit everyone. “Males have always been the baseline for longevity,” Garrison said. “Females should be.”
Updated: 8-3-2025
Longevity Firms Push Montana To Become Hub For Biohacking, Experimental Treatments
‘Right To Try’ laws aim to boost access to unapproved therapies, but hurdles and safety concerns linger.
Montana is well known for luring visitors with outdoor activities such as fly fishing and hiking. Now, longevity companies are exploring investments in the state in a moonshot bid to make it a medical tourism hub.
State legislators have backed measures designed to ease patients’ access to therapies not approved by the Food and Drug Administration. The measures, known as “Right to Try” laws, exist in more than 40 states.
But Montana has pushed even further by adopting rules to make it easier for businesses to provide experimental drugs, therapies or devices and profit from selling them.
Health and Human Services Secretary Robert F. Kennedy Jr. has pledged to increase access to alternative medicine and recently shared publicly that he traveled to Antigua to receive a non-FDA-approved stem-cell therapy for his voice condition, known as spasmodic dysphonia.
“We’re going to end the war at FDA against alternative medicine,” Kennedy said in an interview released in May with self-described biohacker Gary Brecka.
“If you want to take an experimental drug, you ought to be able to do that.”
The laws have drawn the ire of some medical professionals, who fear that hopeful patients will try experimental drugs only to find them ineffective at best and harmful at worst.
They also fear the state is on shaky legal ground with federal regulators who have, in recent years, cracked down on businesses offering certain unapproved therapies.
“If we start hearing about harmed patients, it’s highly likely FDA gets involved,” said Lowell Schiller, former acting chief counsel for the FDA in 2018.
The Access Debate
President Trump in 2018 signed federal legislation that allows patients with life-threatening illnesses to request access to drugs that have shown basic safety in the first phase of clinical trials.
Many legal and medical professionals saw the law as largely symbolic. The FDA already approved a majority of such patient requests under its compassionate-use program, and drugmakers have the final say on who can access their in-development therapies.
Montana’s expanded “Right to Try” law in 2023 allowed any patient to access an experimental treatment, not just those with a terminal illness.
In May, Montana Gov. Greg Gianforte signed a bill to create a framework for licensed treatment centers to offer these interventions.
Patients are unlikely to receive their first therapies under the law before 2026, while the state’s health department establishes operating and safety standards for new and existing facilities, people involved with the legislation said.
Company executives said they are excited about the prospect of gathering real-world data from patients on their drug candidates while the therapies are still in development.
“Anytime I can safely, legally and ethically have more patients trying my medicine, it’s a net benefit because it’ll tell us if it works,” said Chris Bradley, CEO of longevity biotech Matter Bio, which is preparing to start a Phase 1 trial of a cancer therapy.
Positive early data could drive more funding from investors, biotech executives said.
Big Sky Rush?
Roughly 20 businesses, ranging from small biotech companies to medical-clinic operators, have reached out to express interest in operating in Montana under the new law, said Dylan Livingston, founder and CEO of a lobbying group that spearheaded the legislation.
Livingston’s group, the Alliance for Longevity Initiatives, is sponsored by longevity biotechs including the Sam Altman-backed Retro Biosciences.
CSBio, a contract manufacturer of custom peptides for biotechs and pharmaceutical companies, is planning to set up a manufacturing operation in Montana and open treatment clinics.
Chief Executive Jason Chang, who is based in the San Francisco Bay Area, had never been to the state before this year but has now visited twice. Plus, he said, he’s a fan of the set-in-Montana TV drama “Yellowstone.”
Chang recently posted a job opening for a person to act as a liaison among the company, regulators, health providers and patients under the new law.
The peptide-based treatments, which are for cancer patients, cost around $100,000 in total, he said.
He believes he could cut the cost in half by reducing overhead and treating more patients.
In Montana, patients will be required to provide consent that they reviewed other FDA-approved treatment options, received a provider’s recommendation and fully understand the potential risks, benefits and financial liabilities.
State Rep. Melody Cunningham, who was a practicing physician and medical researcher for nearly 30 years, fears the language of the law is designed to protect practitioners and clinics more than patients.
Cunningham voted against the bill, which ultimately passed the House by one vote. “I am afraid that patients will be harmed,” she said.
Company executives said they are clear-eyed about the potential harms with unapproved therapies.
Medical products that haven’t yet been approved might or might not be effective in treating a condition, and could cause unexpected and serious side effects, an FDA representative said.
“You have to be able to tell those people, this is the data we have, and that doesn’t mean we know everything,” Bradley said.
The Montana law grants people access only to therapies that have passed a Phase 1 trial. Phase 1 trials of a drug test for safety, not efficacy, and are often conducted in a small, healthy group of volunteers.
Many therapies fail to move beyond early trials. Only about 8% of drugs that enter clinical development ultimately receive regulatory approval, according to a 2021 report from a biotech trade association and industry analytics firms.
Patients will generally be responsible for paying, though Livingston said companies, philanthropic groups or newly raised tax funds from experimental clinics could defray costs.
If patients are harmed, healthcare providers and drugmakers are largely protected from legal liability under Montana’s laws unless they are negligent or fail to get proper consent from the patient.
Updated: 9-4-2025
Hot Mic In Beijing Captures Leaders’ Drive For Longevity
Xi, Putin and Kim—securely positioned heads of state—enjoy a chat about immortality.
BEIJING—The spectacle of the leaders of China, Russia and North Korea sitting side by side and watching a parade of intercontinental ballistic missiles and other advanced weapons in Beijing on Wednesday raised no shortage of questions of great consequence for the U.S.
A hot-mic moment between them shortly beforehand, as they walked together up a red-carpeted ramp, brought up one such riddle: Just how long will these three leaders be sticking around?
With advances in technology, Russian President Vladimir Putin tells China’s Xi Jinping through a translator, “human organs can be constantly transplanted, to the extent that people can get younger, perhaps even immortal.”
“It is predicted that by the end of the century, people in the future could live to be 150 years old,” Xi responds.
Kim Jong Un, three decades younger than the leaders of Russia and China, looks on smiling.
The simple answer is that the U.S. will likely have to contend with the trio of strongmen for years to come. Each is positioned to remain in power as long as their health allows—if not longer. None has an evident succession plan.
Xi, 72 years old, took power in 2012 and has pushed through overhauls that potentially allow him to serve as leader for life. Putin, also 72, has ruled Russia for 25 years; constitutional changes made on his watch allow him to stay in power until 2036.
Kim, 41, is in position to rule for life, as his father and grandfather did before him.
The health of each is a secret, making their appearance together Wednesday, and their small talk about longevity, fresh fodder for actuarial speculation.
The trio steadily climbed a flight of stairs, side by side, to the parade-viewing rostrum.
While there, Xi, dressed in a Mao suit buttoned to the throat, seemed to manage well on a steamy morning in Beijing, while Kim, seated next to the Chinese leader in a dark suit and tie, appeared to try to cool himself with a hand-held fan.
Putin appeared relaxed throughout the proceedings. In a press conference later, he reiterated his comments about extending human life.
“Modern means of improving health, medically and surgically, through organ transplants, allow humanity to hope that an active life can be extended even beyond what is possible today,” Putin said.
In the past, the smallest hints of infirmity have stirred debate, as when Xi was seen walking with a slight limp during a 2019 trip to Europe.
Xi has cut the number of foreign trips he takes in recent years, but maintains a robust schedule of travel around China.
Whether Xi designates an heir apparent could be the biggest question for China’s next Communist Party congress in 2027. Complicating the picture are Xi’s continuous purges of officials, at times targeting his own protégés.
The prospect that many of his closest allies might retire at the congress could intensify jockeying to succeed him, political analysts say.
Rumors have also often circulated about Putin’s health, such as when video footage showed his hand shaking, his odd posture while seated or a twitch in his leg.
Kim may be the youngest of the trio, but he still has health challenges.
He is a smoker with a family history of cardiovascular illness—his father and grandfather died of heart attacks, according to North Korea’s state media.
South Korea’s spy agency has described him as obese and weighing more than 300 pounds, with symptoms of high blood pressure and diabetes.
North Korea’s dynastic rule is founded on direct lineage to Kim’s grandfather, Kim Il Sung.
High-profile appearances of Kim Jong Un’s young daughter, including in Beijing this week, have stirred speculation that he is grooming her to succeed him.
North Korea’s state media very rarely reports that the country’s dictators are ill, portraying them as eternally strong and capable. Any signs of Kim’s vulnerability could trigger a power struggle.
While in Beijing this week, Kim’s aides appeared to take extraordinary measures to avoid leaving any biological clues, including wiping his chair down after he met with Putin, according to Russian video footage.
The Russian president, for his part, has long sought to portray an image of masculine virility, making appearances shirtless on horseback, playing hockey and hiking in Siberia.
The Kremlin has sought to deflect any rumors of ill health, as well as any parallels made in Russian media between Putin’s long reign and the gerontocracy of the late Soviet Union.
Putin has made a secret of any plans he might have for how to eventually transfer a grip on power that extends over the country’s sprawling security services, which control nearly all aspects of life in wartime Russia.
The personal aspect of Putin’s power has left Kremlin courtiers fearing a power vacuum—similar to the one left following Stalin’s death—if the leader, without warning, were to die.
Putin and Xi have developed visibly warm relations through years in power and scores of in-person meetings.
Throughout the procession of Chinese troops and weaponry on Wednesday, the two leaders could be seen chatting at length. On occasion, Kim chimed in as well.
It was a grouping that could complicate U.S. policy for years—possibly many years —to come.
Updated: 9-6-2025
The Billionaires Fueling The Quest For Longer Life
Investors including Peter Thiel and Sam Altman are making big bets on where longevity science is headed.
How much would you invest in the possibility of living to 150 or beyond? Or having 20 extra healthy years?
For the ultrawealthy, it’s more than $5 billion over the past 2½ decades, according to a Wall Street Journal analysis of longevity investment deals in PitchBook, public company statements and regulatory filings.
Silicon Valley giants Peter Thiel, Sam Altman, Yuri Milner and Marc Andreessen are among the boldface names behind the influx of money in the longevity industry.
Thiel’s quest for longer life spans nearly a dozen companies—some of which were funded by his venture firm and others by a nonprofit foundation he backed—that raised more than $700 million, according to the Journal’s analysis.
They and other wealthy investors have helped push what was once something of an academic backwater into the cultural mainstream.
Many companies ultimately fail, but the ultrawealthy and other enthusiasts are following the money and the science to decide where to invest and what to take.
Who’s Betting Big
Here’s a look at the network of entrepreneurs and big investors building up the longevity space, the sector’s growth and what scientific approaches the ultrawealthy are placing bets on.
These companies are part of a constellation of more than 200 startups and nonprofits involving nearly 1,000 investors. Together they raised more than $12.5 billion in the past 2½ decades from not only the ultrawealthy but also social-media influencers, celebrity scientists and actors chasing a breakthrough, according to the Journal’s analysis.
Thiel’s mentee, OpenAI CEO Altman, placed a big bet–$180 million–on Retro Biosciences, which aims to develop drugs to rejuvenate and reprogram aging cells.
Retro CEO Joe Betts-LaCroix previously worked for Halcyon Molecular, a former biotech funded by Thiel’s Founders Fund.
Thiel’s fund also backed NewLimit, co-founded in 2021 by Coinbase CEO Brian Armstrong.
NewLimit, with a focus on reversing cell aging, raised more than $200 million from ultrawealthy investors including at least nine billionaires or their funds such as Palantir co-founder Joe Lonsdale, ex-Google CEO Eric Schmidt and Sun Microsystems co-founder Vinod Khosla.
Peter Diamandis, a Silicon Valley entrepreneur and longevity enthusiast, also invested in NewLimit.
Lonsdale’s firm, 8VC, backed cellular rejuvenation startup Altos Labs, which drew support from Robert Nelsen’s Arch Venture Partners and tech investor Milner.
Not all longevity bets pay off. Unity Biotechnology raised $355 million since 2013 to develop medicines targeting cells linked to aging, according to company filings. The company was recently delisted from the Nasdaq and announced plans in June to dissolve.
Khosla’s firm invested in several longevity startups, which collectively raised more than $1 billion, the Journal’s analysis shows.
Khosla Ventures and venture capitalist Andreessen’s fund invested in BioAge Labs.
Salesforce co-founder Marc Benioff and Khosla’s fund invested in Viome Life Sciences. BOLD Capital Partners, a venture fund co-founded by Diamandis, also invested in Viome.
Longevity startups are increasingly backed by celebrities and other well-connected people.
Function Health, which focuses on lab tests and lifestyle suggestions, attracted investments from Kevin Hart, Matt Damon and Dr. Casey Means, President Trump’s pick for surgeon general.
How Much Money
Money has been pouring in from some of the wealthiest investors, fueled by these personal connections and passions.
Armstrong’s fundraising for NewLimit includes a $130 million round in May. Among the company’s investors are the funds of fellow billionaires Khosla and Thiel.
Altos, launched in 2022, raised $3 billion—the most of any company identified by the Journal—to develop technology to rejuvenate cells.
Artificial-intelligence drug discovery company Insilico Medicine, which is backed by Diamandis’s BOLD Capital, raised more than $500 million to treat multiple age-related diseases.
Another company, BioAge Labs, which is developing drugs to treat diseases of aging, raised $559 million, which includes investments from Khosla Ventures and Andreessen’s a16z fund.
BioAge’s chief executive, Kristen Fortney, met investors interested in her research at a Stanford lab and through her involvement in a salon for longevity enthusiasts run by Betts-LaCroix.
BioAge went public in 2024, raising the funds to develop an obesity drug and other programs. A trial of an obesity drug halted later that year over safety concerns. BioAge launched a new trial of a different obesity drug this year.
“A lot of people already make it past the age of 100 and they’re healthy. So why can’t that be achievable for all of us?” Fortney said.
That concept has taken hold and is helping drive investors, with the average fundraising round for longevity companies growing by more than 20% in the past decade to nearly $43 million this year, by the Journal’s calculations.
Why They Invest
Many of the ultrawealthy individuals flooding the longevity space are motivated by highly personal reasons.
Naveen Jain says his father’s death from pancreatic cancer led him to found Viome Life Sciences, which has raised over $230 million.
Viome sells at-home health tests and analyzes the data to make personalized nutrition and supplement recommendations.
Jain, a billionaire, invested $30 million of his own money in Viome. Benioff and Khosla Ventures are also investors. “I want to make aging optional,” Jain says.
Stéphane Bancel, CEO of Moderna, tried a diet designed to mimic the effects of fasting developed by longevity scientist Valter Longo.
Bancel later led a $47 million funding round for Longo’s company L-Nutra, which develops fasting nutrition programs.
“I want to be able to have the biggest impact,” says Bancel.
Khosla through his firm is one of the most prolific longevity investors identified by the Journal, with each of the investments focused on different aspects of aging.
“At 70, someone should feel like a 40-year-old,” he said during an interview with the Superteam Podcast, a group of cryptocurrency enthusiasts.
The Science Attracting Big Money
There is no consensus on what makes something a longevity company. The Journal identified three key areas the ultrawealthy are focused on, by companies’ publicly reported missions: Efforts to reverse or change aging, develop treatments for age-related diseases, or sell products or services claiming to improve health and extend lifespan.
The quest for ways to “reprogram” and rejuvenate cells is one of the biggest generating buzz in Silicon Valley circles.
The idea of returning people to a more youthful state is propelling Altos, Retro, Juvenescence and more than 80 other companies to raise some $5 billion.
Retro says it is trying to raise another $1 billion.
Companies seeking treatments for diseases linked to aging include BioAge Labs. Nearly 60 biotechs in this space have raised nearly $5 billion.
For those interested in antiaging strategies that can be tried right now, big money is pouring into companies selling health trackers, supplements and cosmetics, raising some $2.6 billion.
Longevity Personalities
Prominent personalities have helped build longevity’s cachet among wealthy investors and the public, founding companies and building demand.
Peter Attia: Author of a bestselling longevity book and host of a popular podcast, Attia co-founded the high-end clinic Biograph with Silicon Valley entrepreneur John Hering.
David Sinclair: A popular lecturer on the longevity circuit, the Harvard professor’s genetics lab is a frequent stop for wealthy individuals interested in learning about and sometimes funding age-reversal research.
Bryan Johnson: His Silicon Valley-fueled fortune and willingness to spend $1 million a year on self-experiments attempting to slow down his aging led to a Netflix documentary and public attention.
Peter Diamandis: The entrepreneur and investor tapped billionaires and other wealthy individuals as donors for the XPrize Foundation he founded and its XPrize Healthspan, a prize competition with a $101 million purse to develop ways to reverse aspects of aging.
Related Articles:
Bitcoin Information & Resources (#GotBitcoin)
Sean Harribance Shares His Psychic Gifts With The Public
Scientists Achieve Real-Time Communication With Lucid Dreamers In Breakthrough
Does Getting Stoned Help You Get Toned? Gym Rats Embrace Marijuana
Marijuana In Africa Is Like The Gold Rush For America In The 1800s
The Perfect Wine And Weed To Get You Through The Coronavirus Pandemic Lockdown
Mike Tyson’s 420-Acre Weed Ranch Rakes In $500K A Month
What Sex Workers Want To Do With Bitcoin
“Is Bitcoin Reacting To The Chaos Or Is Bitcoin Causing The Chaos?” Max Keiser
How To Safely Store Deposits If You Have More Than $250,000
How To Host A Decentralized Website
Banks Lose Billions (Approx. $52 Billion) As Depositors Seek Higher Deposit Yields #GotBitcoin
Crypto User Recovers Long-Lost Private Keys To Access $4M In Bitcoin
Stripe Stops Processing Payments For Trump Campaign Website
Bitcoin Whales Are Profiting As ‘Weak Hands’ Sell BTC After Price Correction
Pentagon Sees Giant Cargo Cranes As Possible Chinese Spying Tools
Bitcoin’s Volatility Should Burn Investors. It Hasn’t
Bitcoin’s Latest Record Run Is Less Volatile Than The 2017 Boom
“Lettuce Hands” Refers To Investors Who Can’t Deal With The Volatility Of The Cryptocurrency Markets
The Bitcoiners Who Live Off The Grid
US Company Now Lets Travelers Pay For Passports With Bitcoin
After A Year Without Rowdy Tourists, European Cities Want To Keep It That Way
Director Barry Jenkins Is The Travel Nerd’s Travel Nerd
Four Stories Of How People Traveled During Covid
Who Is A Perpetual Traveler (AKA Digital Nomad) Under The US Tax Code
Tricks For Making A Vacation Feel Longer—And More Fulfilling
Travel Has Bounced Back From Coronavirus, But Tourists Stick Close To Home
Nurses Travel From Coronavirus Hot Spot To Hot Spot, From New York To Texas
How To Travel Luxuriously Post- Covid-19, From Private Jets To Hotel Buyouts
Ultimate Travel Resource Covering Business, Personal, Cruise, Flying, Etc.)
Does Bitcoin Boom Mean ‘Better Gold’ Or Bigger Bubble?
Bitcoin’s Slide Dents Price Momentum That Dwarfed Everything
Retail Has Arrived As Paypal Clears $242M In Crypto Sales Nearly Double The Previous Record
Jarlsberg Cheese Offers Significant Bone & Heart-Health Benefits Thanks To Vitamin K2, Says Study
Chrono-Pharmacology Reveals That “When” You Take Your Medication Can Make A Life-Saving Difference
Ultimate Resource For News, Breakthroughs And Innovations In Healthcare
Ultimate Resource For Cooks, Chefs And The Latest Food Trends
Ethereum Use Cases You Might Not Know
Will 1% Yield Force The Fed Into Curve Control?
Ultimate Resource On Hong Kong Vying For World’s Crypto Hub #GotBitcoin
France Moves To Ban Anonymous Crypto Accounts To Prevent Money Laundering
Numerous Times That US (And Other) Regulators Stepped Into Crypto
Where Does This 28% Bitcoin Price Drop Rank In History? Not Even In The Top 5
Traditional Investors View Bitcoin As If It Were A Technology Stock
3 Reasons Why Bitcoin Price Abruptly Dropped 6% After Reaching $15,800
As Bitcoin Approaches $25,000 It Breaks Correlation With Equities
UK Treasury Calls For Feedback On Approach To Cryptocurrency And Stablecoin Regulation
Bitcoin Rebounds While Leaving Everyone In Dark On True Worth
Slow-Twitch vs. Fast-Twitch Muscle Fibers
Biden, Obama Release Campaign Video Applauding Their Achievements
Joe Biden Tops Donald Trump In Polls And Leads In Fundraising (#GotBitcoin)
Trump Gets KPOP’d And Tic Toc’d As Teens Mobilized To Derail Trump’s Tulsa Rally
Schwab’s $200 Million Charge Puts Scrutiny On Robo-Advising
TikTok Is The Place To Go For Financial Advice If You’re A Young Adult
TikTok Is The Place To Go For Financial Advice If You’re A Young Adult
American Shoppers Just Can’t Pass Up A Bargain And Department Stores Pay The Price #GotBitcoin
Motley Fool Adding $5M In Bitcoin To Its ‘10X Portfolio’ — Has A $500K Price Target
Mad Money’s Jim Cramer Invests 1% Of Net Worth In Bitcoin Says, “Gold Is Dangerous”
Ultimate Resource For Financial Advisers By Financial Advisers On Crypto
Anti-ESG Movement Reveals How Blackrock Pulls-off World’s Largest Ponzi Scheme
The Bitcoin Ordinals Protocol Has Caused A Resurgence In Bitcoin Development And Interest
Bitcoin Takes ‘Lion’s Share’ As Institutional Inflows Hit 7-Month High
Bitcoin’s Future Depends On A Handful of Mysterious Coders
Billionaire Hedge Fund Investor Stanley Druckenmiller Says He Owns Bitcoin In CNBC Interview
Bitcoin Billionaire Chamath Palihapitiya Opts Out Of Run For California Governor
Billionaire Took Psychedelics, Got Bitcoin And Is Now Into SPACs
Billionaire’s Bitcoin Dream Shapes His Business Empire In Norway
Trading Firm Of Richest Crypto Billionaire Reveals Buying ‘A Lot More’ Bitcoin Below $30K
Simple Tips To Ensure Your Digital Surveillance Works As It Should
Big (4) Audit Firms Blasted By PCAOB And Gary Gensler, Head Of SEC (#GotBitcoin)
What Crypto Users Need Know About Changes At The SEC
The Ultimate Resource For The Bitcoin Miner And The Mining Industry (Page#2) #GotBitcoin
How Cryptocurrency Can Help In Paying Universal Basic Income (#GotBitcoin)
Gautam Adani Was Briefly World’s Richest Man Only To Be Brought Down By An American Short-Seller
Global Crypto Industry Pledges Aid To Turkey Following Deadly Earthquakes
Money Supply Growth Went Negative Again In December Another Sign Of Recession #GotBitcoin
Here Is How To Tell The Difference Between Bitcoin And Ethereum
Crypto Investors Can Purchase Bankruptcy ‘Put Options’ To Protect Funds On Binance, Coinbase, Kraken
Bitcoin Developers Must Face UK Trial Over Lost Cryptoassets
Google Issues Warning For 2 Billion Chrome Users
How A Lawsuit Against The IRS Is Trying To Expand Privacy For Crypto Users
IRS Uses Cellphone Location Data To Find Suspects
IRS Failed To Collect $2.4 Billion In Taxes From Millionaires
Treasury Calls For Crypto Transfers Over $10,000 To Be Reported To IRS
Can The IRS Be Trusted With Your Data?
US Ransomware Attack Suspect Hails From A Small Ukrainian Town
Alibaba Admits It Was Slow To Report Software Bug After Beijing Rebuke
Japan Defense Ministry Finds Security Threat In Hack
Raoul Pal Believes Institutions Have Finished Taking Profits As Year Winds Up
Yosemite Is Forcing Native American Homeowners To Leave Without Compensation. Here’s Why
What Is Dollar Cost Averaging Bitcoin?
Ultimate Resource On Bitcoin Unit Bias
Best Travel Credit Cards of 2022-2023
A Guarded Generation: How Millennials View Money And Investing (#GotBitcoin)
Bitcoin Enthusiast And CEO Brian Armstrong Buys Los Angeles Home For $133 Million
Nasdaq-Listed Blockchain Firm BTCS To Offer Dividend In Bitcoin; Shares Surge
Ultimate Resource On Kazakhstan As Second In Bitcoin Mining Hash Rate In The World After US
Ultimate Resource On Solana Outages And DDoS Attacks
How Jessica Simpson Almost Lost Her Name And Her Billion Dollar Empire
Sidney Poitier, Actor Who Made Oscars History, Dies At 94
Green Comet Will Be Visible As It Passes By Earth For First Time In 50,000 Years
FTX (SBF) Got Approval From F.D.I.C., State Regulators And Federal Reserve To Buy Tiny Bank!!!
Joe Rogan: I Have A Lot Of Hope For Bitcoin
Teen Cyber Prodigy Stumbled Onto Flaw Letting Him Hijack Teslas
Spyware Finally Got Scary Enough To Freak Lawmakers Out—After It Spied On Them
The First Nuclear-Powered Bitcoin Mine Is Here. Maybe It Can Clean Up Energy FUD
The World’s Best Crypto Policies: How They Do It In 37 Nations
Tonga To Copy El Salvador’s Bill Making Bitcoin Legal Tender, Says Former MP
Wordle Is The New “Lingo” Turning Fans Into Argumentative Strategy Nerds
Prospering In The Pandemic, Some Feel Financial Guilt And Gratitude
Is Art Therapy The Path To Mental Well-Being?
New York, California, New Jersey, And Alabama Move To Ban ‘Forever Chemicals’ In Firefighting Foam
The Mystery Of The Wasting House-Cats
What Pet Owners Should Know About Chronic Kidney Disease In Dogs And Cats
Pets Score Company Perks As The ‘New Dependents’
Why Is My Cat Rubbing His Face In Ants?
Natural Cure For Hyperthyroidism In Cats Including How To Switch Him/Her To A Raw Food Diet
Ultimate Resource For Cat Lovers
FDA Approves First-Ever Arthritis Pain Management Drug For Cats
Ultimate Resource On Duke of York’s Prince Andrew And His Sex Scandal
Walmart Filings Reveal Plans To Create Cryptocurrency, NFTs
Bitcoin’s Dominance of Crypto Payments Is Starting To Erode
T-Mobile Says Hackers Stole Data On About 37 Million Customers
Jack Dorsey Announces Bitcoin Legal Defense Fund
More Than 100 Millionaires Signed An Open Letter Asking To Be Taxed More Heavily
Federal Regulator Says Credit Unions Can Partner With Crypto Providers
What’s Behind The Fascination With Smash-And-Grab Shoplifting?
Train Robberies Are A Problem In Los Angeles, And No One Agrees On How To Stop Them
US Stocks Historically Deliver Strong Gains In Fed Hike Cycles (GotBitcoin)
Ian Alexander Jr., Only Child of Regina King, Dies At Age 26
Amazon Ends Its Charity Donation Program Amazonsmile After Other Cost-Cutting Efforts
Indexing Is Coming To Crypto Funds Via Decentralized Exchanges
Doctors Show Implicit Bias Towards Black Patients
Darkmail Pushes Privacy Into The Hands Of NSA-Weary Customers
3D Printing Make Anything From Candy Bars To Hand Guns
Stealing The Blood Of The Young May Make You More Youthful
Henrietta Lacks And Her Remarkable Cells Will Finally See Some Payback
AL_A Wins Approval For World’s First Magnetized Fusion Power Plant
Want To Be Rich? Bitcoin’s Limited Supply Cap Means You Only Need 0.01 BTC
Smart Money Is Buying Bitcoin Dip. Stocks, Not So Much
McDonald’s Jumps On Bitcoin Memewagon, Crypto Twitter Responds
America COMPETES Act Would Be Disastrous For Bitcoin Cryptocurrency And More
Lyn Alden On Bitcoin, Inflation And The Potential Coming Energy Shock
Inflation And A Tale of Cantillionaires
El Salvador Plans Bill To Adopt Bitcoin As Legal Tender
Miami Mayor Says City Employees Should Be Able To Take Their Salaries In Bitcoin
Vast Troves of Classified Info Undermine National Security, Spy Chief Says
BREAKING: Arizona State Senator Introduces Bill To Make Bitcoin Legal Tender
San Francisco’s Historic Surveillance Law May Get Watered Down
How Bitcoin Contributions Funded A $1.4M Solar Installation In Zimbabwe
California Lawmaker Says National Privacy Law Is a Priority
The Pandemic Turbocharged Online Privacy Concerns
How To Protect Your Online Privacy While Working From Home
Researchers Use GPU Fingerprinting To Track Users Online
Japan’s $1 Trillion Crypto Market May Ease Onerous Listing Rules
Ultimate Resource On A Weak / Strong Dollar’s Impact On Bitcoin
Fed Money Printer Goes Into Reverse (Quantitative Tightening): What Does It Mean For Crypto?
Crypto Market Is Closer To A Bottom Than Stocks (#GotBitcoin)
When World’s Central Banks Get It Wrong, Guess Who Pays The Price😂😹🤣 (#GotBitcoin)
“Better Days Ahead With Crypto Deleveraging Coming To An End” — Joker
Crypto Funds Have Seen Record Investment Inflow In Recent Weeks
Bitcoin’s Epic Run Is Winning More Attention On Wall Street
Ultimate Resource For Crypto Mergers And Acquisitions (M&A) (#GotBitcoin)
Why Wall Street Is Literally Salivating Over Bitcoin
Nasdaq-Listed MicroStrategy And Others Wary Of Looming Dollar Inflation, Turns To Bitcoin And Gold
Bitcoin For Corporations | Michael Saylor | Bitcoin Corporate Strategy
Ultimate Resource On Myanmar’s Involvement With Crypto-Currencies
‘I Cry Every Day’: Olympic Athletes Slam Food, COVID Tests And Conditions In Beijing
Does Your Baby’s Food Contain Toxic Metals? Here’s What Our Investigation Found
Ultimate Resource For Pro-Crypto Lobbying And Non-Profit Organizations
Ultimate Resource On BlockFi, Celsius And Nexo
Petition Calling For Resignation Of U.S. Securities/Exchange Commission Chair Gary Gensler
100 Million Americans Can Legally Bet on the Super Bowl. A Spot Bitcoin ETF? Forget About it!
Green Finance Isn’t Going Where It’s Needed
Shedding Some Light On The Murky World Of ESG Metrics
SEC Targets Greenwashers To Bring Law And Order To ESG
BlackRock (Assets Under Management $7.4 Trillion) CEO: Bitcoin Has Caught Our Attention
Canada’s Major Banks Go Offline In Mysterious (Bank Run?) Hours-Long Outage (#GotBitcoin)
On-Chain Data: A Framework To Evaluate Bitcoin
On Its 14th Birthday, Bitcoin’s 1,690,706,971% Gain Looks Kind of… Well Insane
The Most Important Health Metric Is Now At Your Fingertips
American Bargain Hunters Flock To A New Online Platform Forged In China
Why We Should Welcome Another Crypto Winter
Traders Prefer Gold, Fiat Safe Havens Over Bitcoin As Russia Goes To War
Music Distributor DistroKid Raises Money At $1.3 Billion Valuation
Nas Selling Rights To Two Songs Via Crypto Music Startup Royal
Ultimate Resource On Music Catalog Deals
Ultimate Resource On Music And NFTs And The Implications For The Entertainment Industry
Lead And Cadmium Could Be In Your Dark Chocolate
Catawba, Native-American Tribe Approves First Digital Economic Zone In The United States
The Miracle Of Blockchain’s Triple Entry Accounting
How And Why To Stimulate Your Vagus Nerve!
Housing Boom Brings A Shortage Of Land To Build New Homes
Biden Lays Out His Blueprint For Fair Housing
No Grave Dancing For Sam Zell Now. He’s Paying Up For Hot Properties
Cracks In The Housing Market Are Starting To Show
Ever-Growing Needs Strain U.S. Food Bank Operations
Food Pantry Helps Columbia Students Struggling To Pay Bills
Food Insecurity Driven By Climate Change Has Central Americans Fleeing To The U.S.
Housing Insecurity Is Now A Concern In Addition To Food Insecurity
Families Face Massive Food Insecurity Levels
US Troops Going Hungry (Food Insecurity) Is A National Disgrace
Everything You Should Know About Community Fridges, From Volunteering To Starting Your Own
Russia’s Independent Journalists Including Those Who Revealed The Pandora Papers Need Your Help
10 Women Who Used Crypto To Make A Difference In 2021
Happy International Women’s Day! Leaders Share Their Experiences In Crypto
Dollar On Course For Worst Performance In Over A Decade (#GotBitcoin)
Juice The Stock Market And Destroy The Dollar!! (#GotBitcoin)
Unusual Side Hustles You May Not Have Thought Of
Ultimate Resource On Global Inflation And Rising Interest Rates (#GotBitcoin)
The Fed Is Setting The Stage For Hyper-Inflation Of The Dollar (#GotBitcoin)
An Antidote To Inflation? ‘Buy Nothing’ Groups Gain Popularity
Why Is Bitcoin Dropping If It’s An ‘Inflation Hedge’?
Lyn Alden Talks Bitcoin, Inflation And The Potential Coming Energy Shock
Ultimate Resource On How Black Families Can Fight Against Rising Inflation (#GotBitcoin)
What The Fed’s Rate Hike Means For Inflation, Housing, Crypto And Stocks
Egyptians Buy Bitcoin Despite Prohibitive New Banking Laws
Archaeologists Uncover Five Tombs In Egypt’s Saqqara Necropolis
History of Alchemy From Ancient Egypt To Modern Times
Former World Bank Chief Didn’t Act On Warnings Of Sexual Harassment
Does Your Hospital or Doctor Have A Financial Relationship With Big Pharma?
Ultimate Resource Covering The Crisis Taking Place In The Nickel Market
Apple Along With Meta And Secret Service Agents Fooled By Law Enforcement Impersonators
Handy Tech That Can Support Your Fitness Goals
How To Naturally Increase Your White Blood Cell Count
Ultimate Source For Russians Oligarchs And The Impact Of Sanctions On Them
Ultimate Source For Bitcoin Price Manipulation By Wall Street
Russia, Sri Lanka And Lebanon’s Defaults Could Be The First Of Many (#GotBitcoin)
Will Community Group Buying Work In The US?
Building And Running Businesses In The ‘Spirit Of Bitcoin’
What Is The Mysterious Liver Disease Hurting (And Killing) Children?
Citigroup Trader Is Scapegoat For Flash Crash In European Stocks (#GotBitcoin)
Bird Flu Outbreak Approaches Worst Ever In U.S. With 37 Million Animals Dead
Financial Inequality Grouped By Race For Blacks, Whites And Hispanics
How Black Businesses Can Prosper From Targeting A Trillion-Dollar Black Culture Market (#GotBitcoin)
Ultimate Resource For Central Bank Digital Currencies (#GotBitcoin) Page#2
Meet The Crypto Angel Investor Running For Congress In Nevada (#GotBitcoin?)
Introducing BTCPay Vault – Use Any Hardware Wallet With BTCPay And Its Full Node (#GotBitcoin?)
How Not To Lose Your Coins In 2020: Alternative Recovery Methods (#GotBitcoin?)
H.R.5635 – Virtual Currency Tax Fairness Act of 2020 ($200.00 Limit) 116th Congress (2019-2020)
Adam Back On Satoshi Emails, Privacy Concerns And Bitcoin’s Early Days
The Prospect of Using Bitcoin To Build A New International Monetary System Is Getting Real
How To Raise Funds For Australia Wildfire Relief Efforts (Using Bitcoin And/Or Fiat )
Former Regulator Known As ‘Crypto Dad’ To Launch Digital-Dollar Think Tank (#GotBitcoin?)
Currency ‘Cold War’ Takes Center Stage At Pre-Davos Crypto Confab (#GotBitcoin?)
A Blockchain-Secured Home Security Camera Won Innovation Awards At CES 2020 Las Vegas
Bitcoin’s Had A Sensational 11 Years (#GotBitcoin?)
Sergey Nazarov And The Creation Of A Decentralized Network Of Oracles
Google Suspends MetaMask From Its Play App Store, Citing “Deceptive Services”
Christmas Shopping: Where To Buy With Crypto This Festive Season
At 8,990,000% Gains, Bitcoin Dwarfs All Other Investments This Decade
Coinbase CEO Armstrong Wins Patent For Tech Allowing Users To Email Bitcoin
Bitcoin Has Got Society To Think About The Nature Of Money
How DeFi Goes Mainstream In 2020: Focus On Usability (#GotBitcoin?)
Dissidents And Activists Have A Lot To Gain From Bitcoin, If Only They Knew It (#GotBitcoin?)
At A Refugee Camp In Iraq, A 16-Year-Old Syrian Is Teaching Crypto Basics
Bitclub Scheme Busted In The US, Promising High Returns From Mining
Bitcoin Advertised On French National TV
Germany: New Proposed Law Would Legalize Banks Holding Bitcoin
How To Earn And Spend Bitcoin On Black Friday 2019
The Ultimate List of Bitcoin Developments And Accomplishments
Charities Put A Bitcoin Twist On Giving Tuesday
Family Offices Finally Accept The Benefits of Investing In Bitcoin
An Army Of Bitcoin Devs Is Battle-Testing Upgrades To Privacy And Scaling
Bitcoin ‘Carry Trade’ Can Net Annual Gains With Little Risk, Says PlanB
Max Keiser: Bitcoin’s ‘Self-Settlement’ Is A Revolution Against Dollar
Blockchain Can And Will Replace The IRS
China Seizes The Blockchain Opportunity. How Should The US Respond? (#GotBitcoin?)
Jack Dorsey: You Can Buy A Fraction Of Berkshire Stock Or ‘Stack Sats’
Bitcoin Price Skyrockets $500 In Minutes As Bakkt BTC Contracts Hit Highs
Bitcoin’s Irreversibility Challenges International Private Law: Legal Scholar
Bitcoin Has Already Reached 40% Of Average Fiat Currency Lifespan
Yes, Even Bitcoin HODLers Can Lose Money In The Long-Term: Here’s How (#GotBitcoin?)
Unicef To Accept Donations In Bitcoin (#GotBitcoin?)
Former Prosecutor Asked To “Shut Down Bitcoin” And Is Now Face Of Crypto VC Investing (#GotBitcoin?)
Switzerland’s ‘Crypto Valley’ Is Bringing Blockchain To Zurich
Next Bitcoin Halving May Not Lead To Bull Market, Says Bitmain CEO
Bitcoin Developer Amir Taaki, “We Can Crash National Economies” (#GotBitcoin?)
Veteran Crypto And Stocks Trader Shares 6 Ways To Invest And Get Rich
Is Chainlink Blazing A Trail Independent Of Bitcoin?
Nearly $10 Billion In BTC Is Held In Wallets Of 8 Crypto Exchanges (#GotBitcoin?)
SEC Enters Settlement Talks With Alleged Fraudulent Firm Veritaseum (#GotBitcoin?)
Blockstream’s Samson Mow: Bitcoin’s Block Size Already ‘Too Big’
Attorneys Seek Bank Of Ireland Execs’ Testimony Against OneCoin Scammer (#GotBitcoin?)
OpenLibra Plans To Launch Permissionless Fork Of Facebook’s Stablecoin (#GotBitcoin?)
Tiny $217 Options Trade On Bitcoin Blockchain Could Be Wall Street’s Death Knell (#GotBitcoin?)
Class Action Accuses Tether And Bitfinex Of Market Manipulation (#GotBitcoin?)
Sharia Goldbugs: How ISIS Created A Currency For World Domination (#GotBitcoin?)
Bitcoin Eyes Demand As Hong Kong Protestors Announce Bank Run (#GotBitcoin?)
How To Securely Transfer Crypto To Your Heirs
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
Crypto News From The Spanish-Speaking World (#GotBitcoin?)
Financial Services Giant Morningstar To Offer Ratings For Crypto Assets (#GotBitcoin?)
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
The Original Sins Of Cryptocurrencies (#GotBitcoin?)
Bitcoin Is The Fraud? JPMorgan Metals Desk Fixed Gold Prices For Years (#GotBitcoin?)
Israeli Startup That Allows Offline Crypto Transactions Secures $4M (#GotBitcoin?)
[PSA] Non-genuine Trezor One Devices Spotted (#GotBitcoin?)
Bitcoin Stronger Than Ever But No One Seems To Care: Google Trends (#GotBitcoin?)
First-Ever SEC-Qualified Token Offering In US Raises $23 Million (#GotBitcoin?)
You Can Now Prove A Whole Blockchain With One Math Problem – Really
Crypto Mining Supply Fails To Meet Market Demand In Q2: TokenInsight
$2 Billion Lost In Mt. Gox Bitcoin Hack Can Be Recovered, Lawyer Claims (#GotBitcoin?)
Fed Chair Says Agency Monitoring Crypto But Not Developing Its Own (#GotBitcoin?)
Wesley Snipes Is Launching A Tokenized $25 Million Movie Fund (#GotBitcoin?)
Mystery 94K BTC Transaction Becomes Richest Non-Exchange Address (#GotBitcoin?)
A Crypto Fix For A Broken International Monetary System (#GotBitcoin?)
Four Out Of Five Top Bitcoin QR Code Generators Are Scams: Report (#GotBitcoin?)
Waves Platform And The Abyss To Jointly Launch Blockchain-Based Games Marketplace (#GotBitcoin?)
Bitmain Ramps Up Power And Efficiency With New Bitcoin Mining Machine (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Miss Finland: Bitcoin’s Risk Keeps Most Women Away From Cryptocurrency (#GotBitcoin?)
Artist Akon Loves BTC And Says, “It’s Controlled By The People” (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Co-Founder Of LinkedIn Presents Crypto Rap Video: Hamilton Vs. Satoshi (#GotBitcoin?)
Crypto Insurance Market To Grow, Lloyd’s Of London And Aon To Lead (#GotBitcoin?)
No ‘AltSeason’ Until Bitcoin Breaks $20K, Says Hedge Fund Manager (#GotBitcoin?)
NSA Working To Develop Quantum-Resistant Cryptocurrency: Report (#GotBitcoin?)
Custody Provider Legacy Trust Launches Crypto Pension Plan (#GotBitcoin?)
Vaneck, SolidX To Offer Limited Bitcoin ETF For Institutions Via Exemption (#GotBitcoin?)
Russell Okung: From NFL Superstar To Bitcoin Educator In 2 Years (#GotBitcoin?)
Bitcoin Miners Made $14 Billion To Date Securing The Network (#GotBitcoin?)
Why Does Amazon Want To Hire Blockchain Experts For Its Ads Division?
Argentina’s Economy Is In A Technical Default (#GotBitcoin?)
Blockchain-Based Fractional Ownership Used To Sell High-End Art (#GotBitcoin?)
Portugal Tax Authority: Bitcoin Trading And Payments Are Tax-Free (#GotBitcoin?)
Bitcoin ‘Failed Safe Haven Test’ After 7% Drop, Peter Schiff Gloats (#GotBitcoin?)
Bitcoin Dev Reveals Multisig UI Teaser For Hardware Wallets, Full Nodes (#GotBitcoin?)
Bitcoin Price: $10K Holds For Now As 50% Of CME Futures Set To Expire (#GotBitcoin?)
Bitcoin Realized Market Cap Hits $100 Billion For The First Time (#GotBitcoin?)
Stablecoins Begin To Look Beyond The Dollar (#GotBitcoin?)
Bank Of England Governor: Libra-Like Currency Could Replace US Dollar (#GotBitcoin?)
Binance Reveals ‘Venus’ — Its Own Project To Rival Facebook’s Libra (#GotBitcoin?)
The Real Benefits Of Blockchain Are Here. They’re Being Ignored (#GotBitcoin?)
CommBank Develops Blockchain Market To Boost Biodiversity (#GotBitcoin?)
SEC Approves Blockchain Tech Startup Securitize To Record Stock Transfers (#GotBitcoin?)
SegWit Creator Introduces New Language For Bitcoin Smart Contracts (#GotBitcoin?)
You Can Now Earn Bitcoin Rewards For Postmates Purchases (#GotBitcoin?)
Bitcoin Price ‘Will Struggle’ In Big Financial Crisis, Says Investor (#GotBitcoin?)
Fidelity Charitable Received Over $100M In Crypto Donations Since 2015 (#GotBitcoin?)
Would Blockchain Better Protect User Data Than FaceApp? Experts Answer (#GotBitcoin?)
Just The Existence Of Bitcoin Impacts Monetary Policy (#GotBitcoin?)
What Are The Biggest Alleged Crypto Heists And How Much Was Stolen? (#GotBitcoin?)
IRS To Cryptocurrency Owners: Come Clean, Or Else!
Coinbase Accidentally Saves Unencrypted Passwords Of 3,420 Customers (#GotBitcoin?)
Bitcoin Is A ‘Chaos Hedge, Or Schmuck Insurance‘ (#GotBitcoin?)
Bakkt Announces September 23 Launch Of Futures And Custody
Coinbase CEO: Institutions Depositing $200-400M Into Crypto Per Week (#GotBitcoin?)
Researchers Find Monero Mining Malware That Hides From Task Manager (#GotBitcoin?)
Crypto Dusting Attack Affects Nearly 300,000 Addresses (#GotBitcoin?)
A Case For Bitcoin As Recession Hedge In A Diversified Investment Portfolio (#GotBitcoin?)
SEC Guidance Gives Ammo To Lawsuit Claiming XRP Is Unregistered Security (#GotBitcoin?)
15 Countries To Develop Crypto Transaction Tracking System: Report (#GotBitcoin?)
US Department Of Commerce Offering 6-Figure Salary To Crypto Expert (#GotBitcoin?)
Mastercard Is Building A Team To Develop Crypto, Wallet Projects (#GotBitcoin?)
Canadian Bitcoin Educator Scams The Scammer And Donates Proceeds (#GotBitcoin?)
Amazon Wants To Build A Blockchain For Ads, New Job Listing Shows (#GotBitcoin?)
Shield Bitcoin Wallets From Theft Via Time Delay (#GotBitcoin?)
Blockstream Launches Bitcoin Mining Farm With Fidelity As Early Customer (#GotBitcoin?)
Commerzbank Tests Blockchain Machine To Machine Payments With Daimler (#GotBitcoin?)
Man Takes Bitcoin Miner Seller To Tribunal Over Electricity Bill And Wins (#GotBitcoin?)
Bitcoin’s Computing Power Sets Record As Over 100K New Miners Go Online (#GotBitcoin?)
Walmart Coin And Libra Perform Major Public Relations For Bitcoin (#GotBitcoin?)
Judge Says Buying Bitcoin Via Credit Card Not Necessarily A Cash Advance (#GotBitcoin?)
Poll: If You’re A Stockowner Or Crypto-Currency Holder. What Will You Do When The Recession Comes?
1 In 5 Crypto Holders Are Women, New Report Reveals (#GotBitcoin?)
Beating Bakkt, Ledgerx Is First To Launch ‘Physical’ Bitcoin Futures In Us (#GotBitcoin?)
Facebook Warns Investors That Libra Stablecoin May Never Launch (#GotBitcoin?)
Government Money Printing Is ‘Rocket Fuel’ For Bitcoin (#GotBitcoin?)
Bitcoin-Friendly Square Cash App Stock Price Up 56% In 2019 (#GotBitcoin?)
Safeway Shoppers Can Now Get Bitcoin Back As Change At 894 US Stores (#GotBitcoin?)
TD Ameritrade CEO: There’s ‘Heightened Interest Again’ With Bitcoin (#GotBitcoin?)
Venezuela Sets New Bitcoin Volume Record Thanks To 10,000,000% Inflation (#GotBitcoin?)
Newegg Adds Bitcoin Payment Option To 73 More Countries (#GotBitcoin?)
China’s Schizophrenic Relationship With Bitcoin (#GotBitcoin?)
More Companies Build Products Around Crypto Hardware Wallets (#GotBitcoin?)
Bakkt Is Scheduled To Start Testing Its Bitcoin Futures Contracts Today (#GotBitcoin?)
Bitcoin Network Now 8 Times More Powerful Than It Was At $20K Price (#GotBitcoin?)
Crypto Exchange BitMEX Under Investigation By CFTC: Bloomberg (#GotBitcoin?)
“Bitcoin An ‘Unstoppable Force,” Says US Congressman At Crypto Hearing (#GotBitcoin?)
Bitcoin Network Is Moving $3 Billion Daily, Up 210% Since April (#GotBitcoin?)
Cryptocurrency Startups Get Partial Green Light From Washington
Fundstrat’s Tom Lee: Bitcoin Pullback Is Healthy, Fewer Searches Аre Good (#GotBitcoin?)
Bitcoin Lightning Nodes Are Snatching Funds From Bad Actors (#GotBitcoin?)
The Provident Bank Now Offers Deposit Services For Crypto-Related Entities (#GotBitcoin?)
Bitcoin Could Help Stop News Censorship From Space (#GotBitcoin?)
US Sanctions On Iran Crypto Mining — Inevitable Or Impossible? (#GotBitcoin?)
US Lawmaker Reintroduces ‘Safe Harbor’ Crypto Tax Bill In Congress (#GotBitcoin?)
EU Central Bank Won’t Add Bitcoin To Reserves — Says It’s Not A Currency (#GotBitcoin?)
The Miami Dolphins Now Accept Bitcoin And Litecoin Crypt-Currency Payments (#GotBitcoin?)
Trump Bashes Bitcoin And Alt-Right Is Mad As Hell (#GotBitcoin?)
Goldman Sachs Ramps Up Development Of New Secret Crypto Project (#GotBitcoin?)
Blockchain And AI Bond, Explained (#GotBitcoin?)
Grayscale Bitcoin Trust Outperformed Indexes In First Half Of 2019 (#GotBitcoin?)
XRP Is The Worst Performing Major Crypto Of 2019 (GotBitcoin?)
Bitcoin Back Near $12K As BTC Shorters Lose $44 Million In One Morning (#GotBitcoin?)
As Deutsche Bank Axes 18K Jobs, Bitcoin Offers A ‘Plan ฿”: VanEck Exec (#GotBitcoin?)
Argentina Drives Global LocalBitcoins Volume To Highest Since November (#GotBitcoin?)
‘I Would Buy’ Bitcoin If Growth Continues — Investment Legend Mobius (#GotBitcoin?)
Lawmakers Push For New Bitcoin Rules (#GotBitcoin?)
Facebook’s Libra Is Bad For African Americans (#GotBitcoin?)
Crypto Firm Charity Announces Alliance To Support Feminine Health (#GotBitcoin?)
Canadian Startup Wants To Upgrade Millions Of ATMs To Sell Bitcoin (#GotBitcoin?)
Trump Says US ‘Should Match’ China’s Money Printing Game (#GotBitcoin?)
Casa Launches Lightning Node Mobile App For Bitcoin Newbies (#GotBitcoin?)
Bitcoin Rally Fuels Market In Crypto Derivatives (#GotBitcoin?)
World’s First Zero-Fiat ‘Bitcoin Bond’ Now Available On Bloomberg Terminal (#GotBitcoin?)
Buying Bitcoin Has Been Profitable 98.2% Of The Days Since Creation (#GotBitcoin?)
Another Crypto Exchange Receives License For Crypto Futures
From ‘Ponzi’ To ‘We’re Working On It’ — BIS Chief Reverses Stance On Crypto (#GotBitcoin?)
These Are The Cities Googling ‘Bitcoin’ As Interest Hits 17-Month High (#GotBitcoin?)
Venezuelan Explains How Bitcoin Saves His Family (#GotBitcoin?)
Quantum Computing Vs. Blockchain: Impact On Cryptography
This Fund Is Riding Bitcoin To Top (#GotBitcoin?)
Bitcoin’s Surge Leaves Smaller Digital Currencies In The Dust (#GotBitcoin?)
Bitcoin Exchange Hits $1 Trillion In Trading Volume (#GotBitcoin?)
Bitcoin Breaks $200 Billion Market Cap For The First Time In 17 Months (#GotBitcoin?)
You Can Now Make State Tax Payments In Bitcoin (#GotBitcoin?)
Religious Organizations Make Ideal Places To Mine Bitcoin (#GotBitcoin?)
Goldman Sacs And JP Morgan Chase Finally Concede To Crypto-Currencies (#GotBitcoin?)
Bitcoin Heading For Fifth Month Of Gains Despite Price Correction (#GotBitcoin?)
Breez Reveals Lightning-Powered Bitcoin Payments App For IPhone (#GotBitcoin?)
Big Four Auditing Firm PwC Releases Cryptocurrency Auditing Software (#GotBitcoin?)
Amazon-Owned Twitch Quietly Brings Back Bitcoin Payments (#GotBitcoin?)
JPMorgan Will Pilot ‘JPM Coin’ Stablecoin By End Of 2019: Report (#GotBitcoin?)
Is There A Big Short In Bitcoin? (#GotBitcoin?)
Coinbase Hit With Outage As Bitcoin Price Drops $1.8K In 15 Minutes
Samourai Wallet Releases Privacy-Enhancing CoinJoin Feature (#GotBitcoin?)
There Are Now More Than 5,000 Bitcoin ATMs Around The World (#GotBitcoin?)
You Can Now Get Bitcoin Rewards When Booking At Hotels.Com (#GotBitcoin?)
North America’s Largest Solar Bitcoin Mining Farm Coming To California (#GotBitcoin?)
Bitcoin On Track For Best Second Quarter Price Gain On Record (#GotBitcoin?)
Bitcoin Hash Rate Climbs To New Record High Boosting Network Security (#GotBitcoin?)
Bitcoin Exceeds 1Million Active Addresses While Coinbase Custodies $1.3B In Assets
Why Bitcoin’s Price Suddenly Surged Back $5K (#GotBitcoin?)
Zebpay Becomes First Exchange To Add Lightning Payments For All Users (#GotBitcoin?)
Coinbase’s New Customer Incentive: Interest Payments, With A Crypto Twist (#GotBitcoin?)
The Best Bitcoin Debit (Cashback) Cards Of 2019 (#GotBitcoin?)
Real Estate Brokerages Now Accepting Bitcoin (#GotBitcoin?)
Ernst & Young Introduces Tax Tool For Reporting Cryptocurrencies (#GotBitcoin?)
Recession Is Looming, or Not. Here’s How To Know (#GotBitcoin?)
How Will Bitcoin Behave During A Recession? (#GotBitcoin?)
Many U.S. Financial Officers Think a Recession Will Hit Next Year (#GotBitcoin?)
Definite Signs of An Imminent Recession (#GotBitcoin?)
What A Recession Could Mean for Women’s Unemployment (#GotBitcoin?)
Investors Run Out of Options As Bitcoin, Stocks, Bonds, Oil Cave To Recession Fears (#GotBitcoin?)
Goldman Is Looking To Reduce “Marcus” Lending Goal On Credit (Recession) Caution (#GotBitcoin?)

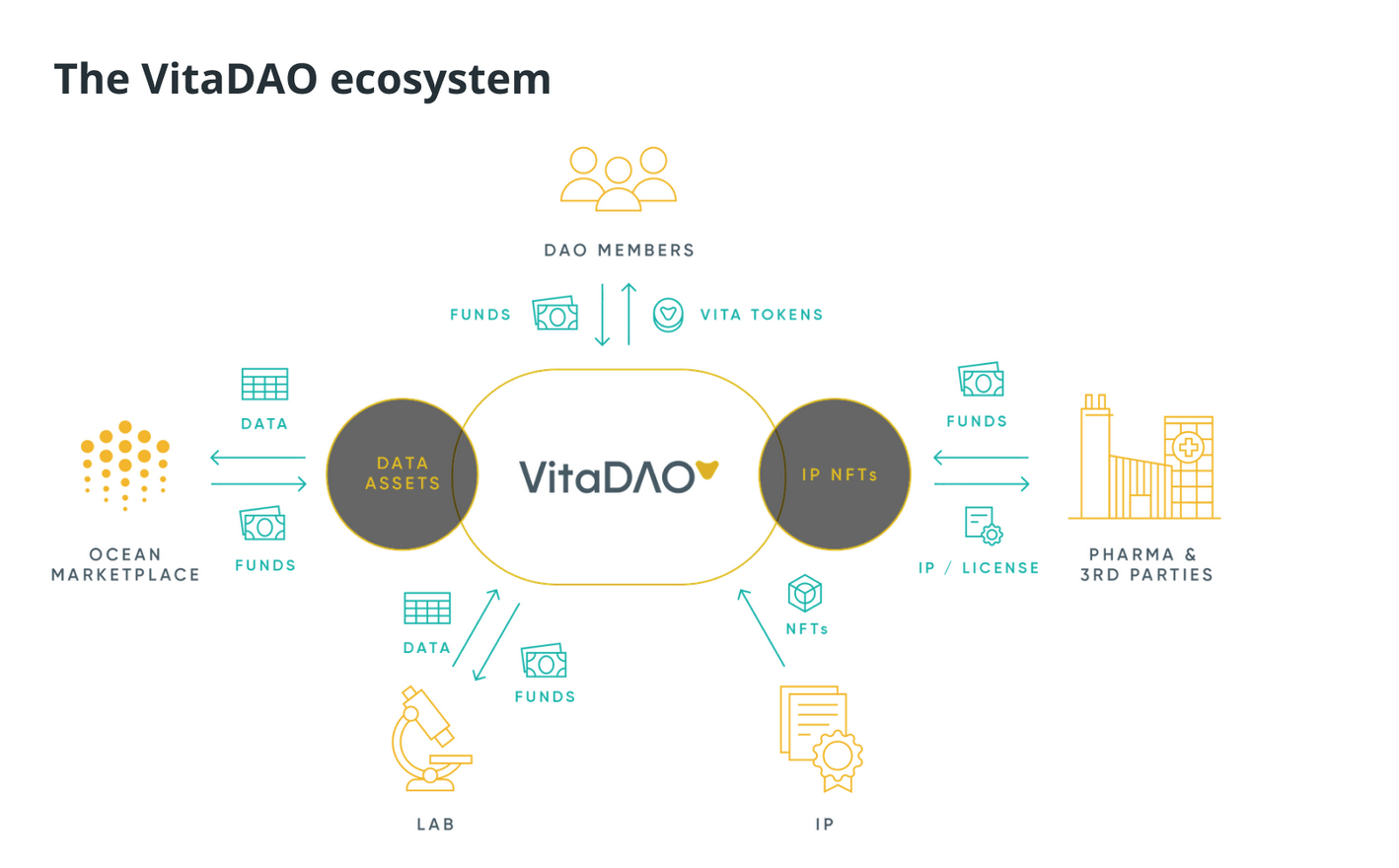
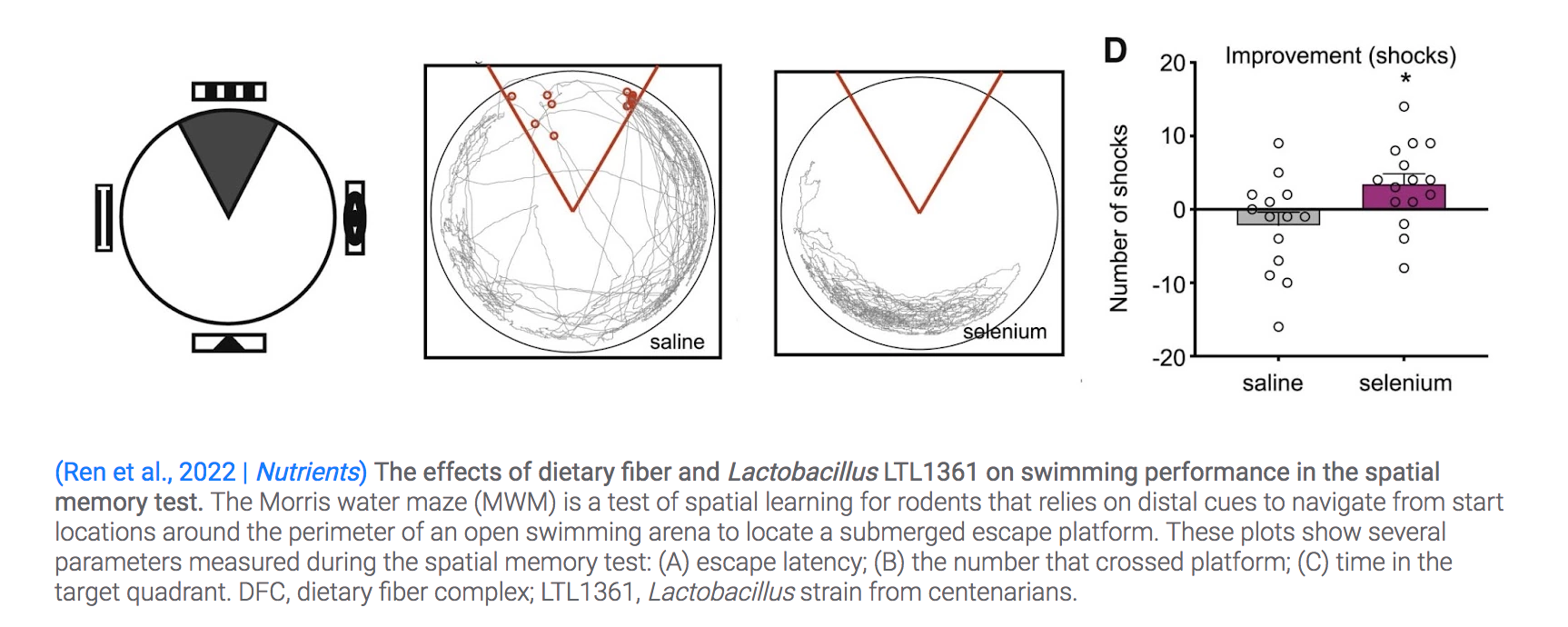



































Leave a Reply
You must be logged in to post a comment.