Freedom of Information Act Document Reveals Antidepressant Drugs Are Less Effective Than Placebos
Freedom of Information Act Document Reveals Antidepressant Drugs Are Less Effective Than Placebos
Janis Schonfeld recalls the events that started her on her recovery from 30 years of depression with snapshot clarity: the newspaper ad she saw in 1997 seeking subjects for an antidepressant study; the chair she was sitting in when she called UCLA’s Neuropsychiatric Institute; the window she was looking out of when she first spoke with Michelle Abrams, the research nurse who shepherded her through the trial.
Related Articles:
Opiate-Addicted Babies Are The New Crack Babies of 2025-26
Fentanyl’s New Foe: A Quick Test Strip That Can Prevent Overdoses
Pharmaceuticals Found In Our Drinking Water!
What Do You Get When Legal Drug Dealers Peddle “Heroin-in-a-Pill” To It’s “Clientele”?
Short Sellers Bet On Opioid Fallout Sinking Drug Companies’ Stocks
She remembers being both nervous and hopeful when she arrived at the institute, and a little uncomfortable when a technician put gel on her head, attached a nylon cap shot through with electrodes, and recorded her brain activity for 45 minutes. But most of all she remembers getting the bottle of her new pills in a brown paper bag from the hospital pharmacy. “I was so excited,” she told me. “I couldn’t wait to get started on them.”
Within a couple of weeks, Schonfeld, then a 46-year-old interior designer, got quickly and dramatically better, able once again to care for herself and her husband and daughter, no longer so convinced of her own worthlessness that she’d consider killing herself. For the next two months, she came back weekly for more interviews and tests and EEGs. And by the end of the study, Schonfeld seemed to be yet another person who owed a nearly miraculous recovery to the new generation of antidepressants — in this case, venlafaxine, better known as Effexor.
But during her final visit to the institute, one of the doctors directing the research sat her down to deliver some disturbing news. “He told me I hadn’t been taking a medicine at all. I’d been on a placebo. I was totally shocked.” So was nurse Abrams. Both women knew that half the test subjects were getting placebos and that Schonfeld might be among them.
But not only was she feeling better — she’d even experienced nausea, a side effect commonly associated with Effexor, so they had each assumed that she was in the drug group. Schonfeld was so certain of this that at first she didn’t believe the doctor.
“I said to him, ‘Are you sure? Check those records again.'” But there was no doubt. The brown bag contained nothing but sugar pills. Which didn’t mean, he was quick to add, that she was making anything up, but only that her improvement couldn’t possibly be due to the pharmacological effects of the pills.
Schonfeld’s experience is hardly unique, although you wouldn’t know it from the ubiquitous advertisements for antidepressants — nor, if you were a doctor, would you know just how common it is from reading the medical journals. Psychiatrists and other mental-health professionals (I am a practicing therapist) know that any given antidepressant has only about a 50 percent chance of working with any given person.
But what most people — patients and clinicians alike — don’t know is that in more than half of the 47 trials used by the Food and Drug Administration to approve the six leading antidepressants on the market, the drugs failed to outperform sugar pills, and in the trials that were successful, the advantage of drugs over placebo was slight.
As it would hardly help drug sales, pharmaceutical companies don’t publish unsuccessful trials, so University of Connecticut psychology professor Irving Kirsch and his co-authors used the Freedom of Information Act to extract the data from the FDA.
What they found has led them, and other researchers who’ve investigated antidepressants’ relatively poor showing against placebos, to conclude that millions of people may be spending billions of dollars on medicines that owe their popularity as much to clever marketing as to chemistry, and suffering serious side effects — not to mention becoming dependent on drugs for healing they might be able to do without them — in the bargain.
But many doctors remain convinced that antidepressants do work, that the flaw lies not in the capsules themselves but in the studies used to evaluate them. Clinical trials can consume half a drug’s patent life.
And so pressure to bring the medicine to market leads researchers to adopt strategies — such as recruiting people whose depression is too mild to yield powerful results — better suited to clearing regulatory hurdles than generating useful scientific knowledge. That, and not the power of suggestion, is why antidepressants barely outperform placebos, these scientists say.
While some of this debate breaks down along familiar lines — psychologists resisting the tendency to reduce all mental suffering to biology versus psychiatrists more comfortable with matter than spirit — no one disputes that the statistics about antidepressant efficacy are dismal, and that they do little to clarify the question of whether people who get better on antidepressants do so because they are taking Prozac or Zoloft or because they are taking a pill — any pill.
BEFORE SCIENCE TOOK OVER the healing arts and focused physicians’ attention on biological causes of disease, mystics and alchemists and flimflam artists alike offered potions and powders to the ailing. Some of these remedies were bizarre, like usnea — the moss from the skull of a hanged man, used to treat nervous illness — and others merely fanciful, like powdered unicorn horn.
Some were truly dangerous, like calomel, a mercury-based laxative that may have hastened George Washington’s death from the cold he famously caught while riding on a rainy night.
Some — notably cinchona bark, the source of quinine — turned out to have actual healing powers, but there were so few of these that in 1860 Oliver Wendell Holmes, the doctor who fathered a Supreme Court justice, wrote, “If the whole materia medica could be sunk to the bottom of the sea, it would be all the better for mankind and all the worse for the fishes.”
But Holmes was not entirely correct. Despite their lack of specific healing properties, many ancient medicines worked — or at least people often got better after taking them, as they still do. Most illnesses remit as part of their natural course, but the placebo effect occurs far too frequently to be mere coincidence. No one really understands why, but doing something for an illness — especially if that something involves a pill — is usually better than doing nothing at all.
There’s no money to be made in sugar pills, so drug companies, which fund much of the drug research in the United States, have not looked very hard into this question. But placebos do figure prominently in their studies — as a stalking-horse for the potential new medications. Because any drug may well be acting as a placebo, it is not a sufficient test simply to give a new compound to sick people to see if they get better.
To rule out the possibility that patients are recovering because of faith or a good sales pitch, and to ensure that the drug works by virtue of its biochemical properties, the FDA has, since the late 1970s, required that all drugs be tested against placebos.
Typically, between 35 and 45 percent of people given placebos improve. If a candidate drug outperforms a placebo in two independent studies, and if it does so without untoward side effects, the FDA will approve it for use.
The FDA does not consider, however, the relative advantage that new drugs show over placebo. So long as the difference is statistically significant — meaning that the results are not merely random — a drug can be advertised as “safe and effective” whether clinical trials proved it to be 5 percent or 50 percent or 500 percent more effective than an inert pill.
In the case of the Prozac generation of antidepressants, marketing efforts have paid off wildly.
Some 92 million prescriptions were written for the top six antidepressants in 2002, a ubiquity that has, far more than any research, helped to bolster the theory that depression is the result of a biochemical imbalance that the drugs cure — a theory that has not been proved, despite more than 40 years of trying.
But critics, psychologists and psychiatrists alike, have been suspicious of the drugs since they were introduced, and it turns out they have some striking data on their side. “In the early ’90s, many of our psychiatric colleagues felt that patients did not do as wonderfully as all these reports of ‘magic pills’ would suggest,” recalled psychologist Roger Greenberg, a professor at the State University of New York’s Upstate Medical University.
“So we went back to the literature.” Greenberg [no relation to the author] and his team analyzed all the data from Prozac’s clinical trials that had been published. They determined that the new drug showed negligible advantage over earlier antidepressants and that two-thirds of the patients would do as well or better with placebos.
Greenberg started with material hidden in the plain light of professional journals, but a bit of detective work by Irving Kirsch and his research team has turned up even more disturbing evidence about the low rates of antidepressant effectiveness. Kirsch is a soft-spoken and slight man who has spent more than 30 years studying the placebo effect.
He has a native suspicion of biological explanations of depression and sees in the placebo effect the potential for self-healing without resorting to expensive and possibly dangerous drugs.
While many researchers duplicated and refined Greenberg’s initial findings, Kirsch knew that there was a body of results that no one was looking at. Manufacturers don’t have to publish all their data in journals, but they do have to report every trial to the FDA.
“This was all so controversial,” he told me. “And the defenders claimed that our data didn’t tell the whole story. So we figured, why not use the Freedom of Information Act to investigate?”
Kirsch requested the complete files on the six most widely prescribed antidepressants approved between 1987 and 1999: Prozac, Zoloft, Paxil, Effexor, Serzone, and Celexa — drugs that together had $8.3 billion in worldwide sales in 2002. Within a month, he had an even less drug-friendly story than the one told in the journals.
In “The Emperor’s New Drugs,” published in the July 2002 issue of the American Psychological Association’s Prevention & Treatment, Kirsch’s team presented their findings: Of the 47 trials conducted for the six drugs, only 20 of them showed any measurable advantage of drugs over placebos, a much lower number than turns up in published research.
This was not entirely unexpected — “publication bias” has long been known to be a problem in assessing the effectiveness of drugs — and Kirsch is quick to point out that even these meager numbers “leave no doubt that there is a difference between drug and placebo.
But I was surprised at how small the difference was in clinical terms. The studies all used the same measure” — the Hamilton Depression Rating Scale, the nearly universal way clinicians assess a patient’s level of depression — “so it was easy to see how much clinical improvement there really was.”
And there really wasn’t much at all: The average patient on drugs improved by about 10 points on the 52-point Hamilton, while a placebo patient improved by a little more than eight. “A two-point difference on the Hamilton — it’s just clinically meaningless. Trivial,” Kirsch says.
“You can get that from having an improvement in sleep patterns, and if one of the side effects of the drugs is to induce drowsiness, the whole difference could be right there.” (Indeed, critics say the Hamilton is skewed toward physical symptoms of depression, those most likely to be affected by medication.)
Kirsch received copies of memos indicating that regulators had, in at least one case, raised questions about clinical significance. In 1998, Paul Leber, then director of the FDA’s Division of Neuropharmacological Drug Products, wrote of Celexa, “There is clear evidence from more than one adequate and well-controlled clinical investigation that [Celexa] exerts an antidepressant effect.
The size of that effect, and more importantly, the clinical value of that effect is not something that can be validly measured, at least not in the kind of experiments conducted.”
A deputy agreed: “It is difficult to judge the clinical significance of this difference,” he wrote, but added that this shouldn’t be an impediment for bringing Celexa to market because “similar findings for…other recently approved antidepressants have been considered sufficient.”
Kirsch argues that by the FDA’s own logic, it’s not even clear if the drugs’ small advantage is truly pharmacological. In trials, every drug response is assumed to be partially a placebo response, and the drug effect is only the additional benefit — in the case of the antidepressant studies, less than two points out of ten, or 20 percent of the overall improvement.
This means, he said, that “80 percent of the drug effect is the placebo effect.” And even the remaining 20 percent could be due to placebo effects enhanced by the drugs’ side effects, amplified by the way the trials are conducted.
“A person is brought into a clinical trial and told, ‘You may be getting placebo or drug. The real drug has the following side effects.’ Put yourself in this position. You’re certainly curious about what you’re getting. And you want to get better. You notice that your mouth is getting dry, which is one of the side effects they told you about, and that leads you to conclude that you’ve been assigned to the drug condition.
Presumably, a placebo works by affecting a person’s expectancy about what is going to happen. If you know you’ve been assigned to the drug condition, you may have a stronger placebo effect because you’re now more convinced that you’re getting something that’s going to help you.”
Greenberg’s research shows that both patients and raters in clinical trials often “break the blind” by guessing which condition they have been assigned and that the most powerful drug effects are reported when this occurs.
The guesses don’t even have to be accurate. Janis Schonfeld experienced side effects on placebo, and this was part of what led her (and nurse Abrams, who was scoring the Hamilton) to assume she was on drugs. According to Kirsch’s theory, Schonfeld’s strong response (and Abrams’ rating of her progress) may have come about because they thought — due to symptoms caused by the power of suggestion — that she was on the drug.
Kirsch thinks it is possible to test his theory, but only with a radical redesign of the method used to validate drugs. Instead of two groups, a study would have four. Researchers would tell two groups of patients they were getting placebo and the other two that they were being given the drugs. But only half the patients would be told the truth.
And the placebo would be a nonpsychoactive substance designed to mimic at least some of the side effects of the real drug. This way researchers could look directly at the role of suggestion in response to both placebo and drug. It is, however, currently considered unethical to deceive patients in this fashion.
But there is plenty of indirect evidence for Kirsch’s position, including a peculiar recent finding: Both placebo response and drug response for antidepressants have steadily increased over time, so much so that the best predictor of whether research shows positive results is the year the study was published.
This result has yet to be explained, but Kirsch thinks it indicates the way the wide- spread publicity about antidepressants shapes patients’ expectations.
“It suggests that over time the drugs have gotten more potent for reasons other than chemistry. I would suspect that it’s because of increased marketing.” Kirsch explains the way that marketing can capitalize on a central mechanism of depression: “The hopelessness of depression is the expectancy that a terrible state of affairs is not going to get better.
Now you give somebody a treatment that’s been touted as the cure for the worst thing in their lives. What that does is to instill a hope, which is the opposite of depression.” Kirsch’s theory leads to an unsettling conclusion: Drug companies may have marketed their antidepressants beyond what statistics justify, but the barrage of advertising may also have inadvertently amplified the placebo effect and thus increased the effectiveness of the drugs they are selling.
WHEN I FILL OUT A treatment report explaining to an insurance company why they ought to pay for someone’s therapy, I am asked for a diagnosis.
If the patient is depressed and not on antidepressants, I often must explain why not. Were it not for these bureaucratic demands — and for all the miracle-drug testimony found in advertising and casual talk — the FDA statistics would hardly be surprising or disturbing, because, like many clinicians, I have come to see that the effects of Prozac and its cousins are just about as pallid as those numbers would predict: The drugs are not panaceas, not solid evidence that depression is a chemical imbalance, but have proved to be moderately useful for some people (and moderately harmful to others).
No scientist doubts the existence of the disconnect between the data and the way antidepressants are perceived and used, but Kirsch’s theory about it is far from the industry standard.
Indeed, some simply dismiss it out of hand — like Donald Klein, a renowned psychiatry professor at Columbia University’s New York State Psychiatric Institute, who thinks that Kirsch’s work is so biased against antidepressants that, though asked, he declined to be among the respondents to “The Emperor’s New Drugs” — “for the same reason,” he told me, “that I don’t argue with creationists.”
Klein, who has conducted antidepressant trials for pharmaceutical companies, acknowledges that the data can leave the impression that the drugs don’t work very well. But he is among those who think this says more about the trials than the drugs.
According to Klein, the FDA standard — two successful trials without untoward side effects — won’t elicit a full body of knowledge about new drugs, and may even limit what the tests can tell us.
“The job of the pharmaceutical company is to get FDA approval,” he says. “So you want to go in with a dose which is effective but doesn’t create side effects. It’s a real problem.
Drugs are not being tested for their optimum efficacy.” Nor, given this strategy, are they tested for their maximum side effects — which may be why reports of agitation and suicidal impulses in excess of what the trials found have dogged the Prozac generation of antidepressants since they were introduced.
Clinical trials can become a game for drug companies to win rather than a venue for generating scientific knowledge. And it’s a game that establishes perverse incentives, in part because drugs’ limited patent lives — usually 20 years — begin before clinical trials, which can take a decade, start.
“We’re talking real money here,” says Klein, noting it takes between $300 to $500 million to develop a new drug. Klein told me that within the industry the clinical trial period is thought to cost “a million dollars a day. That adds some pressure for finishing trials fast.”
Despite the bottom-line approach, “there are lots and lots of compounds that get evaluated and never approved,” notes Lawrence Price, a psychiatrist who directs research at Brown University’s Butler Hospital. A more nuanced criterion for a successful trial is possible, but, says Price, “it would just take forever. It’s not that there aren’t important questions, but you would get so bogged down in trying to nail down the details that you would just never make any progress with newer compounds.”
You also might not make any progress if you waited around for severely depressed people to test drugs on. “The problem with antidepressant studies,” according to Klein, “is that anything that can be confused with ordinary unhappiness gets in” — which means that subjects in clinical trials are insufficiently depressed, too close to normal to show dramatic improvement.
Price, who has conducted clinical trials of antidepressants for 25 years, points out that recruitment techniques like the one that attracted Janis Schonfeld to UCLA can lead to a skewed sample. “If you go out and advertise in the newspaper for depressed people,” says Price, “you are going to get less ill people than if you are taking people who are brought in via the emergency room.”
Relatively high-functioning, moderately depressed people, those most likely to enroll in and finish a trial, are, as it happens, more likely to register a high placebo response. There are no biochemical markers of depression, no blood test or X-ray that confirms its presence, so it can be judged only by its appearance — which means, in trials, by the Hamilton, a test of subjective states scored by clinicians whose employers are paid up to $10,000 for each patient who completes a study.
“If the investigator has directed his/her research assistant to rate liberally on the Hamilton,” says Price, “then you are going to have more people meeting the entry criterion,” typically, at least 17 points — the line dividing mildly and moderately depressed. (One of Price’s colleagues estimates that Hamilton scores are inflated by up to five points for clinical trials.)
The drug companies, of course, want more than speedy trials. They want successful ones. “Placebo is a killer for them,” Price explains, “because if they spend $40 million on a trial and get a placebo response rate of 50 percent, then they’ve just wasted that $40 million. There’s a huge interest in trying to address the high placebo response rate in depression. How can it be lowered? How can you identify the sample of people in whom these compounds are really going to work?”
The study in which Janis Schonfeld enrolled may provide some answers to these questions — although somewhat inadvertently.
Hoping to eliminate the trial-and-error method used to match patients with antidepressants, the UCLA doctors were using electroencephalograms to determine if there was some neurochemical difference between the brains of people who respond to Effexor and those who respond to Prozac. The researchers found the differences they were looking for, but they also got a surprise.
The EEGs of placebo responders were different from those of the drug responders, and similar to each other, a phenomenon that had never before been observed and that may be the first step to identifying the neurochemistry of the placebo response. This was welcome news to the drug companies, who’d like nothing more than to eliminate placebo responders from their studies.
Take away the people most likely to show a strong placebo effect, include the people most likely to respond to a drug, and the statistics become more favorable for the manufacturers and provide less ammunition for critics like Greenberg and Kirsch. Psychologist David Antonuccio, a professor at the University of Nevada, claims that the deck is already stacked.
In addition to publication bias, inflated Hamilton scores, and broken blinds, he points to the placebo washout period that starts every clinical trial: All patients are given a week of placebo treatment, and the strongest responders are eliminated from the study.
The idea, of course, is to get a more accurate estimate of the true drug effect, but “if you put everybody on an antidepressant and washed out everyone who responds, people would say, ‘That’s a very biased strategy against the drugs.’ Well, I believe we have a strategy here that’s biased against the placebo condition.”
AFTER JANIS SCHONFELD was debriefed, she was given her reward for participating in the study: a one-year supply of Effexor. She didn’t consider not taking the drug. “They told me that I’d gotten a good start, that if I’d done well on placebo, I’d probably do better on the drug.” And so she did. “After about a week or maybe two weeks it was like a fog was lifted from my eyes.
I realized I had spent much of the last 20 years in that fog.” Schonfeld took Effexor for two and a half years and then “one day I just thought, ‘You know, I don’t think I need this medication anymore.’ I spent three weeks weaning off of it. That was about a year and a half ago, and I haven’t really felt that I needed it since.” She emphatically rules out the possibility that her improvement was a result of placebo effects, amplified or otherwise.
To Kirsch, Schonfeld’s is a case of lost opportunity. “Why not say to her, ‘You did this’? People respond the way they were expecting to respond, so why not work on that expectation?
Why not teach her the strategies that she can use to make herself feel better?” Antonuccio says, “Placebo is a valid intervention in and of itself,” adding that people like Schonfeld have ample contact with trained staff during trials, which may itself be what accounts for the high placebo rates.
“It’s possible that psychological treatments are mostly placebo as well,” he says — not, as he is quick to add, that there’s any- thing wrong with that. “We just ought not to see the placebo effect as some sort of inferior response or condition.”
But even though, as Kirsch notes, “more placebos have been administered to research participants than any single experimental drug,” they remain poorly understood and used, for the most part, only inadvertently and haphazardly. The discovery of biological underpinnings of the placebo effect may change this, as drug researchers grasp the potential of turning yet another neurochemical pathway into a pharmaceutical market by developing a placebo drug.
Bizarre as this sounds, it may be the only incentive that will lead a profit-driven health care industry toward an understanding of humanity’s oldest means of healing.
Updated: 7-29-2025
New Opioid, 100 Times Stronger Than Fentanyl!!
LONDON—Fentanyl fueled the worst drug crisis the West has ever seen. Now, an even more dangerous drug is wreaking havoc faster than authorities can keep up.
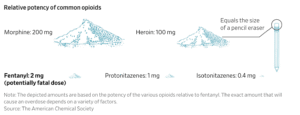
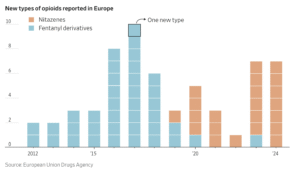
The looming danger is an emerging wave of highly potent synthetic opioids called nitazenes, which often pack a far stronger punch than fentanyl.
Nitazenes have already killed hundreds of people in Europe and left law enforcement and scientists scrambling to detect them in the drug supply and curb their spread.
The opioids, most of which originate in China, are so strong that even trace amounts can trigger a fatal overdose. They have been found mixed into heroin and recreational drugs, counterfeit painkillers and antianxiety medication. Their enormous risk is only dawning on authorities.
Europe, which has skirted the kind of opioid pandemic plaguing the U.S., is now on the front line as nitazenes push into big heroin and opioid markets such as Britain and the Baltic states.
At least 400 people died in the U.K. from overdoses involving nitazenes over 18 months until January of this year, according to the government.
“This is probably the biggest public health crisis for people who use drugs in the U.K. since the AIDS crisis in the 1980s,” said Vicki Markiewicz, executive director for Change Grow Live, a leading treatment provider for drugs and alcohol in the U.K. Particularly worrying, she said, is that most people take nitazenes unwittingly, as contaminants in other drugs.
The U.K.’s National Crime Agency has warned that partly due to nitazenes, “there has never been a more dangerous time to take drugs.”
In the U.S., where fentanyl dominated the opioid market, nitazenes had as of last year been found in at least 4,300 drug seizures since 2019, usually in fentanyl mixtures, and have led to dozens of deaths.
The Drug Enforcement Administration has warned that Mexican cartels could use their existing relations with China-based suppliers to obtain nitazenes and funnel them into America.
The most common street nitazenes are roughly 50 to 250 times as potent as heroin, or up to five times the strength of fentanyl.
They are likely much more prevalent than official statistics suggest, due to limited testing. Authorities say official death tolls are almost certainly undercounts.
On an early summer morning in 2023, police arrived at Anne Jacques’s door in north Wales. Her 23-year-old son had died in his sleep in his student apartment in London, they told her.
Her son, Alex Harpum, was a rising opera singer and healthy. Police found Xanax tablets in his room, and evidence on his phone that he had bought pills illegally, which Jacques said he occasionally did to sleep while on medication for his attention-deficit hyperactivity disorder.
Yet, the coroner established the cause of death as unexplained cardiac arrest, known as sudden adult death syndrome. Jacques, not satisfied with the explanation, researched drug contaminants and requested the coroner test for nitazenes.
Seven months after her son’s death, police confirmed that his tablets had been contaminated with the potent opioid.
“I basically had to investigate my own son’s death,” Jacques said. “You feel like your child has been murdered.”
Harpum wasn’t alone. While most known overdoses affect heroin users, nitazenes have also been found in party drugs like cocaine, ketamine and ecstasy, in illegal nasal sprays and vapes, and detected in benzodiazepines like Xanax and Valium.
In May, two young Londoners died after taking what authorities believe was oxycodone laced with nitazenes upon returning home from a nightclub.
Dealers aren’t trying to kill customers, but the globalized drug trade leads gangs to traffic a wider variety of increasingly potent substances, partly because smuggling gets easier as the volumes involved shrink.
U.K. National Crime Agency Deputy Director Charles Yates said dealers are driven mainly by greed. “They buy potent nitazenes cheaply and mix them with bulking agents such as caffeine and paracetamol to strengthen the product being sold and make significant profits,” he said.
Nitazenes are also ravaging West Africa as a prevalent ingredient in kush, a synthetic drug that has killed thousands of people and led Sierra Leone and Liberia to declare national emergencies.
“It’s an international concern. They have been detected on every continent,” said Adam Holland, an expert on synthetic opioids at the University of Bristol.
“You can produce them with different chemicals that are relatively easy to get a hold of, and you can do it in an underground laboratory. And because they’re so potent, you need less for the same size of market so they’re easier to smuggle.”
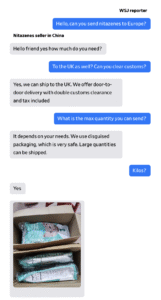
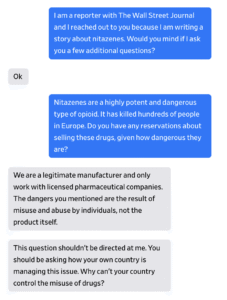
Chinese suppliers sell nitazenes openly on online marketplaces sometimes using photos of young women as their profile picture. They list phone numbers, social-media handles and business addresses linked to China or Hong Kong.
The drugs are sometimes labeled as research chemicals but also often explicitly as nitazenes.
Four suppliers told a Journal reporter they could send any quantity to Europe, including the U.K., and promised they could evade customs.
A spokesperson for TradeKey said the company has a “zero-tolerance policy toward the listing or sale of any controlled substances, including synthetic opioids such as nitazenes.” It said it had added various types of nitazenes to its banned products registry and blocked hundreds of accounts seen to violate its compliance rules.
On “rare occasions,” a prohibited product may pass initial approvals and get listed, but the company worked to routinely clean the site, it said.
“We take this issue very seriously and are fully committed to ensuring our platform is not misused in any way. We also cooperate with regulatory and law enforcement bodies as needed,” the spokesperson said.
Nitazenes were never approved for medical use in Europe. Developed in the 1950s, they were found in trials to cause fatal breathing problems.
They were detected sporadically over the years: in a lab in Germany in 1987; in 1998 in Moscow, where they were linked to a dozen deaths; and in 2003 in Utah, where a chemist manufactured them apparently for personal use.
Nitazenes appeared in drug seizures in Europe and the U.S. beginning in 2019, and began spreading quickly in Europe in 2023, their high potency leaving a trail of fatal overdoses even among seasoned drug users.
In Scotland, whose population of 5.5 million has the highest overdose death rate per capita in Europe, nitazenes have been involved in 150 to 200 drug-related deaths in the past two years alone, said Austin Smith, head of policy with the Scottish Drug Forum charity.
“Imagine mixing salt in sand on a beach, it’s impossible to do that evenly,” he said.
Europe’s medical practices have protected it from fentanyl, which first took off in the U.S. in the 1990s due to private prescriptions and aggressive marketing.
However, Europe is vulnerable to opioids in ways that echo the American experience. The second big boost in fentanyl usage in the U.S. came in the 2010s, when drug cartels began adulterating the heroin supply with fentanyl.
So far, nitazenes appear to be supplied by individual brokers and sellers, but Europe is rife with international drug gangs that could turn to nitazenes.
“Synthetic opioids in the U.S. have not been driven by demand, they have been driven wholesale by supply,” said Vanda Felbab-Brown, senior fellow and expert on the global opioid trade with the Brookings Institution, a think tank.
“If large criminal groups such as Albanian mafia groups, Turkish criminal groups or Italian or Mexican groups get into supplying nitazenes to Europe on a large scale, we can anticipate a massive public healthcare catastrophe.”
They may be prompted to do so. Since the Afghan Taliban most recently banned in 2022 the cultivation of poppies, which supplied about 90% of the world’s heroin, experts have warned that a heroin shortage could lead gangs to cut the drug with other, more dangerous substances. Nitazenes are at the top of the list.
“If the heroin supply is interrupted, that will have a knock-on effect on drug use within Europe, and on things users can turn to in the absence of heroin, such as synthetic opioids and synthetic crystal meth,” said Andrew Cunningham, expert on drug markets with the European Union Drugs Agency.
The tiny nation of Estonia has firsthand experience of what that is like. When the Taliban first banned poppy cultivation in 2000, fentanyl flooded the Estonian drug market as a replacement for heroin.
Drug-related deaths grew fourfold in two years, and put the Baltic country in a fentanyl grip that it was unable to shake.
For a decade, from 2007 to 2017, Estonia had the highest per capita overdose death rate in Europe. And Estonia is already feeling the influx of nitazenes, which since 2023 have been involved in nearly half of all drug-induced deaths in the tiny Baltic nation.
When a batch of drugs contaminated with nitazenes hits the streets, it often results in a cluster of overdoses. Late last year, about 80 people overdosed and needed medical treatment in Dublin over a weekend.
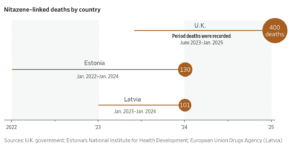
In March of this year, at least 31 users overdosed over a few days in Camden, north London.
One of them, Tina Harris, 41, who has been using heroin since her early teens, said she bought a £5 bag of what she thought was fentanyl from a drug dealer in Camden.
“He told me, ‘be careful because it’s strong.’ I thought he was just chatting sh—,” she said. After smoking the drug, she passed out, and survived only because a friend administered shots of naloxone, an antidote that users carry for emergencies, until the ambulance arrived.
Harris woke up in the hospital, rattling from withdrawal. Since then, she has twice saved the life of friends who mistakenly took nitazenes, by providing naloxone and CPR.
She has become increasingly worried about the drug supply in London, but said her addiction is impossible to kick.
“It’s a devil’s trap,” she said.
Related Articles:
Bitcoin Information & Resources (#GotBitcoin)
The Price Of Parenting. TL;DR, It Takes A Helluva Lot of Money
Ultimate Resource On Mushrooms From Foraging, Medicinal, Cultivation And More
Humans Can Echolocate Like Bats And Dolphins. We Explain How It Works
Our Arctic Vacation Itineraries (2026)
Goodbye Gentle Parenting, Hello ‘F—Around And Find Out’
The Many Health Benefits of Nitric Oxide
How To Eliminate Mouth Breathing In Children And Teenagers
We RFK’d The Fries And Food Dyes
Declining Dollar Hits Amazon Forcing It To Raise Prices
Red Yeast Rice VS Prescription Statin Drugs
European Commission Plans A “Reverse Robinhood” Via Savings And Investments Union (SIU)
Un Día Sin Mexicanos (“A Day Without A Mexican”) Is Today!
Is Perplexity AI A Bloomberg Terminal Killer? Not Even Close!
Compounding Pharmacies Explained: Safety, Regulations And More
Sell Stock In Yourself Or Some Of Your Home Equity For Bitcoin
North Korea Infiltrates U.S. Remote Jobs—With The Help of Everyday Americans
America’s Leading Alien Hunters Depend On AI To Speed Their Search
Bitcoin’s Nouveau Riche Executives And Wealthy Investors In Search Of Ways To Protect Themselves
How Anthocyanins Shield Us From Microplastics
Origins Of Toxic Algae (Domoic Acid) Killing Sea Lions, Birds And Dolphins In Southern California
The Human Brain And It’s Ability To Time-Travel
Wall Street’s New Tariff Safe Haven: Biotech Companies Adopting Bitcoin Treasury Strategy
How Natural “Short Sleepers” Thrive On 4 Hours Of Sleep Per Night Thanks To A Gene Mutation
Nine Unknown Benefits of Sleep Including Penal And Clitoral Erections
Elite Athletes Try A New Training Tactic: More Vitamin D3
Bitcoin Industry On A Winning Streak As Sec. Drops Lawsuit After Lawsuit!!
FOIA Files: Elon Musk’s DOGE Wants A Heads Up On FOIA Requests
When Russia Came To Defend The United States
Ultimate Resource Covering DOGE AKA The Department of Government Efficiency
Your Money Is Currently Lawful. However, That Is Subject To Change #GotBitcoin #BitcoinFixesThis
Is The United Nations Going Bankrupt?
Bitcoin Industry Founders Should Stop Chasing Narratives And Start Creating Them
Consumer Finance Watchdog Finalizes ‘Open Banking’ Rules Aimed At Driving Competition
US Government Crypto Wallets Hacked For $20M — Arkham Intelligence
Coinbase Files FOIAs Against US Regulators Probing Banks’ Bitcoin Crackdown
The Botanist Daring To Ask: What If Plants Have Intelligence?
Theoretical Physicist Sylvester James Gates
FOIA Request Reveals SEC’s Gensler, Lawmakers Target Of Violent Threats
Ultimate Resource For Prediction Markets Covering Politics, Pop Culture, Sports And More
September (AKA Rektember) Is Once Again A Tough Month For Stocks AND Bitcoin
Ultimate Resource For Cloning humans. Biological, Ethical, And Social Considerations
FOIA Reveals Aurora Borealis Imperiled US Infrastructure From Power Grids To Satellites (5-10-2024)
Ultimate Resource Covering How “Silent Payments” Are Bringing New Privacy Protections To Bitcoin
Nostr Allows Bitcoiners To Build-Out A Decentralized, CENSORSHIP-RESISTANT Social-Media!!
Joe Rogan: I Have A Lot Of Hope For Bitcoin
Teen Cyber Prodigy Stumbled Onto Flaw Letting Him Hijack Teslas
Spyware Finally Got Scary Enough To Freak Lawmakers Out—After It Spied On Them
The First Nuclear-Powered Bitcoin Mine Is Here. Maybe It Can Clean Up Energy FUD
The World’s Best Bitcoin Policies: How They Do It In 37 Nations
Tonga To Copy El Salvador’s Bill Making Bitcoin Legal Tender, Says Former MP
Wordle Is The New “Lingo” Turning Fans Into Argumentative Strategy Nerds
Prospering In The Pandemic, Some Feel Financial Guilt And Gratitude
Is Art Therapy The Path To Mental Well-Being?
New York, California, New Jersey, And Alabama Move To Ban ‘Forever Chemicals’ In Firefighting Foam
The Mystery Of The Wasting House-Cats
What Pet Owners Should Know About Chronic Kidney Disease In Dogs And Cats
Pets Score Company Perks As The ‘New Dependents’
Why Is My Cat Rubbing His Face In Ants?
Natural Cure For Hyperthyroidism In Cats Including How To Switch Him/Her To A Raw Food Diet
Ultimate Resource For Cat Lovers
FDA Approves First-Ever Arthritis Pain Management Drug For Cats
Ultimate Resource On Duke of York’s Prince Andrew And His Sex Scandal
Walmart Filings Reveal Plans To Create Cryptocurrency, NFTs
Bitcoin’s Dominance of Crypto Payments Is Starting To Erode
T-Mobile Says Hackers Stole Data On About 37 Million Customers
Jack Dorsey Announces Bitcoin Legal Defense Fund
More Than 100 Millionaires Signed An Open Letter Asking To Be Taxed More Heavily
Federal Regulator Says Credit Unions Can Partner With Bitcoin Providers
What’s Behind The Fascination With Smash-And-Grab Shoplifting?
Train Robberies Are A Problem In Los Angeles, And No One Agrees On How To Stop Them
US Stocks Historically Deliver Strong Gains In Fed Hike Cycles (GotBitcoin)
Ian Alexander Jr., Only Child of Regina King, Dies At Age 26
Amazon Ends Its Charity Donation Program Amazonsmile After Other Cost-Cutting Efforts
BTC Panics, Then Jeers at DOJ Announcement of ‘Major Action’ Against Tiny Chinese Exchange Bitzlato
Indexing Is Coming To BTC Funds Via Decentralized Exchanges
Doctors Show Implicit Bias Towards Black Patients
Darkmail Pushes Privacy Into The Hands Of NSA-Weary Customers
3D Printing Make Anything From Candy Bars To Hand Guns
Stealing The Blood Of The Young May Make You More Youthful
Henrietta Lacks And Her Remarkable Cells Will Finally See Some Payback
AL_A Wins Approval For World’s First Magnetized Fusion Power Plant
Want To Be Rich? Bitcoin’s Limited Supply Cap Means You Only Need 0.01 BTC
Smart Money Is Buying Bitcoin Dip. Stocks, Not So Much
McDonald’s Jumps On Bitcoin Memewagon, Crypto Twitter Responds
America COMPETES Act Would Be Disastrous For Bitcoin Inustry (LOL) And More
Lyn Alden On Bitcoin, Inflation And The Potential Coming Energy Shock
Inflation And A Tale of Cantillionaires
El Salvador Plans Bill To Adopt Bitcoin As Legal Tender
Miami Mayor Says City Employees Should Be Able To Take Their Salaries In Bitcoin
Vast Troves of Classified Info Undermine National Security, Spy Chief Says
BREAKING: Arizona State Senator Introduces Bill To Make Bitcoin Legal Tender
San Francisco’s Historic Surveillance Law May Get Watered Down
How Bitcoin Contributions Funded A $1.4M Solar Installation In Zimbabwe
California Lawmaker Says National Privacy Law Is a Priority
The Pandemic Turbocharged Online Privacy Concerns
How To Protect Your Online Privacy While Working From Home
Researchers Use GPU Fingerprinting To Track Users Online
Japan’s $1 Trillion Bitcoin Industry May Ease Onerous Listing Rules
Ultimate Resource On A Weak / Strong Dollar’s Impact On Bitcoin
Fed Money Printer Goes Into Reverse (Quantitative Tightening): What Does It Mean For Crypto?
BTC Market Is Closer To A Bottom Than Stocks (#GotBitcoin)
When World’s Central Banks Get It Wrong, Guess Who Pays The Price??? (#GotBitcoin)
“Better Days Ahead With Crypto Deleveraging Coming To An End” — Joker
Bitcoin Funds Have Seen Record Investment Inflow In Recent Weeks
Bitcoin’s Epic Run Is Winning More Attention On Wall Street
Ultimate Resource For Bitcoin Industry Mergers And Acquisitions (M&A) (#GotBitcoin)
Why Wall Street Is Literally Salivating Over Bitcoin
Nasdaq-Listed MicroStrategy And Others Wary Of Looming Dollar Inflation, Turns To Bitcoin And Gold
Bitcoin For Corporations | Michael Saylor | Bitcoin Corporate Strategy
Ultimate Resource On Myanmar’s Involvement With Bitcoin Industry
‘I Cry Every Day’: Olympic Athletes Slam Food, COVID Tests And Conditions In Beijing
Does Your Baby’s Food Contain Toxic Metals? Here’s What Our Investigation Found
Ultimate Resource For Pro-Bitcoin Lobbying And Non-Profit Organizations
Ultimate Resource On BlockFi, Celsius And Nexo
Petition Calling For Resignation Of U.S. Securities/Exchange Commission Chair Gary Gensler
100 Million Americans Can Legally Bet on the Super Bowl. A Spot Bitcoin ETF? Forget About it!
Green Finance Isn’t Going Where It’s Needed
Shedding Some Light On The Murky World Of ESG Metrics
SEC Targets Greenwashers To Bring Law And Order To ESG
BlackRock (Assets Under Management $7.4 Trillion) CEO: Bitcoin Has Caught Our Attention
Canada’s Major Banks Go Offline In Mysterious (Bank Run?) Hours-Long Outage (#GotBitcoin)
On-Chain Data: A Framework To Evaluate Bitcoin
On Its 14th Birthday, Bitcoin’s 1,690,706,971% Gain Looks Kind of… Well Insane
The Most Important Health Metric Is Now At Your Fingertips
American Bargain Hunters Flock To A New Online Platform Forged In China
Why We Should Welcome Another Crypto Winter
Traders Prefer Gold, Fiat Safe Havens Over Bitcoin As Russia Goes To War
Music Distributor DistroKid Raises Money At $1.3 Billion Valuation
Nas Selling Rights To Two Songs Via Crypto Music Startup Royal
Ultimate Resource On Music Catalog Deals
Ultimate Resource On Music And NFTs And The Implications For The Entertainment Industry
Lead And Cadmium Could Be In Your Dark Chocolate
Catawba, Native-American Tribe Approves First Digital Economic Zone In The United States
The Miracle Of Blockchain’s Triple Entry Accounting
How And Why To Stimulate Your Vagus Nerve!
Housing Boom Brings A Shortage Of Land To Build New Homes
Biden Lays Out His Blueprint For Fair Housing
No Grave Dancing For Sam Zell Now. He’s Paying Up For Hot Properties
Cracks In The Housing Market Are Starting To Show
Ever-Growing Needs Strain U.S. Food Bank Operations
Food Pantry Helps Columbia Students Struggling To Pay Bills
Food Insecurity Driven By Climate Change Has Central Americans Fleeing To The U.S.
Housing Insecurity Is Now A Concern In Addition To Food Insecurity
Families Face Massive Food Insecurity Levels
US Troops Going Hungry (Food Insecurity) Is A National Disgrace
Everything You Should Know About Community Fridges, From Volunteering To Starting Your Own
Russia’s Independent Journalists Including Those Who Revealed The Pandora Papers Need Your Help
10 Women Who Used Crypto To Make A Difference In 2021
Happy International Women’s Day! Leaders Share Their Experiences In Crypto
Dollar On Course For Worst Performance In Over A Decade (#GotBitcoin)
Juice The Stock Market And Destroy The Dollar!! (#GotBitcoin)
Unusual Side Hustles You May Not Have Thought Of
Ultimate Resource On Global Inflation And Rising Interest Rates (#GotBitcoin)
The Fed Is Setting The Stage For Hyper-Inflation Of The Dollar (#GotBitcoin)
An Antidote To Inflation? ‘Buy Nothing’ Groups Gain Popularity
Why Is Bitcoin Dropping If It’s An ‘Inflation Hedge’?
Lyn Alden Talks Bitcoin, Inflation And The Potential Coming Energy Shock
Ultimate Resource On How Black Families Can Fight Against Rising Inflation (#GotBitcoin)
What The Fed’s Rate Hike Means For Inflation, Housing, Crypto And Stocks
Egyptians Buy Bitcoin Despite Prohibitive New Banking Laws
Archaeologists Uncover Five Tombs In Egypt’s Saqqara Necropolis
History of Alchemy From Ancient Egypt To Modern Times
Former World Bank Chief Didn’t Act On Warnings Of Sexual Harassment
Does Your Hospital or Doctor Have A Financial Relationship With Big Pharma?
Ultimate Resource Covering The Crisis Taking Place In The Nickel Market
Apple Along With Meta And Secret Service Agents Fooled By Law Enforcement Impersonators
Handy Tech That Can Support Your Fitness Goals
How To Naturally Increase Your White Blood Cell Count
Ultimate Source For Russians Oligarchs And The Impact Of Sanctions On Them
Ultimate Source For Bitcoin Price Manipulation By Wall Street
Russia, Sri Lanka And Lebanon’s Defaults Could Be The First Of Many (#GotBitcoin)
Will Community Group Buying Work In The US?
Building And Running Businesses In The ‘Spirit Of Bitcoin’
What Is The Mysterious Liver Disease Hurting (And Killing) Children?
Citigroup Trader Is Scapegoat For Flash Crash In European Stocks (#GotBitcoin)
Bird Flu Outbreak Approaches Worst Ever In U.S. With 37 Million Animals Dead
Financial Inequality Grouped By Race For Blacks, Whites And Hispanics
How Black Businesses Can Prosper From Targeting A Trillion-Dollar Black Culture Market (#GotBitcoin)
Ultimate Resource For Central Bank Digital Currencies (#GotBitcoin) Page#2
Meet The Crypto Angel Investor Running For Congress In Nevada (#GotBitcoin?)
Introducing BTCPay Vault – Use Any Hardware Wallet With BTCPay And Its Full Node (#GotBitcoin?)
How Not To Lose Your Coins In 2020: Alternative Recovery Methods (#GotBitcoin?)
H.R.5635 – Virtual Currency Tax Fairness Act of 2020 ($200.00 Limit) 116th Congress (2019-2020)
Adam Back On Satoshi Emails, Privacy Concerns And Bitcoin’s Early Days
The Prospect of Using Bitcoin To Build A New International Monetary System Is Getting Real
How To Raise Funds For Australia Wildfire Relief Efforts (Using Bitcoin And/Or Fiat )
Former Regulator Known As ‘Crypto Dad’ To Launch Digital-Dollar Think Tank (#GotBitcoin?)
Currency ‘Cold War’ Takes Center Stage At Pre-Davos Crypto Confab (#GotBitcoin?)
A Blockchain-Secured Home Security Camera Won Innovation Awards At CES 2020 Las Vegas
Bitcoin’s Had A Sensational 11 Years (#GotBitcoin?)
Sergey Nazarov And The Creation Of A Decentralized Network Of Oracles
Google Suspends MetaMask From Its Play App Store, Citing “Deceptive Services”
Christmas Shopping: Where To Buy With Crypto This Festive Season
At 8,990,000% Gains, Bitcoin Dwarfs All Other Investments This Decade
Coinbase CEO Armstrong Wins Patent For Tech Allowing Users To Email Bitcoin
Bitcoin Has Got Society To Think About The Nature Of Money
How DeFi Goes Mainstream In 2020: Focus On Usability (#GotBitcoin?)
Dissidents And Activists Have A Lot To Gain From Bitcoin, If Only They Knew It (#GotBitcoin?)
At A Refugee Camp In Iraq, A 16-Year-Old Syrian Is Teaching Crypto Basics
Bitclub Scheme Busted In The US, Promising High Returns From Mining
Bitcoin Advertised On French National TV
Germany: New Proposed Law Would Legalize Banks Holding Bitcoin
How To Earn And Spend Bitcoin On Black Friday 2019
The Ultimate List of Bitcoin Developments And Accomplishments
Charities Put A Bitcoin Twist On Giving Tuesday
Family Offices Finally Accept The Benefits of Investing In Bitcoin
An Army Of Bitcoin Devs Is Battle-Testing Upgrades To Privacy And Scaling
Bitcoin ‘Carry Trade’ Can Net Annual Gains With Little Risk, Says PlanB
Max Keiser: Bitcoin’s ‘Self-Settlement’ Is A Revolution Against Dollar
Blockchain Can And Will Replace The IRS
China Seizes The Blockchain Opportunity. How Should The US Respond? (#GotBitcoin?)
Jack Dorsey: You Can Buy A Fraction Of Berkshire Stock Or ‘Stack Sats’
Bitcoin Price Skyrockets $500 In Minutes As Bakkt BTC Contracts Hit Highs
Bitcoin’s Irreversibility Challenges International Private Law: Legal Scholar
Bitcoin Has Already Reached 40% Of Average Fiat Currency Lifespan
Yes, Even Bitcoin HODLers Can Lose Money In The Long-Term: Here’s How (#GotBitcoin?)
Unicef To Accept Donations In Bitcoin (#GotBitcoin?)
Former Prosecutor Asked To “Shut Down Bitcoin” And Is Now Face Of Crypto VC Investing (#GotBitcoin?)
Switzerland’s ‘Crypto Valley’ Is Bringing Blockchain To Zurich
Next Bitcoin Halving May Not Lead To Bull Market, Says Bitmain CEO
Bitcoin Developer Amir Taaki, “We Can Crash National Economies” (#GotBitcoin?)
Veteran Crypto And Stocks Trader Shares 6 Ways To Invest And Get Rich
Is Chainlink Blazing A Trail Independent Of Bitcoin?
Nearly $10 Billion In BTC Is Held In Wallets Of 8 Crypto Exchanges (#GotBitcoin?)
SEC Enters Settlement Talks With Alleged Fraudulent Firm Veritaseum (#GotBitcoin?)
Blockstream’s Samson Mow: Bitcoin’s Block Size Already ‘Too Big’
Attorneys Seek Bank Of Ireland Execs’ Testimony Against OneCoin Scammer (#GotBitcoin?)
OpenLibra Plans To Launch Permissionless Fork Of Facebook’s Stablecoin (#GotBitcoin?)
Tiny $217 Options Trade On Bitcoin Blockchain Could Be Wall Street’s Death Knell (#GotBitcoin?)
Class Action Accuses Tether And Bitfinex Of Market Manipulation (#GotBitcoin?)
Sharia Goldbugs: How ISIS Created A Currency For World Domination (#GotBitcoin?)
Bitcoin Eyes Demand As Hong Kong Protestors Announce Bank Run (#GotBitcoin?)
How To Securely Transfer Crypto To Your Heirs
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
Crypto News From The Spanish-Speaking World (#GotBitcoin?)
Financial Services Giant Morningstar To Offer Ratings For Crypto Assets (#GotBitcoin?)
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
The Original Sins Of Cryptocurrencies (#GotBitcoin?)
Bitcoin Is The Fraud? JPMorgan Metals Desk Fixed Gold Prices For Years (#GotBitcoin?)
Israeli Startup That Allows Offline Crypto Transactions Secures $4M (#GotBitcoin?)
[PSA] Non-genuine Trezor One Devices Spotted (#GotBitcoin?)
Bitcoin Stronger Than Ever But No One Seems To Care: Google Trends (#GotBitcoin?)
First-Ever SEC-Qualified Token Offering In US Raises $23 Million (#GotBitcoin?)
You Can Now Prove A Whole Blockchain With One Math Problem – Really
Crypto Mining Supply Fails To Meet Market Demand In Q2: TokenInsight
$2 Billion Lost In Mt. Gox Bitcoin Hack Can Be Recovered, Lawyer Claims (#GotBitcoin?)
Fed Chair Says Agency Monitoring Crypto But Not Developing Its Own (#GotBitcoin?)
Wesley Snipes Is Launching A Tokenized $25 Million Movie Fund (#GotBitcoin?)
Mystery 94K BTC Transaction Becomes Richest Non-Exchange Address (#GotBitcoin?)
A Crypto Fix For A Broken International Monetary System (#GotBitcoin?)
Four Out Of Five Top Bitcoin QR Code Generators Are Scams: Report (#GotBitcoin?)
Waves Platform And The Abyss To Jointly Launch Blockchain-Based Games Marketplace (#GotBitcoin?)
Bitmain Ramps Up Power And Efficiency With New Bitcoin Mining Machine (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Miss Finland: Bitcoin’s Risk Keeps Most Women Away From Cryptocurrency (#GotBitcoin?)
Artist Akon Loves BTC And Says, “It’s Controlled By The People” (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Co-Founder Of LinkedIn Presents Crypto Rap Video: Hamilton Vs. Satoshi (#GotBitcoin?)
Crypto Insurance Market To Grow, Lloyd’s Of London And Aon To Lead (#GotBitcoin?)
No ‘AltSeason’ Until Bitcoin Breaks $20K, Says Hedge Fund Manager (#GotBitcoin?)
NSA Working To Develop Quantum-Resistant Cryptocurrency: Report (#GotBitcoin?)
Custody Provider Legacy Trust Launches Crypto Pension Plan (#GotBitcoin?)
Vaneck, SolidX To Offer Limited Bitcoin ETF For Institutions Via Exemption (#GotBitcoin?)
Russell Okung: From NFL Superstar To Bitcoin Educator In 2 Years (#GotBitcoin?)
Bitcoin Miners Made $14 Billion To Date Securing The Network (#GotBitcoin?)
Why Does Amazon Want To Hire Blockchain Experts For Its Ads Division?
Argentina’s Economy Is In A Technical Default (#GotBitcoin?)
Blockchain-Based Fractional Ownership Used To Sell High-End Art (#GotBitcoin?)
Portugal Tax Authority: Bitcoin Trading And Payments Are Tax-Free (#GotBitcoin?)
Bitcoin ‘Failed Safe Haven Test’ After 7% Drop, Peter Schiff Gloats (#GotBitcoin?)
Bitcoin Dev Reveals Multisig UI Teaser For Hardware Wallets, Full Nodes (#GotBitcoin?)
Bitcoin Price: $10K Holds For Now As 50% Of CME Futures Set To Expire (#GotBitcoin?)
Bitcoin Realized Market Cap Hits $100 Billion For The First Time (#GotBitcoin?)
Stablecoins Begin To Look Beyond The Dollar (#GotBitcoin?)
Bank Of England Governor: Libra-Like Currency Could Replace US Dollar (#GotBitcoin?)
Binance Reveals ‘Venus’ — Its Own Project To Rival Facebook’s Libra (#GotBitcoin?)
The Real Benefits Of Blockchain Are Here. They’re Being Ignored (#GotBitcoin?)
CommBank Develops Blockchain Market To Boost Biodiversity (#GotBitcoin?)
SEC Approves Blockchain Tech Startup Securitize To Record Stock Transfers (#GotBitcoin?)
SegWit Creator Introduces New Language For Bitcoin Smart Contracts (#GotBitcoin?)
You Can Now Earn Bitcoin Rewards For Postmates Purchases (#GotBitcoin?)
Bitcoin Price ‘Will Struggle’ In Big Financial Crisis, Says Investor (#GotBitcoin?)
Fidelity Charitable Received Over $100M In Crypto Donations Since 2015 (#GotBitcoin?)
Would Blockchain Better Protect User Data Than FaceApp? Experts Answer (#GotBitcoin?)
Just The Existence Of Bitcoin Impacts Monetary Policy (#GotBitcoin?)
What Are The Biggest Alleged Crypto Heists And How Much Was Stolen? (#GotBitcoin?)
IRS To Cryptocurrency Owners: Come Clean, Or Else!
Coinbase Accidentally Saves Unencrypted Passwords Of 3,420 Customers (#GotBitcoin?)
Bitcoin Is A ‘Chaos Hedge, Or Schmuck Insurance‘ (#GotBitcoin?)
Bakkt Announces September 23 Launch Of Futures And Custody
Coinbase CEO: Institutions Depositing $200-400M Into Crypto Per Week (#GotBitcoin?)
Researchers Find Monero Mining Malware That Hides From Task Manager (#GotBitcoin?)
Crypto Dusting Attack Affects Nearly 300,000 Addresses (#GotBitcoin?)
A Case For Bitcoin As Recession Hedge In A Diversified Investment Portfolio (#GotBitcoin?)
SEC Guidance Gives Ammo To Lawsuit Claiming XRP Is Unregistered Security (#GotBitcoin?)
15 Countries To Develop Crypto Transaction Tracking System: Report (#GotBitcoin?)
US Department Of Commerce Offering 6-Figure Salary To Crypto Expert (#GotBitcoin?)
Mastercard Is Building A Team To Develop Crypto, Wallet Projects (#GotBitcoin?)
Canadian Bitcoin Educator Scams The Scammer And Donates Proceeds (#GotBitcoin?)
Amazon Wants To Build A Blockchain For Ads, New Job Listing Shows (#GotBitcoin?)
Shield Bitcoin Wallets From Theft Via Time Delay (#GotBitcoin?)
Blockstream Launches Bitcoin Mining Farm With Fidelity As Early Customer (#GotBitcoin?)
Commerzbank Tests Blockchain Machine To Machine Payments With Daimler (#GotBitcoin?)
Man Takes Bitcoin Miner Seller To Tribunal Over Electricity Bill And Wins (#GotBitcoin?)
Bitcoin’s Computing Power Sets Record As Over 100K New Miners Go Online (#GotBitcoin?)
Walmart Coin And Libra Perform Major Public Relations For Bitcoin (#GotBitcoin?)
Judge Says Buying Bitcoin Via Credit Card Not Necessarily A Cash Advance (#GotBitcoin?)
Poll: If You’re A Stockowner Or Crypto-Currency Holder. What Will You Do When The Recession Comes?
1 In 5 Crypto Holders Are Women, New Report Reveals (#GotBitcoin?)
Beating Bakkt, Ledgerx Is First To Launch ‘Physical’ Bitcoin Futures In Us (#GotBitcoin?)
Facebook Warns Investors That Libra Stablecoin May Never Launch (#GotBitcoin?)
Government Money Printing Is ‘Rocket Fuel’ For Bitcoin (#GotBitcoin?)
Bitcoin-Friendly Square Cash App Stock Price Up 56% In 2019 (#GotBitcoin?)
Safeway Shoppers Can Now Get Bitcoin Back As Change At 894 US Stores (#GotBitcoin?)
TD Ameritrade CEO: There’s ‘Heightened Interest Again’ With Bitcoin (#GotBitcoin?)
Venezuela Sets New Bitcoin Volume Record Thanks To 10,000,000% Inflation (#GotBitcoin?)
Newegg Adds Bitcoin Payment Option To 73 More Countries (#GotBitcoin?)
China’s Schizophrenic Relationship With Bitcoin (#GotBitcoin?)
More Companies Build Products Around Crypto Hardware Wallets (#GotBitcoin?)
Bakkt Is Scheduled To Start Testing Its Bitcoin Futures Contracts Today (#GotBitcoin?)
Bitcoin Network Now 8 Times More Powerful Than It Was At $20K Price (#GotBitcoin?)
Crypto Exchange BitMEX Under Investigation By CFTC: Bloomberg (#GotBitcoin?)
“Bitcoin An ‘Unstoppable Force,” Says US Congressman At Crypto Hearing (#GotBitcoin?)
Bitcoin Network Is Moving $3 Billion Daily, Up 210% Since April (#GotBitcoin?)
Cryptocurrency Startups Get Partial Green Light From Washington
Fundstrat’s Tom Lee: Bitcoin Pullback Is Healthy, Fewer Searches Аre Good (#GotBitcoin?)
Bitcoin Lightning Nodes Are Snatching Funds From Bad Actors (#GotBitcoin?)
The Provident Bank Now Offers Deposit Services For Crypto-Related Entities (#GotBitcoin?)
Bitcoin Could Help Stop News Censorship From Space (#GotBitcoin?)
US Sanctions On Iran Crypto Mining — Inevitable Or Impossible? (#GotBitcoin?)
US Lawmaker Reintroduces ‘Safe Harbor’ Crypto Tax Bill In Congress (#GotBitcoin?)
EU Central Bank Won’t Add Bitcoin To Reserves — Says It’s Not A Currency (#GotBitcoin?)
The Miami Dolphins Now Accept Bitcoin And Litecoin Crypt-Currency Payments (#GotBitcoin?)
Trump Bashes Bitcoin And Alt-Right Is Mad As Hell (#GotBitcoin?)
Goldman Sachs Ramps Up Development Of New Secret Crypto Project (#GotBitcoin?)
Blockchain And AI Bond, Explained (#GotBitcoin?)
Grayscale Bitcoin Trust Outperformed Indexes In First Half Of 2019 (#GotBitcoin?)
XRP Is The Worst Performing Major Crypto Of 2019 (GotBitcoin?)
Bitcoin Back Near $12K As BTC Shorters Lose $44 Million In One Morning (#GotBitcoin?)
As Deutsche Bank Axes 18K Jobs, Bitcoin Offers A ‘Plan ฿”: VanEck Exec (#GotBitcoin?)
Argentina Drives Global LocalBitcoins Volume To Highest Since November (#GotBitcoin?)
‘I Would Buy’ Bitcoin If Growth Continues — Investment Legend Mobius (#GotBitcoin?)
Lawmakers Push For New Bitcoin Rules (#GotBitcoin?)
Facebook’s Libra Is Bad For African Americans (#GotBitcoin?)
Crypto Firm Charity Announces Alliance To Support Feminine Health (#GotBitcoin?)
Canadian Startup Wants To Upgrade Millions Of ATMs To Sell Bitcoin (#GotBitcoin?)
Trump Says US ‘Should Match’ China’s Money Printing Game (#GotBitcoin?)
Casa Launches Lightning Node Mobile App For Bitcoin Newbies (#GotBitcoin?)
Bitcoin Rally Fuels Market In Crypto Derivatives (#GotBitcoin?)
World’s First Zero-Fiat ‘Bitcoin Bond’ Now Available On Bloomberg Terminal (#GotBitcoin?)
Buying Bitcoin Has Been Profitable 98.2% Of The Days Since Creation (#GotBitcoin?)
Another Crypto Exchange Receives License For Crypto Futures
From ‘Ponzi’ To ‘We’re Working On It’ — BIS Chief Reverses Stance On Crypto (#GotBitcoin?)
These Are The Cities Googling ‘Bitcoin’ As Interest Hits 17-Month High (#GotBitcoin?)
Venezuelan Explains How Bitcoin Saves His Family (#GotBitcoin?)
Quantum Computing Vs. Blockchain: Impact On Cryptography
This Fund Is Riding Bitcoin To Top (#GotBitcoin?)
Bitcoin’s Surge Leaves Smaller Digital Currencies In The Dust (#GotBitcoin?)
Bitcoin Exchange Hits $1 Trillion In Trading Volume (#GotBitcoin?)
Bitcoin Breaks $200 Billion Market Cap For The First Time In 17 Months (#GotBitcoin?)
You Can Now Make State Tax Payments In Bitcoin (#GotBitcoin?)
Religious Organizations Make Ideal Places To Mine Bitcoin (#GotBitcoin?)
Goldman Sacs And JP Morgan Chase Finally Concede To Crypto-Currencies (#GotBitcoin?)
Bitcoin Heading For Fifth Month Of Gains Despite Price Correction (#GotBitcoin?)
Breez Reveals Lightning-Powered Bitcoin Payments App For IPhone (#GotBitcoin?)
Big Four Auditing Firm PwC Releases Cryptocurrency Auditing Software (#GotBitcoin?)
Amazon-Owned Twitch Quietly Brings Back Bitcoin Payments (#GotBitcoin?)
JPMorgan Will Pilot ‘JPM Coin’ Stablecoin By End Of 2019: Report (#GotBitcoin?)
Is There A Big Short In Bitcoin? (#GotBitcoin?)
Coinbase Hit With Outage As Bitcoin Price Drops $1.8K In 15 Minutes
Samourai Wallet Releases Privacy-Enhancing CoinJoin Feature (#GotBitcoin?)
There Are Now More Than 5,000 Bitcoin ATMs Around The World (#GotBitcoin?)
You Can Now Get Bitcoin Rewards When Booking At Hotels.Com (#GotBitcoin?)
North America’s Largest Solar Bitcoin Mining Farm Coming To California (#GotBitcoin?)
Bitcoin On Track For Best Second Quarter Price Gain On Record (#GotBitcoin?)
Bitcoin Hash Rate Climbs To New Record High Boosting Network Security (#GotBitcoin?)
Bitcoin Exceeds 1Million Active Addresses While Coinbase Custodies $1.3B In Assets
Why Bitcoin’s Price Suddenly Surged Back $5K (#GotBitcoin?)
Zebpay Becomes First Exchange To Add Lightning Payments For All Users (#GotBitcoin?)
Coinbase’s New Customer Incentive: Interest Payments, With A Crypto Twist (#GotBitcoin?)
The Best Bitcoin Debit (Cashback) Cards Of 2019 (#GotBitcoin?)
Real Estate Brokerages Now Accepting Bitcoin (#GotBitcoin?)
Ernst & Young Introduces Tax Tool For Reporting Cryptocurrencies (#GotBitcoin?)
Recession Is Looming, or Not. Here’s How To Know (#GotBitcoin?)
How Will Bitcoin Behave During A Recession? (#GotBitcoin?)
Many U.S. Financial Officers Think a Recession Will Hit Next Year (#GotBitcoin?)
Definite Signs of An Imminent Recession (#GotBitcoin?)
What A Recession Could Mean for Women’s Unemployment (#GotBitcoin?)
Investors Run Out of Options As Bitcoin, Stocks, Bonds, Oil Cave To Recession Fears (#GotBitcoin?)
Goldman Is Looking To Reduce “Marcus” Lending Goal On Credit (Recession) Caution (#GotBitcoin?)
Your Questions And Comments Are Greatly Appreciated.
Monty H. & Carolyn A.
Go back


Leave a Reply
You must be logged in to post a comment.