Parabens: A Cancer-Causing And DNA-Damaging Preservative Used In The Food And Cosmetic Industries
Parabens are a class of chemicals widely used as preservatives by cosmetic and pharmaceutical industries. Parabens Are A Cancer-Causing And DNA-Damaging Preservative Used In The Food And Cosmetic Industries
Parabens are effective preservatives in many types of formulas. These compounds, and their salts, are used primarily for their bactericidal and fungicidal properties. They can be found in shampoos, commercial moisturizers, shaving gels, personal lubricants, topical/parenteral pharmaceuticals, spray tanning solution, makeup, and toothpaste.
They are also used as food additives. Parabens: A Cancer-Causing And DNA-Damaging Preservative Used In The Food And Cosmetic Industries

Parabens have been widely used in products to prevent bacteria growth since the 1950s. “About 85 percent of cosmetics have them,” says Arthur Rich, Ph.D., a cosmetic chemist in Chestnut Ridge, New York. “They’re inexpensive.”
Related:
Does Your Baby’s Food Contain Toxic Metals? Here’s What Our Investigation Found
Lead And Cadmium Could Be In Your Dark Chocolate
Which Lipsticks Contain The Most Lead, Cadmium, Aluminum And Other Heavy Metals?
Poll: What Are You (Men/Women) Doing To Avoid The Toxins/Poisons In Commercial Beauty Products?
Secrets of ‘UltraGeezer,’ Earth’s Fastest 70-Year-Old Distance Runner
The Best Exercises For Your 50s, 60s, 70s—And Beyond
She’s Powerlifting at 76, So You’re Officially Out of Excuses
This Training Tweak Keeps Him Running At 70
33 Resistance Band Exercises You Can Do Literally Anywhere
Fighting Cancer By Releasing The Brakes On The Immune System
Over-Diagnosis And Over-Treatment Of Cancer In America Reaches Crisis Levels
Cancer Super-Survivors Use Their Own Bodies To Fight The Disease
FDA Says Up To Two Million People Exposed To Likely Carcinogens In Blood-Pressure Drugs
Carolyn’s Natural Organic Handmade Soap
Why Interior Designers And Home Stagers Prefer Bar Soap Over Liquid
We Now Live In A World With Customized Bar Soaps, Lotions And Shampoos
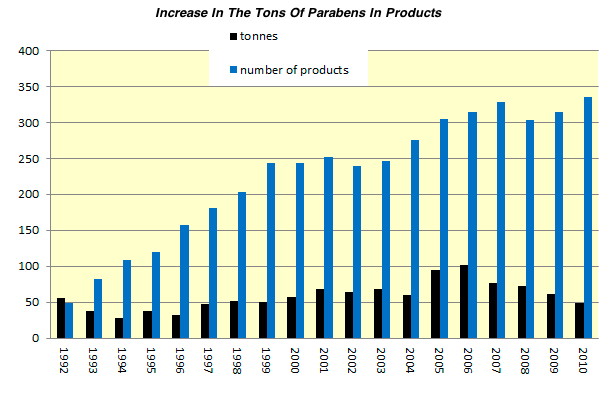
Their efficacy as preservatives, in combination with their low cost, probably explains why parabens are so commonplace. They are becoming increasingly controversial, however, because they have been found in breast cancer tumors.
Parabens have also displayed the ability to slightly mimic estrogen (a hormone known to play a role in the development of breast cancer). No effective direct links between parabens and cancer have been established, however. Another concern is that the estrogen-mimic aspect of parabens may be a factor in the increasing prevalence of early puberty in girls.
Typically, more than one form of the ingredient is used in a product. The most common are butylparaben, methylparaben, and propylparaben. Over the last few years, however, in response to customer concerns, many brands have started to manufacture (and label) paraben-free products, including lotions, lipsticks, shampoos, scrubs, and more.
Parabens are esters of para-hydroxybenzoic acid, from which the name is derived. Common parabens include methylparaben (E number E218), ethylparaben (E214), propylparaben (E216), butylparaben and heptylparaben (E209). Less common parabens include isobutylparaben, isopropylparaben, benzylparaben and their sodium salts.
Synthetically Produced
All commercially used parabens are synthetically produced, although some are identical to those found in nature. They are produced by the esterification of para-hydroxybenzoic acid with the appropriate alcohol, such as methanol, ethanol, or n-propanol. para-Hydroxybenzoic acid is in turn produced industrially from a modification of the Kolbe-Schmitt reaction, using potassium phenoxide and carbon dioxide.
So What’s The Problem?
In the 1990s, parabens were deemed xenoestrogens―agents that mimic estrogen in the body. “Estrogen disruption” has been linked to breast cancer and reproductive issues. And in 2004 British cancer researcher Philippa Darbre, Ph.D., found parabens present in malignant breast tumors.
As a result, experts in many countries are recommending limits on paraben levels in cosmetic and food products. What’s more, watchdog organizations worry that if parabens can be stored in the body, over time they could have a cumulative effect and pose a health risk.
Allergic Reactions
Parabens can cause skin irritation and contact dermatitis and rosacea in individuals with paraben allergies.
There’s reason to be mindful, but no reason to have an all-consuming concern about these chemicals. If it helps you rest easy, use a paraben-free body lotion (which coats a large area of skin). Today there are a number of formulas available from paraben-free brands (see below).
Breast Cancer
High levels of parabens have been detected in breast tumors. These findings, along with the demonstrated ability of some parabens to partially mimic estrogen, a hormone known to play a role in the development of breast cancers, have led some scientists to conclude that the presence of parabens may be associated with the occurrence of breast cancer, and to call for investigation into whether or not a causal link exists.
The lead researcher of the UK study, molecular biologist Philippa Darbre, reported that the ester-bearing form of the parabens found in the tumors indicate that they came from something applied to the skin, such as an underarm deodorant, cream or body spray, and stated that the results helped to explain why up to 60% of all breast tumors are found in just one-fifth of the breast – the upper-outer quadrant, nearest the underarm.
“From this research it is not possible to say whether parabens actually caused these tumors, but they may certainly be associated with the overall rise in breast cancer cases. Given that breast cancer is a large killer of women and a very high percentage of young women use underarm deodorants, I think we should be carrying out properly funded, further investigations into parabens and where they are found in the body,” says Philip Harvey, an editor of the Journal of Applied Toxicology, which published the research.
A 2004 study at Northwestern University found that an earlier age of breast cancer diagnosis related to more frequent use of antiperspirants/deodorants and underarm shaving.
“I personally feel there is a very strong correlation between the underarm hygiene habits and breast cancer,” said immunologist Dr. Kris McGrath, the author of the study.
This research has fueled a popular belief that the parabens in underarm deodorants and other cosmetics can migrate into breast tissue and contribute to the development of tumors.
If you want to play it extremely safe, use a few oil-based organic products that don’t contain water (which calls for a preservative).
They often come in dark containers with a pump so that light and air don’t degrade them quickly. “With truly natural products, just stay within their use-by date,” says Kane. “It’s like milk―the date is there for a reason.”

Medicinal Herb Ginger Helps Protect Against The Effect of Chemical Parabens
Traditional healers often use ginger to treat ailments ranging from nausea to arthritis pain. Recent studies highlight another use for ginger, the amelioration of the impact of parabens, a chemical widely used in the food and beverage industry and as a preservative in personal care products, drugs, and cosmetics.
Parabens have been in the news recently thanks to studies which show the vast majority of Americans show signs of exposure. If ginger does indeed reduce parabens’ impact that gives it increased importance as a significant natural health therapy.
Studies Show Parabens Are Widespread In The Human Population
Parabens role as a pseudo or xenoestrogen means they may be implicated in a number of health conditions such as the early onset of puberty and hormonally related illnesses such as breast and colon cancer. Parabens have also been linked to oxidative degradation of fats and reduced levels of anti-oxidants in the livers of mice.
How common are parabens? In a 2010 study in the journal Environmental Health Perspectives, scientists evaluated over 2,500 urine samples from Americans over the age of six and found exposure to methyl parabens in over 99% of the sample and to propyl parabens in over 92%.
Of special interest is their evaluation of the sample by age and sex which found adolescent and adult females had significantly higher concentration of methyl and propyl parabens than did any males. The authors attribute this to the fact that women and girls use many more personal care products than do males at any age.
These results also suggests the importance of considering the cumulative impact of parabens. Manufacturers may be correct in claiming that the amount in any one product does not pose a health risk, however since most women use multiple products, their cumulative impact can be significant.
Parabens May Be Linked To Breast Cancer
Studies of biopsy tissue from women with breast cancer have found measurable levels of parabens which are associated with the growth of cancer cells in test tubes.
An article published in January of 2012 in the Journal of Applied Toxicology measured parabens found in breast tissue from 40 women who had undergone a mastectomy. Measurements were taken at four locations across the breast. One or more parabens were found in 99% of the samples and five different parabens were found in 60% of the samples.
The study results also showed that the highest concentrations of parabens were found in the underarm area and entered the body through the skin.
Ginger Can Help Ameliorate The Impact of Parabens
In a 2009 animal study reported in the journal Acta Poloniae Pharmaceutica-Drug Research, scientists in India orally administered parabens to mice. The result was increased oxidative degradation of fats and decreased levels of anti-oxidants in the livers of of the treated mice compared to the controls.
Another group was given both parabens and ginger. In this group there was a significant decrease in paraben induced liver damage along with an increase in anti-oxidant levels. The scientists concluded that ginger can significantly reduce paraben induced liver damage.
In a 2006 study in the same journal human red blood cells were treated with parabens in a test tube. The result was a significant increase in the rate of hemolysis, or breakdown of the cells. When ginger was introduced to the samples along with parabens the result was a significant decrease in the rate of breakdown.
Paraben-Free Brands:
Aveda (aveda.com)
Burt’s Bees (burtsbees.com)
Dr. Hauschka (drhauschka.com)
John Masters Organics (johnmasters.com)
Josie Maran Cosmetics (josiemarancosmetics.com)
Korres (korresusa.com)
Origins (origins.com)
Pangea Organics (pangeaorganics.com)
Sun Exposure
Studies indicate that methylparaben applied on the skin may react with UVB leading to increased skin aging and DNA damage.
The Dangers of Sodium Lauryl Sulfate (SLS)
This section will focus on a compound called sodium lauryl/laureth sulfate (SLS/SLES), a very common chemical used throughout the cosmetic industry.
A great deal of misinformation, myth, and rumor surround SLS/SLES, and I would like to discuss what is really known about this chemical and its potential risk to you.
What You Put ON Your Skin Can Be More Dangerous Than What You Eat
Putting chemicals on your skin or scalp, such as getting a hair dye, may actually be worse than eating them. When you eat something, the enzymes in your saliva and stomach help to break it down and flush it out of your body.
However, when you put these chemicals on your skin, they are absorbed straight into your bloodstream without filtering of any kind, going directly to your delicate organs.
Once these chemicals find their way into your body, they tend to accumulate over time because you typically lack the necessary enzymes to break them down.
There are literally thousands of chemicals used in personal care products, and the U. S. government does not require any mandatory testing for these products before they are sold.
The Environmental Working Group (EWG) estimates that one out of five cosmetics might be contaminated with a cancer-causing agent. This nonprofit public-interest research group is known for making connections between chemical exposure and adverse health conditions.
The United Nations Environmental Programme estimates that approximately 70,000 chemicals are in common use across the world, with 1,000 new chemicals being introduced every year. Of all the chemicals used in cosmetics, the National Institute of Occupational Safety and Health reported that nearly 900 are toxic, and that estimate might be low.3
Many of the same poisons that pollute your environment are also lurking in the jars and bottles that line your bathroom shelves. We all risk becoming a toxic waste dump from the products we use, the foods we eat, and the environment in which we live.
Why Worry About Your Skin?
Your skin is much more than a wrap to keep you from sliding down into a puddle of formless bio-goo. It is your body’s largest organ.
You might not be aware of the many protective functions your skin serves. Consider that your skin:
1. Protects Your Internal Organs From Injury And Infection And Is Your Primary And Most Important Defense Against Infections.
2. Helps Eliminate Wastes Through Perspiration.
3. Assists Your Immune System By Providing A Protective Barrier To Viruses And Bad Bacteria, Thus Preventing Infections.
4. Provides A Friendly Habitat For Good Bacteria.
5. Helps Maintain Body Temperature By Controlling Heat Flow Between You And Your Environment.
6. Seals In Moisture, Maintaining Your Body’s Delicate Fluid Balance.
7. Produces Vitamin D, Which Is Crucial For Your Health.
8. Sends Sensory Feedback To Your Brain Because It Is Rich In Receptors, Such As Hard/Soft And Hot/Cold, So That You Can React To Dangerous Conditions Around You.
Your skin is vital to your health, yet many people fail to take care of it. Because your skin has the ability to absorb much of what you put on it, informed choices are critical to optimize your health.
You should give your skin the same thoughtful care you give your diet, because much of what goes ON you ends up going IN you.
Choose Your ‘Natural’ Cosmetics Carefully
There are no federal regulations for beauty products; anyone can claim their product is “natural” or “organic.” A label with the word “natural” does not mean the product contains only natural or organic ingredients.
According to the Organic Consumers Association, whose current “Coming Clean Campaign” aims to clean up the organic personal care product industry, the word “organic” is not properly regulated with personal care products as it is with food products, unless the product is certified by the USDA National Organic Program.4
In fact, some “organic” beauty products contain only a single-digit percentage of organic ingredients. Some brands use ingredients that were simply derived from natural sources but are highly processed and contain synthetic and petrochemical compounds.
When it comes to the labeling of cosmetics and body care products, it’s kind of a free-for-all.
In an OCA report released on March 14, 2008, at least one toxic, cancer-linked chemical was found in over 40 percent of products that call themselves “natural.”
Sodium Lauryl Sulfate (SLS), Sodium Laureth Sulfate (SLES), and Ammonium Laurel Sulfate (ALS)
Sodium lauryl sulfate is a surfactant, detergent, and emulsifier used in thousands of cosmetic products, as well as in industrial cleaners. It is present in nearly all shampoos, scalp treatments, hair color and bleaching agents, toothpastes, body washes and cleansers, make-up foundations, liquid hand soaps, laundry detergents, and bath oils/bath salts.
Although SLS Originates From Coconuts, The Chemical Is Anything But Natural.
The real problem with SLES/SLS is that the manufacturing process (ethoxylation) results in SLES/SLS being contaminated with 1,4 dioxane, a carcinogenic by-product, which will be discussed in more detail later.
SLS is the sodium salt of lauryl sulfate, and is classified by the EWG Cosmetics Database as a “denaturant, surfactant cleansing agent, emulsifier and foamer,” rated as a “moderate hazard.”
Similar to sodium lauryl sulfate (SLS) is sodium laureth sulfate (short for sodium lauryl ether sulfate, or SLES), a yellow detergent with higher foaming ability. SLES is considered to be slightly less irritating than SLS.
Ammonium lauryl sulfate (ALS) is another surfactant variation commonly put into cosmetics and cleansers to make them foam. ALS is similar to SLS, with similar risks.
SLS Goes By Other Names, Including:
* Sodium Dodecyl Sulfate
* A13-00356
* Sulfuric Acid, Monododecyl Ester, Sodium Salt
* Akyposal SDS
* Sodium Salt Sulfuric Acid
* Aquarex ME
* Monododecyl Ester Sodium Salt Sulfuric Acid
* Aquarex Methyl
Can 16,000 Studies About SLS Be Wrong?
According to the Environmental Working Group’s Skin Deep: Cosmetic Safety Reviews, research studies on SLS have shown links to:
* Irritation Of The Skin And Eyes
* Organ Toxicity
* Developmental/Reproductive Toxicity
* Neurotoxicity, Endocrine Disruption, Ecotoxicology, And Biochemical Or Cellular Changes
* Possible Mutations And Cancer
If you visit the SLS page on the Environmental Working Group‘s (EWG) website, you will see a very long list of health concerns and associated research studies. In fact, you will also see their mention of nearly 16,000 studies in the PubMed science library (as well as their link to that list) about the toxicity of this chemical.
There are clearly grounds for concern about using products containing this agent. Yet skeptics abound, claiming that these concerns are overblown and unfounded. It’s no wonder that consumers are completely confused about just how much risk this chemical poses.
Since most of the research studies are done on SLS itself—not on products containing it—the EWG states:
“Actual health risks will vary based on the level of exposure to the ingredient and individual susceptibility.”
Many of the studies on laboratory animals have involved applying SLS directly to the eyes of the animals and feeding them straight SLS. As would be expected with ANY chemical, eating it or putting it in your eyes would be bad news!
Even natural substances applied in high concentration (for example, cinnamon oil or oregano oil) can have harmful effects.
But high levels of SLS intake, either orally or through the skin, are not ordinarily experienced in normal cosmetics use—it’s the gradual, cumulative effects of long-term, repeated exposures that are the real concern. And there is a serious lack of long-term studies on ALL of the chemicals in these products—so we don’t really know what the long-term effects are.
It’s not just repeated exposure to one chemical—it’s the combined effect of thousands of little chemical exposures, day in and day out, that is of concern.
Sorting through the evidence is even more complicated when research findings are exaggerated and misquoted, and then circulated around the Internet as if it were fact.
The Green Study Debacle
A huge source of misinformation arose from a gross misinterpretation (or misrepresentation) of a study done by Dr. Keith Green of the Medical College of Georgia, Department of Ophthalmology, which looked at the uptake of SLS by eye tissues. Paula Begoun (aka “The Cosmetics Cop“) explains on her website how the Green controversy occurred.
Dr. Green investigated SLS uptake into the eye, but he did NOT study the effect of SLS on vision, nor did he study children or cataracts.
However, his findings were misquoted by anti-SLS zealots, to the point that he spent years trying to set the record straight about his findings and conclusions.
Dr. Green found that SLS is rapidly taken up and accumulated by eye tissues, where it is retained for up to five days. He also found that SLS uptake is greater in younger rabbits than in adult rabbits, and that SLS causes changes in some eye proteins.
However, someone quoted him as writing (in a report to the Research to Prevent Blindness conference):
“SLS is a systemic that can penetrate and be retained in the eye, brain, heart, liver, etc., with potentially harmful long-term effects. It can retard healing and cause cataracts in adults, and can keep children’s eyes from developing properly.”
Of course, this statement went far beyond the reaches of his study—and he denied ever saying it. The controversy that ensued led to a whole slew of articles and statements, based on this misinformation, that have done nothing but add to the confusion about SLS and fueling both sides of the issue.
Dr. Green later stated in an interview with Paula Begoun:
“There is no part of my study that indicated any eye development or cataract problems from SLS or SLES and the body does not retain those ingredients at all.”
He also said that he did not even look at the issue with children, and later claimed his findings were so insignificant that he no longer had any interest in further researching the subject.
In spite of Green’s later statements dismissing the importance of his findings, there are legitimate concerns about SLS and its systemic effects—based on multiple other studies.
The fact that one study’s findings were misrepresented doesn’t mean the risks aren’t real. Naysayers are fond of citing the Green study debacle but NOT mentioning the other evidence of potential health risks of SLS.
Real Dangers of SLS—Rumors Aside
A number of studies report SLS being damaging to oral mucosa and skin. This is not at all surprising since SLS is actually used as a skin irritant during studies where medical treatments for skin irritation require an intentionally irritating agent.
A study at the Stern College for Women at Yeshiva University in New York in 1997 examined SLS in mouthwash. They found that SLS in mouth rinses caused desquamation of oral epithelium and a burning sensation in human volunteers.
A study appearing in Exogenous Dermatology confirmed SLS to be a very “corrosive irritant” to the skin—irritation which persisted in research subjects for 3 weeks. SLS exerts its damage by stripping your skin of protective oils and moisture.
SLS is associated with increased aphthous ulcers (canker sores) due to the denaturing effect and irritation of the oral mucosa.
Swallowing SLS will likely lead to nausea and diarrhea and is even used as a laxative in enemas. So be careful not to swallow much of your toothpaste if it contains SLS.
According to Judi Vance, author of Beauty to Die For, SLS can cause cellular DNA damage. In an article for ConsumerHealth.org, she states that a dental association in Japan tested the effects of SLS on bacteria, finding it to be mutagenic. She also states that hair follicles are significant transporters of harmful chemicals into your body.
Links Between SLS, Ethylene Oxide, 1,4 Dioxane, and Cancer
The evidence linking SLS to cancer is a bit challenging due to the paucity of scientific studies. However, carcinogenic effects are quite possible when you consider that SLS/SLES is often contaminated by two known carcinogens:
Ethylene oxide (which is what the “E” in SLES represents). A return to the Skin Deep website for ethylene oxide reveals a rating of “high hazard,” which appears as an impurity in thousands of personal care products. It is used to “ethoxylate” SLS and other chemicals, to make them less harsh.
1,4 dioxane, a byproduct of ethylene oxide, also receives a “high hazard” rating from Skin Deep and is associated with an even longer list of common personal care products. On the CDC site, 1,4 dioxane is described as “probably carcinogenic to humans,” toxic to the brain and central nervous system, kidneys, and liver. It is also a leading groundwater contaminant.
To avoid 1,4 dioxane, the Organic Consumers Association (OCA) recommends avoiding products with indications of ethoxylation.
To do this, look for the following suffixes in the ingredient list: “myreth,” “oleth,” “laureth,” “ceteareth,” any other “eth,” “PEG,” “polyethylene,” “polyethylene glycol,” “polyoxyethylene,” or “oxynol.”
For Example—Sodium Laureth Sulfate.
Both polysorbate 60 and polysorbate 80 are also often contaminated with 1,4 dioxane, according to Dr. Samuel Epstein.
The FDA continues to take the stance that the levels of 1,4 dioxane in body care products are too low to be considered harmful. But given that there are products available that have NO 1,4 dioxane, why take a chance with your health?
Your best bet is to purchase products that are certified under the USDA National Organic Program, and if those aren’t available, select products whose ingredients you recognize—and can pronounce!
SLS and Nitrosamines
SLS has also been linked to nitrosamines. Nitrosamines are potent carcinogens that cause your body to absorb nitrates, which are known to be carcinogenic as well.
According to one article by Greenfeet, at least one study linked SLS to nitrate absorption.
The Greenfeet Article States:
“A study cited in the Wall Street Journal (November 1, 1988) linked SLS to cataracts and nitrate absorption (nitrates are carcinogens—or cancer causing substances). Apparently, this absorption occurs when the SLS becomes contaminated with NDELA (N-nitrosodiethanolamine) during processing.
This contamination comes about as a result of SLS coming into contact with any number of chemicals including TEA (triethanolamine), which is a commonly used ingredient in shampoos as a detergent.”
So, the SLS combines with the TEA, resulting in NDELA, which is a nitrosamine and a recognized carcinogen.
The biochemistry is very complex due to the “chemical cocktail” that is your shampoo or hand wash. When these chemical ingredients come into contact with each other, all sorts of molecular bonds begin to form and new and unintended chemicals are produced.
Unfortunately, Some Of These Unintended Chemicals Are Nitrosamines.
As the above article points out, there is no way the FDA can possibly test all of the combinations of chemicals available, in every unique blend.
So, while the individual ingredients may be considered safe, once you mix them up into a brew, all bets are off. Just because SLS doesn’t contain nitrogen, doesn’t mean it can’t GET a nitrogen from the chemical soup and bond with it to form deadly nitrosamine.
How to Evaluate Your Toxic Toiletry Burden
Lest you shrug these findings off, thinking that your exposure is “insignificant,” think again.
Did you know that, if you use conventional cosmetics on a daily basis, you can absorb almost five pounds of chemicals and toxins into your body each year?
Daily use of ordinary, seemingly benign personal care products like shampoo, toothpaste and shower gel can easily result in exposure to thousands of chemicals, and many will make their way into your body and become “stuck” there, since you lack the means to break them down.
This toxic load can become a significant contributing factor to health problems and serious diseases, especially if your diet and exercise habits are lacking.
Women seem to be predisposed to more autoimmune disorders than men. Diseases such as thyroid disease, fibromyalgia, and multiple sclerosis are far more common in women. Perhaps one of the major contributing factors is that women tend to use far more personal products than men.
If you are a woman, acting on the information in this report is particularly important. Is your make-up cabinet a toxic wasteland?
It is especially challenging to establish a link between these routine chemical exposures and health problems down the road, because the adverse effects might not show up for years.
As Theo Colburn discusses in Our Stolen Future, in some cases, effects are not seen in the person exposed but DO appear in her offspring.
This has been seen in the animal kingdom, as well as in humans. Some adults have been known to suddenly show a disease many decades after prenatal exposure.
If you would like to learn more about the health effects of the chemicals you are routinely exposed to, I strongly urge you to read Our Toxic World: A Wake Up Call by Dr. Doris Rapp. She does a thorough job of uncovering the many ways we are exposed to toxic chemicals and how they contribute to chronic disease.
A Newer, Greener YOU!
With the jury still out about long-term exposure to SLS and its associated contaminants, the best advice is to avoid them and avoid the risk altogether—since there are safe alternatives available.
The easiest way to ensure that you’re not being exposed to potentially hazardous agents is to make your own personal care products, using simple all-natural ingredients that you may already have in your home.
Finding recipes for your own homemade beauty products is a breeze when you have access to the Internet. Just Google “homemade cosmetics” for more than 400,000 pages of recipes and instructions.
Updated: 7-31-2022
A Banana Boat Sunscreen Spray Was Recalled Due To Presence Of Carcinogen Benzene
Trace amounts of the cancer-causing chemical were found in some samples of Hair & Scalp SPF 30 sunscreen aerosol products.
Edgewell Personal Care Co. issued a voluntary recall of one of its Banana Boat sunscreen spray products after an internal review found trace levels of the carcinogen benzene in some samples.
Edgewell pulled several batches of its Banana Boat Hair & Scalp Sunscreen Spray SPF 30 after tests found that the product contained small amounts of the cancer-causing chemical, the manufacturer said Friday in a statement to the U.S. Food and Drug Administration.
The company said benzene isn’t an ingredient in any Banana Boat product, but an internal review showed that unexpected levels of the chemical came from the propellant that sprays the sunscreen out of the can.
Exposure to benzene could potentially result in blood disorders, leukemia and other cancers, the company said. Edgewell said it hasn’t received any notice of adverse events related to the recall to date.
“Daily exposure to benzene in the recalled products would not be expected to cause adverse health consequences according to an independent health assessment using established exposure modeling guidelines,” the company said.
The Shelton, Conn.-based company said in its voluntary recall that the affected products have expiration dates of December 2022, February 2023 and April 2024.
Consumers can identify possibly tainted products by the lot numbers on the bottom of Banana Boat Hair & Scalp Sunscreen Spray SPF 30 cans.
Lot numbers 20016AF, 20084BF and 21139AF shouldn’t be used, the company said.
Banana Boat will offer reimbursement to consumers who bought the recalled sunscreen spray products packaged in aerosol cans.
The products were distributed nationwide in the U.S. through various retailers and online. Edgewell said it has notified its retailers to remove any remaining recalled product from shelves.
Benzene, a colorless chemical with a sweet odor, is found in natural sources like volcanoes and forest fires, and can also be man-made, according to the Centers for Disease Control and Prevention.
It is widely used in the U.S., ranking in the top 20 chemicals for production volume, the CDC said.
In July 2021, Johnson & Johnson recalled most of its Neutrogena and Aveeno spray sunscreens from U.S. stores after detecting benzene in some samples. Later that year, Procter & Gamble Co. issued a recall for its Old Spice and Secret antiperspirant sprays after benzene was detected in some of the products.
Related Articles:
Bitcoin Information & Resources (#GotBitcoin)
North Korea Infiltrates U.S. Remote Jobs—With The Help of Everyday Americans
America’s Leading Alien Hunters Depend On AI To Speed Their Search
Bitcoin’s Nouveau Riche Executives And Wealthy Investors In Search Of Ways To Protect Themselves
How Anthocyanins Shield Us From Microplastics
Origins Of Toxic Algae (Domoic Acid) Killing Sea Lions, Birds And Dolphins In Southern California
The Human Brain And It’s Ability To Time-Travel
Wall Street’s New Tariff Safe Haven: Biotech Companies Adopting Bitcoin Treasury Strategy
How Natural “Short Sleepers” Thrive On 4 Hours Of Sleep Per Night Thanks To A Gene Mutation
Nine Unknown Benefits of Sleep Including Penal And Clitoral Erections
Elite Athletes Try A New Training Tactic: More Vitamin D3
Bitcoin Industry On A Winning Streak As Sec. Drops Lawsuit After Lawsuit!!
FOIA Files: Elon Musk’s DOGE Wants A Heads Up On FOIA Requests
When Russia Came To Defend The United States
Ultimate Resource Covering DOGE AKA The Department of Government Efficiency
Your Money Is Currently Lawful. However, That Is Subject To Change #GotBitcoin #BitcoinFixesThis
Is The United Nations Going Bankrupt?
Bitcoin Industry Founders Should Stop Chasing Narratives And Start Creating Them
Consumer Finance Watchdog Finalizes ‘Open Banking’ Rules Aimed At Driving Competition
US Government Crypto Wallets Hacked For $20M — Arkham Intelligence
Coinbase Files FOIAs Against US Regulators Probing Banks’ Bitcoin Crackdown
The Botanist Daring To Ask: What If Plants Have Intelligence?
Theoretical Physicist Sylvester James Gates
FOIA Request Reveals SEC’s Gensler, Lawmakers Target Of Violent Threats
Ultimate Resource For Prediction Markets Covering Politics, Pop Culture, Sports And More
September (AKA Rektember) Is Once Again A Tough Month For Stocks AND Bitcoin
Ultimate Resource For Cloning humans. Biological, Ethical, And Social Considerations
FOIA Reveals Aurora Borealis Imperiled US Infrastructure From Power Grids To Satellites (5-10-2024)
Ultimate Resource Covering How “Silent Payments” Are Bringing New Privacy Protections To Bitcoin
Nostr Allows Bitcoiners To Build-Out A Decentralized, CENSORSHIP-RESISTANT Social-Media!!
Joe Rogan: I Have A Lot Of Hope For Bitcoin
Teen Cyber Prodigy Stumbled Onto Flaw Letting Him Hijack Teslas
Spyware Finally Got Scary Enough To Freak Lawmakers Out—After It Spied On Them
The First Nuclear-Powered Bitcoin Mine Is Here. Maybe It Can Clean Up Energy FUD
The World’s Best Bitcoin Policies: How They Do It In 37 Nations
Tonga To Copy El Salvador’s Bill Making Bitcoin Legal Tender, Says Former MP
Wordle Is The New “Lingo” Turning Fans Into Argumentative Strategy Nerds
Prospering In The Pandemic, Some Feel Financial Guilt And Gratitude
Is Art Therapy The Path To Mental Well-Being?
New York, California, New Jersey, And Alabama Move To Ban ‘Forever Chemicals’ In Firefighting Foam
The Mystery Of The Wasting House-Cats
What Pet Owners Should Know About Chronic Kidney Disease In Dogs And Cats
Pets Score Company Perks As The ‘New Dependents’
Why Is My Cat Rubbing His Face In Ants?
Natural Cure For Hyperthyroidism In Cats Including How To Switch Him/Her To A Raw Food Diet
Ultimate Resource For Cat Lovers
FDA Approves First-Ever Arthritis Pain Management Drug For Cats
Ultimate Resource On Duke of York’s Prince Andrew And His Sex Scandal
Walmart Filings Reveal Plans To Create Cryptocurrency, NFTs
Bitcoin’s Dominance of Crypto Payments Is Starting To Erode
T-Mobile Says Hackers Stole Data On About 37 Million Customers
Jack Dorsey Announces Bitcoin Legal Defense Fund
More Than 100 Millionaires Signed An Open Letter Asking To Be Taxed More Heavily
Federal Regulator Says Credit Unions Can Partner With Bitcoin Providers
What’s Behind The Fascination With Smash-And-Grab Shoplifting?
Train Robberies Are A Problem In Los Angeles, And No One Agrees On How To Stop Them
US Stocks Historically Deliver Strong Gains In Fed Hike Cycles (GotBitcoin)
Ian Alexander Jr., Only Child of Regina King, Dies At Age 26
Amazon Ends Its Charity Donation Program Amazonsmile After Other Cost-Cutting Efforts
BTC Panics, Then Jeers at DOJ Announcement of ‘Major Action’ Against Tiny Chinese Exchange Bitzlato
Indexing Is Coming To BTC Funds Via Decentralized Exchanges
Doctors Show Implicit Bias Towards Black Patients
Darkmail Pushes Privacy Into The Hands Of NSA-Weary Customers
3D Printing Make Anything From Candy Bars To Hand Guns
Stealing The Blood Of The Young May Make You More Youthful
Henrietta Lacks And Her Remarkable Cells Will Finally See Some Payback
AL_A Wins Approval For World’s First Magnetized Fusion Power Plant
Want To Be Rich? Bitcoin’s Limited Supply Cap Means You Only Need 0.01 BTC
Smart Money Is Buying Bitcoin Dip. Stocks, Not So Much
McDonald’s Jumps On Bitcoin Memewagon, Crypto Twitter Responds
America COMPETES Act Would Be Disastrous For Bitcoin Inustry (LOL) And More
Lyn Alden On Bitcoin, Inflation And The Potential Coming Energy Shock
Inflation And A Tale of Cantillionaires
El Salvador Plans Bill To Adopt Bitcoin As Legal Tender
Miami Mayor Says City Employees Should Be Able To Take Their Salaries In Bitcoin
Vast Troves of Classified Info Undermine National Security, Spy Chief Says
BREAKING: Arizona State Senator Introduces Bill To Make Bitcoin Legal Tender
San Francisco’s Historic Surveillance Law May Get Watered Down
How Bitcoin Contributions Funded A $1.4M Solar Installation In Zimbabwe
California Lawmaker Says National Privacy Law Is a Priority
The Pandemic Turbocharged Online Privacy Concerns
How To Protect Your Online Privacy While Working From Home
Researchers Use GPU Fingerprinting To Track Users Online
Japan’s $1 Trillion Bitcoin Industry May Ease Onerous Listing Rules
Ultimate Resource On A Weak / Strong Dollar’s Impact On Bitcoin
Fed Money Printer Goes Into Reverse (Quantitative Tightening): What Does It Mean For Crypto?
BTC Market Is Closer To A Bottom Than Stocks (#GotBitcoin)
When World’s Central Banks Get It Wrong, Guess Who Pays The Price??? (#GotBitcoin)
“Better Days Ahead With Crypto Deleveraging Coming To An End” — Joker
Bitcoin Funds Have Seen Record Investment Inflow In Recent Weeks
Bitcoin’s Epic Run Is Winning More Attention On Wall Street
Ultimate Resource For Bitcoin Industry Mergers And Acquisitions (M&A) (#GotBitcoin)
Why Wall Street Is Literally Salivating Over Bitcoin
Nasdaq-Listed MicroStrategy And Others Wary Of Looming Dollar Inflation, Turns To Bitcoin And Gold
Bitcoin For Corporations | Michael Saylor | Bitcoin Corporate Strategy
Ultimate Resource On Myanmar’s Involvement With Bitcoin Industry
‘I Cry Every Day’: Olympic Athletes Slam Food, COVID Tests And Conditions In Beijing
Does Your Baby’s Food Contain Toxic Metals? Here’s What Our Investigation Found
Ultimate Resource For Pro-Bitcoin Lobbying And Non-Profit Organizations
Ultimate Resource On BlockFi, Celsius And Nexo
Petition Calling For Resignation Of U.S. Securities/Exchange Commission Chair Gary Gensler
100 Million Americans Can Legally Bet on the Super Bowl. A Spot Bitcoin ETF? Forget About it!
Green Finance Isn’t Going Where It’s Needed
Shedding Some Light On The Murky World Of ESG Metrics
SEC Targets Greenwashers To Bring Law And Order To ESG
BlackRock (Assets Under Management $7.4 Trillion) CEO: Bitcoin Has Caught Our Attention
Canada’s Major Banks Go Offline In Mysterious (Bank Run?) Hours-Long Outage (#GotBitcoin)
On-Chain Data: A Framework To Evaluate Bitcoin
On Its 14th Birthday, Bitcoin’s 1,690,706,971% Gain Looks Kind of… Well Insane
The Most Important Health Metric Is Now At Your Fingertips
American Bargain Hunters Flock To A New Online Platform Forged In China
Why We Should Welcome Another Crypto Winter
Traders Prefer Gold, Fiat Safe Havens Over Bitcoin As Russia Goes To War
Music Distributor DistroKid Raises Money At $1.3 Billion Valuation
Nas Selling Rights To Two Songs Via Crypto Music Startup Royal
Ultimate Resource On Music Catalog Deals
Ultimate Resource On Music And NFTs And The Implications For The Entertainment Industry
Lead And Cadmium Could Be In Your Dark Chocolate
Catawba, Native-American Tribe Approves First Digital Economic Zone In The United States
The Miracle Of Blockchain’s Triple Entry Accounting
How And Why To Stimulate Your Vagus Nerve!
Housing Boom Brings A Shortage Of Land To Build New Homes
Biden Lays Out His Blueprint For Fair Housing
No Grave Dancing For Sam Zell Now. He’s Paying Up For Hot Properties
Cracks In The Housing Market Are Starting To Show
Ever-Growing Needs Strain U.S. Food Bank Operations
Food Pantry Helps Columbia Students Struggling To Pay Bills
Food Insecurity Driven By Climate Change Has Central Americans Fleeing To The U.S.
Housing Insecurity Is Now A Concern In Addition To Food Insecurity
Families Face Massive Food Insecurity Levels
US Troops Going Hungry (Food Insecurity) Is A National Disgrace
Everything You Should Know About Community Fridges, From Volunteering To Starting Your Own
Russia’s Independent Journalists Including Those Who Revealed The Pandora Papers Need Your Help
10 Women Who Used Crypto To Make A Difference In 2021
Happy International Women’s Day! Leaders Share Their Experiences In Crypto
Dollar On Course For Worst Performance In Over A Decade (#GotBitcoin)
Juice The Stock Market And Destroy The Dollar!! (#GotBitcoin)
Unusual Side Hustles You May Not Have Thought Of
Ultimate Resource On Global Inflation And Rising Interest Rates (#GotBitcoin)
The Fed Is Setting The Stage For Hyper-Inflation Of The Dollar (#GotBitcoin)
An Antidote To Inflation? ‘Buy Nothing’ Groups Gain Popularity
Why Is Bitcoin Dropping If It’s An ‘Inflation Hedge’?
Lyn Alden Talks Bitcoin, Inflation And The Potential Coming Energy Shock
Ultimate Resource On How Black Families Can Fight Against Rising Inflation (#GotBitcoin)
What The Fed’s Rate Hike Means For Inflation, Housing, Crypto And Stocks
Egyptians Buy Bitcoin Despite Prohibitive New Banking Laws
Archaeologists Uncover Five Tombs In Egypt’s Saqqara Necropolis
History of Alchemy From Ancient Egypt To Modern Times
Former World Bank Chief Didn’t Act On Warnings Of Sexual Harassment
Does Your Hospital or Doctor Have A Financial Relationship With Big Pharma?
Ultimate Resource Covering The Crisis Taking Place In The Nickel Market
Apple Along With Meta And Secret Service Agents Fooled By Law Enforcement Impersonators
Handy Tech That Can Support Your Fitness Goals
How To Naturally Increase Your White Blood Cell Count
Ultimate Source For Russians Oligarchs And The Impact Of Sanctions On Them
Ultimate Source For Bitcoin Price Manipulation By Wall Street
Russia, Sri Lanka And Lebanon’s Defaults Could Be The First Of Many (#GotBitcoin)
Will Community Group Buying Work In The US?
Building And Running Businesses In The ‘Spirit Of Bitcoin’
What Is The Mysterious Liver Disease Hurting (And Killing) Children?
Citigroup Trader Is Scapegoat For Flash Crash In European Stocks (#GotBitcoin)
Bird Flu Outbreak Approaches Worst Ever In U.S. With 37 Million Animals Dead
Financial Inequality Grouped By Race For Blacks, Whites And Hispanics
How Black Businesses Can Prosper From Targeting A Trillion-Dollar Black Culture Market (#GotBitcoin)
Ultimate Resource For Central Bank Digital Currencies (#GotBitcoin) Page#2
Meet The Crypto Angel Investor Running For Congress In Nevada (#GotBitcoin?)
Introducing BTCPay Vault – Use Any Hardware Wallet With BTCPay And Its Full Node (#GotBitcoin?)
How Not To Lose Your Coins In 2020: Alternative Recovery Methods (#GotBitcoin?)
H.R.5635 – Virtual Currency Tax Fairness Act of 2020 ($200.00 Limit) 116th Congress (2019-2020)
Adam Back On Satoshi Emails, Privacy Concerns And Bitcoin’s Early Days
The Prospect of Using Bitcoin To Build A New International Monetary System Is Getting Real
How To Raise Funds For Australia Wildfire Relief Efforts (Using Bitcoin And/Or Fiat )
Former Regulator Known As ‘Crypto Dad’ To Launch Digital-Dollar Think Tank (#GotBitcoin?)
Currency ‘Cold War’ Takes Center Stage At Pre-Davos Crypto Confab (#GotBitcoin?)
A Blockchain-Secured Home Security Camera Won Innovation Awards At CES 2020 Las Vegas
Bitcoin’s Had A Sensational 11 Years (#GotBitcoin?)
Sergey Nazarov And The Creation Of A Decentralized Network Of Oracles
Google Suspends MetaMask From Its Play App Store, Citing “Deceptive Services”
Christmas Shopping: Where To Buy With Crypto This Festive Season
At 8,990,000% Gains, Bitcoin Dwarfs All Other Investments This Decade
Coinbase CEO Armstrong Wins Patent For Tech Allowing Users To Email Bitcoin
Bitcoin Has Got Society To Think About The Nature Of Money
How DeFi Goes Mainstream In 2020: Focus On Usability (#GotBitcoin?)
Dissidents And Activists Have A Lot To Gain From Bitcoin, If Only They Knew It (#GotBitcoin?)
At A Refugee Camp In Iraq, A 16-Year-Old Syrian Is Teaching Crypto Basics
Bitclub Scheme Busted In The US, Promising High Returns From Mining
Bitcoin Advertised On French National TV
Germany: New Proposed Law Would Legalize Banks Holding Bitcoin
How To Earn And Spend Bitcoin On Black Friday 2019
The Ultimate List of Bitcoin Developments And Accomplishments
Charities Put A Bitcoin Twist On Giving Tuesday
Family Offices Finally Accept The Benefits of Investing In Bitcoin
An Army Of Bitcoin Devs Is Battle-Testing Upgrades To Privacy And Scaling
Bitcoin ‘Carry Trade’ Can Net Annual Gains With Little Risk, Says PlanB
Max Keiser: Bitcoin’s ‘Self-Settlement’ Is A Revolution Against Dollar
Blockchain Can And Will Replace The IRS
China Seizes The Blockchain Opportunity. How Should The US Respond? (#GotBitcoin?)
Jack Dorsey: You Can Buy A Fraction Of Berkshire Stock Or ‘Stack Sats’
Bitcoin Price Skyrockets $500 In Minutes As Bakkt BTC Contracts Hit Highs
Bitcoin’s Irreversibility Challenges International Private Law: Legal Scholar
Bitcoin Has Already Reached 40% Of Average Fiat Currency Lifespan
Yes, Even Bitcoin HODLers Can Lose Money In The Long-Term: Here’s How (#GotBitcoin?)
Unicef To Accept Donations In Bitcoin (#GotBitcoin?)
Former Prosecutor Asked To “Shut Down Bitcoin” And Is Now Face Of Crypto VC Investing (#GotBitcoin?)
Switzerland’s ‘Crypto Valley’ Is Bringing Blockchain To Zurich
Next Bitcoin Halving May Not Lead To Bull Market, Says Bitmain CEO
Bitcoin Developer Amir Taaki, “We Can Crash National Economies” (#GotBitcoin?)
Veteran Crypto And Stocks Trader Shares 6 Ways To Invest And Get Rich
Is Chainlink Blazing A Trail Independent Of Bitcoin?
Nearly $10 Billion In BTC Is Held In Wallets Of 8 Crypto Exchanges (#GotBitcoin?)
SEC Enters Settlement Talks With Alleged Fraudulent Firm Veritaseum (#GotBitcoin?)
Blockstream’s Samson Mow: Bitcoin’s Block Size Already ‘Too Big’
Attorneys Seek Bank Of Ireland Execs’ Testimony Against OneCoin Scammer (#GotBitcoin?)
OpenLibra Plans To Launch Permissionless Fork Of Facebook’s Stablecoin (#GotBitcoin?)
Tiny $217 Options Trade On Bitcoin Blockchain Could Be Wall Street’s Death Knell (#GotBitcoin?)
Class Action Accuses Tether And Bitfinex Of Market Manipulation (#GotBitcoin?)
Sharia Goldbugs: How ISIS Created A Currency For World Domination (#GotBitcoin?)
Bitcoin Eyes Demand As Hong Kong Protestors Announce Bank Run (#GotBitcoin?)
How To Securely Transfer Crypto To Your Heirs
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
Crypto News From The Spanish-Speaking World (#GotBitcoin?)
Financial Services Giant Morningstar To Offer Ratings For Crypto Assets (#GotBitcoin?)
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
The Original Sins Of Cryptocurrencies (#GotBitcoin?)
Bitcoin Is The Fraud? JPMorgan Metals Desk Fixed Gold Prices For Years (#GotBitcoin?)
Israeli Startup That Allows Offline Crypto Transactions Secures $4M (#GotBitcoin?)
[PSA] Non-genuine Trezor One Devices Spotted (#GotBitcoin?)
Bitcoin Stronger Than Ever But No One Seems To Care: Google Trends (#GotBitcoin?)
First-Ever SEC-Qualified Token Offering In US Raises $23 Million (#GotBitcoin?)
You Can Now Prove A Whole Blockchain With One Math Problem – Really
Crypto Mining Supply Fails To Meet Market Demand In Q2: TokenInsight
$2 Billion Lost In Mt. Gox Bitcoin Hack Can Be Recovered, Lawyer Claims (#GotBitcoin?)
Fed Chair Says Agency Monitoring Crypto But Not Developing Its Own (#GotBitcoin?)
Wesley Snipes Is Launching A Tokenized $25 Million Movie Fund (#GotBitcoin?)
Mystery 94K BTC Transaction Becomes Richest Non-Exchange Address (#GotBitcoin?)
A Crypto Fix For A Broken International Monetary System (#GotBitcoin?)
Four Out Of Five Top Bitcoin QR Code Generators Are Scams: Report (#GotBitcoin?)
Waves Platform And The Abyss To Jointly Launch Blockchain-Based Games Marketplace (#GotBitcoin?)
Bitmain Ramps Up Power And Efficiency With New Bitcoin Mining Machine (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Miss Finland: Bitcoin’s Risk Keeps Most Women Away From Cryptocurrency (#GotBitcoin?)
Artist Akon Loves BTC And Says, “It’s Controlled By The People” (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Co-Founder Of LinkedIn Presents Crypto Rap Video: Hamilton Vs. Satoshi (#GotBitcoin?)
Crypto Insurance Market To Grow, Lloyd’s Of London And Aon To Lead (#GotBitcoin?)
No ‘AltSeason’ Until Bitcoin Breaks $20K, Says Hedge Fund Manager (#GotBitcoin?)
NSA Working To Develop Quantum-Resistant Cryptocurrency: Report (#GotBitcoin?)
Custody Provider Legacy Trust Launches Crypto Pension Plan (#GotBitcoin?)
Vaneck, SolidX To Offer Limited Bitcoin ETF For Institutions Via Exemption (#GotBitcoin?)
Russell Okung: From NFL Superstar To Bitcoin Educator In 2 Years (#GotBitcoin?)
Bitcoin Miners Made $14 Billion To Date Securing The Network (#GotBitcoin?)
Why Does Amazon Want To Hire Blockchain Experts For Its Ads Division?
Argentina’s Economy Is In A Technical Default (#GotBitcoin?)
Blockchain-Based Fractional Ownership Used To Sell High-End Art (#GotBitcoin?)
Portugal Tax Authority: Bitcoin Trading And Payments Are Tax-Free (#GotBitcoin?)
Bitcoin ‘Failed Safe Haven Test’ After 7% Drop, Peter Schiff Gloats (#GotBitcoin?)
Bitcoin Dev Reveals Multisig UI Teaser For Hardware Wallets, Full Nodes (#GotBitcoin?)
Bitcoin Price: $10K Holds For Now As 50% Of CME Futures Set To Expire (#GotBitcoin?)
Bitcoin Realized Market Cap Hits $100 Billion For The First Time (#GotBitcoin?)
Stablecoins Begin To Look Beyond The Dollar (#GotBitcoin?)
Bank Of England Governor: Libra-Like Currency Could Replace US Dollar (#GotBitcoin?)
Binance Reveals ‘Venus’ — Its Own Project To Rival Facebook’s Libra (#GotBitcoin?)
The Real Benefits Of Blockchain Are Here. They’re Being Ignored (#GotBitcoin?)
CommBank Develops Blockchain Market To Boost Biodiversity (#GotBitcoin?)
SEC Approves Blockchain Tech Startup Securitize To Record Stock Transfers (#GotBitcoin?)
SegWit Creator Introduces New Language For Bitcoin Smart Contracts (#GotBitcoin?)
You Can Now Earn Bitcoin Rewards For Postmates Purchases (#GotBitcoin?)
Bitcoin Price ‘Will Struggle’ In Big Financial Crisis, Says Investor (#GotBitcoin?)
Fidelity Charitable Received Over $100M In Crypto Donations Since 2015 (#GotBitcoin?)
Would Blockchain Better Protect User Data Than FaceApp? Experts Answer (#GotBitcoin?)
Just The Existence Of Bitcoin Impacts Monetary Policy (#GotBitcoin?)
What Are The Biggest Alleged Crypto Heists And How Much Was Stolen? (#GotBitcoin?)
IRS To Cryptocurrency Owners: Come Clean, Or Else!
Coinbase Accidentally Saves Unencrypted Passwords Of 3,420 Customers (#GotBitcoin?)
Bitcoin Is A ‘Chaos Hedge, Or Schmuck Insurance‘ (#GotBitcoin?)
Bakkt Announces September 23 Launch Of Futures And Custody
Coinbase CEO: Institutions Depositing $200-400M Into Crypto Per Week (#GotBitcoin?)
Researchers Find Monero Mining Malware That Hides From Task Manager (#GotBitcoin?)
Crypto Dusting Attack Affects Nearly 300,000 Addresses (#GotBitcoin?)
A Case For Bitcoin As Recession Hedge In A Diversified Investment Portfolio (#GotBitcoin?)
SEC Guidance Gives Ammo To Lawsuit Claiming XRP Is Unregistered Security (#GotBitcoin?)
15 Countries To Develop Crypto Transaction Tracking System: Report (#GotBitcoin?)
US Department Of Commerce Offering 6-Figure Salary To Crypto Expert (#GotBitcoin?)
Mastercard Is Building A Team To Develop Crypto, Wallet Projects (#GotBitcoin?)
Canadian Bitcoin Educator Scams The Scammer And Donates Proceeds (#GotBitcoin?)
Amazon Wants To Build A Blockchain For Ads, New Job Listing Shows (#GotBitcoin?)
Shield Bitcoin Wallets From Theft Via Time Delay (#GotBitcoin?)
Blockstream Launches Bitcoin Mining Farm With Fidelity As Early Customer (#GotBitcoin?)
Commerzbank Tests Blockchain Machine To Machine Payments With Daimler (#GotBitcoin?)
Man Takes Bitcoin Miner Seller To Tribunal Over Electricity Bill And Wins (#GotBitcoin?)
Bitcoin’s Computing Power Sets Record As Over 100K New Miners Go Online (#GotBitcoin?)
Walmart Coin And Libra Perform Major Public Relations For Bitcoin (#GotBitcoin?)
Judge Says Buying Bitcoin Via Credit Card Not Necessarily A Cash Advance (#GotBitcoin?)
Poll: If You’re A Stockowner Or Crypto-Currency Holder. What Will You Do When The Recession Comes?
1 In 5 Crypto Holders Are Women, New Report Reveals (#GotBitcoin?)
Beating Bakkt, Ledgerx Is First To Launch ‘Physical’ Bitcoin Futures In Us (#GotBitcoin?)
Facebook Warns Investors That Libra Stablecoin May Never Launch (#GotBitcoin?)
Government Money Printing Is ‘Rocket Fuel’ For Bitcoin (#GotBitcoin?)
Bitcoin-Friendly Square Cash App Stock Price Up 56% In 2019 (#GotBitcoin?)
Safeway Shoppers Can Now Get Bitcoin Back As Change At 894 US Stores (#GotBitcoin?)
TD Ameritrade CEO: There’s ‘Heightened Interest Again’ With Bitcoin (#GotBitcoin?)
Venezuela Sets New Bitcoin Volume Record Thanks To 10,000,000% Inflation (#GotBitcoin?)
Newegg Adds Bitcoin Payment Option To 73 More Countries (#GotBitcoin?)
China’s Schizophrenic Relationship With Bitcoin (#GotBitcoin?)
More Companies Build Products Around Crypto Hardware Wallets (#GotBitcoin?)
Bakkt Is Scheduled To Start Testing Its Bitcoin Futures Contracts Today (#GotBitcoin?)
Bitcoin Network Now 8 Times More Powerful Than It Was At $20K Price (#GotBitcoin?)
Crypto Exchange BitMEX Under Investigation By CFTC: Bloomberg (#GotBitcoin?)
“Bitcoin An ‘Unstoppable Force,” Says US Congressman At Crypto Hearing (#GotBitcoin?)
Bitcoin Network Is Moving $3 Billion Daily, Up 210% Since April (#GotBitcoin?)
Cryptocurrency Startups Get Partial Green Light From Washington
Fundstrat’s Tom Lee: Bitcoin Pullback Is Healthy, Fewer Searches Аre Good (#GotBitcoin?)
Bitcoin Lightning Nodes Are Snatching Funds From Bad Actors (#GotBitcoin?)
The Provident Bank Now Offers Deposit Services For Crypto-Related Entities (#GotBitcoin?)
Bitcoin Could Help Stop News Censorship From Space (#GotBitcoin?)
US Sanctions On Iran Crypto Mining — Inevitable Or Impossible? (#GotBitcoin?)
US Lawmaker Reintroduces ‘Safe Harbor’ Crypto Tax Bill In Congress (#GotBitcoin?)
EU Central Bank Won’t Add Bitcoin To Reserves — Says It’s Not A Currency (#GotBitcoin?)
The Miami Dolphins Now Accept Bitcoin And Litecoin Crypt-Currency Payments (#GotBitcoin?)
Trump Bashes Bitcoin And Alt-Right Is Mad As Hell (#GotBitcoin?)
Goldman Sachs Ramps Up Development Of New Secret Crypto Project (#GotBitcoin?)
Blockchain And AI Bond, Explained (#GotBitcoin?)
Grayscale Bitcoin Trust Outperformed Indexes In First Half Of 2019 (#GotBitcoin?)
XRP Is The Worst Performing Major Crypto Of 2019 (GotBitcoin?)
Bitcoin Back Near $12K As BTC Shorters Lose $44 Million In One Morning (#GotBitcoin?)
As Deutsche Bank Axes 18K Jobs, Bitcoin Offers A ‘Plan ฿”: VanEck Exec (#GotBitcoin?)
Argentina Drives Global LocalBitcoins Volume To Highest Since November (#GotBitcoin?)
‘I Would Buy’ Bitcoin If Growth Continues — Investment Legend Mobius (#GotBitcoin?)
Lawmakers Push For New Bitcoin Rules (#GotBitcoin?)
Facebook’s Libra Is Bad For African Americans (#GotBitcoin?)
Crypto Firm Charity Announces Alliance To Support Feminine Health (#GotBitcoin?)
Canadian Startup Wants To Upgrade Millions Of ATMs To Sell Bitcoin (#GotBitcoin?)
Trump Says US ‘Should Match’ China’s Money Printing Game (#GotBitcoin?)
Casa Launches Lightning Node Mobile App For Bitcoin Newbies (#GotBitcoin?)
Bitcoin Rally Fuels Market In Crypto Derivatives (#GotBitcoin?)
World’s First Zero-Fiat ‘Bitcoin Bond’ Now Available On Bloomberg Terminal (#GotBitcoin?)
Buying Bitcoin Has Been Profitable 98.2% Of The Days Since Creation (#GotBitcoin?)
Another Crypto Exchange Receives License For Crypto Futures
From ‘Ponzi’ To ‘We’re Working On It’ — BIS Chief Reverses Stance On Crypto (#GotBitcoin?)
These Are The Cities Googling ‘Bitcoin’ As Interest Hits 17-Month High (#GotBitcoin?)
Venezuelan Explains How Bitcoin Saves His Family (#GotBitcoin?)
Quantum Computing Vs. Blockchain: Impact On Cryptography
This Fund Is Riding Bitcoin To Top (#GotBitcoin?)
Bitcoin’s Surge Leaves Smaller Digital Currencies In The Dust (#GotBitcoin?)
Bitcoin Exchange Hits $1 Trillion In Trading Volume (#GotBitcoin?)
Bitcoin Breaks $200 Billion Market Cap For The First Time In 17 Months (#GotBitcoin?)
You Can Now Make State Tax Payments In Bitcoin (#GotBitcoin?)
Religious Organizations Make Ideal Places To Mine Bitcoin (#GotBitcoin?)
Goldman Sacs And JP Morgan Chase Finally Concede To Crypto-Currencies (#GotBitcoin?)
Bitcoin Heading For Fifth Month Of Gains Despite Price Correction (#GotBitcoin?)
Breez Reveals Lightning-Powered Bitcoin Payments App For IPhone (#GotBitcoin?)
Big Four Auditing Firm PwC Releases Cryptocurrency Auditing Software (#GotBitcoin?)
Amazon-Owned Twitch Quietly Brings Back Bitcoin Payments (#GotBitcoin?)
JPMorgan Will Pilot ‘JPM Coin’ Stablecoin By End Of 2019: Report (#GotBitcoin?)
Is There A Big Short In Bitcoin? (#GotBitcoin?)
Coinbase Hit With Outage As Bitcoin Price Drops $1.8K In 15 Minutes
Samourai Wallet Releases Privacy-Enhancing CoinJoin Feature (#GotBitcoin?)
There Are Now More Than 5,000 Bitcoin ATMs Around The World (#GotBitcoin?)
You Can Now Get Bitcoin Rewards When Booking At Hotels.Com (#GotBitcoin?)
North America’s Largest Solar Bitcoin Mining Farm Coming To California (#GotBitcoin?)
Bitcoin On Track For Best Second Quarter Price Gain On Record (#GotBitcoin?)
Bitcoin Hash Rate Climbs To New Record High Boosting Network Security (#GotBitcoin?)
Bitcoin Exceeds 1Million Active Addresses While Coinbase Custodies $1.3B In Assets
Why Bitcoin’s Price Suddenly Surged Back $5K (#GotBitcoin?)
Zebpay Becomes First Exchange To Add Lightning Payments For All Users (#GotBitcoin?)
Coinbase’s New Customer Incentive: Interest Payments, With A Crypto Twist (#GotBitcoin?)
The Best Bitcoin Debit (Cashback) Cards Of 2019 (#GotBitcoin?)
Real Estate Brokerages Now Accepting Bitcoin (#GotBitcoin?)
Ernst & Young Introduces Tax Tool For Reporting Cryptocurrencies (#GotBitcoin?)
Recession Is Looming, or Not. Here’s How To Know (#GotBitcoin?)
How Will Bitcoin Behave During A Recession? (#GotBitcoin?)
Many U.S. Financial Officers Think a Recession Will Hit Next Year (#GotBitcoin?)
Definite Signs of An Imminent Recession (#GotBitcoin?)
What A Recession Could Mean for Women’s Unemployment (#GotBitcoin?)
Investors Run Out of Options As Bitcoin, Stocks, Bonds, Oil Cave To Recession Fears (#GotBitcoin?)
Goldman Is Looking To Reduce “Marcus” Lending Goal On Credit (Recession) Caution (#GotBitcoin?)
Your Questions And Comments Are Greatly Appreciated.
Monty H. & Carolyn A.
Go back


Leave a Reply
You must be logged in to post a comment.