Elon Musk’s Neuralink Shows Off Advances To Brain-Computer Interface
Company putting together submission to the FDA to start testing the technology in humans. Elon Musk’s Neuralink Shows Off Advances To Brain-Computer Interface
Elon Musk and top-level scientists from his neuroscience startup Neuralink Corp., who are developing a next-generation brain-computer interface, unveiled what they billed as a significant advance toward a therapeutic device Tuesday night.
Related:
Next Target For Hackers In 2024 Is Your Brain
Brain Scans Can Reveal Your Decisions 7 Seconds Before You “Decide”
The Botanist Daring To Ask: What If Plants Have Intelligence?
Our Brains Are Quantum Computers. Well, Maybe Not Yours
Scientists Restore Some Brain Function After Death In Animal Experiments
Electro-Magnetic Radiation Reducing Brain Shield
Neuralink is putting together a submission to the U.S. Food and Drug Administration to start testing the technology in humans. The goal is to use the platform to treat neurological conditions like movement disorders, spinal-cord injury and blindness.
“It’s not like suddenly Neuralink will have this neural lace and take over people’s brain,” said Mr. Musk. He also mentioned the announcement was meant to recruit talent to the company, which has about 100 employees.
The company said it is first focusing on patients with severe neurological conditions, but wants to make it safe enough to turn the implantation surgery into an elective procedure, like Lasik.
“We hope we’re less than a year from the first safety study on the order of five patients,” said Neuralink President Max Hodak in an interview. He emphasized that it could take years before the device could help a range of patients. “The road is long,” he said.
Among the most important tests: showing they can monitor brain activity and then decode it, meaning they can correlate certain patterns of activity to actions, such as movement, vision or speech. The company didn’t specify what behavioral experiments were performed or how reliably they were able to translate brain activity into smooth, well-controlled movement.
The device has been tested on monkeys, according to Mr. Musk. The primate was able to control a computer with its brain, he said in a surprise announcement during a question and answer session. He didn’t provide any other details. A company spokeswoman confirmed the experiment had been done.
Neuralink is one of several companies, including Facebook Inc., Kernel, CTRL-Labs and Paradromics Inc., trying to build neural interfaces for clinical and nonclinical applications. In recent years, neurotechnology development has been spurred by public and private investment, including the U.S. Brain Initiative, which was started by the Obama administration in 2013.
The goal of many of these projects is to access as many neurons as possible because that would give scientists more precise reads on activity that underpins walking, speech and mood, among other brain functions.
They can then turn neural recordings into electrical signals that can be fed into a robotic device or back into the nervous system to produce movement or vision to help patients, according to experts.
At the event Tuesday in San Francisco, Neuralink described a tiny probe with nearly 3,100 electrodes laid out across about 100 flexible wires, or threads, each individually inserted into rat brains by a custom-made surgical robot. The device can monitor the activity of upward of 1,000 neurons at a time, according to the company.
The sewing-machine-like robot can target very specific brain areas, helping surgeons avoid major blood vessels—an important consideration for minimizing inflammation and long-term damage, according to a paper from the company. Data were processed and analyzed by proprietary chips and software.
Neuroscientists and neurotechnologists said that a platform that can insert tiny electrodes robotically throughout the brain and then analyze activity with custom software is exciting, but cautioned it is too early to tell how quickly Neuralink’s device could safely be used in patients.
“If you’re trying to walk yourself toward human prosthetics, this is a more promising direction than currently available technology,” said Tim Harris, a senior fellow at the Howard Hughes Medical Institute’s Janelia Research Campus and a developer of research-grade neural interfaces. Among the questions that are left unanswered by the company’s paper, he said, is how long it lasts in the brain.
“If you’re going to do this for people, you should be aiming for at least five years, minimum,” he said. “To do an implantation surgery of this level of intricacy, a year or two is not enough.”
The paper, which wasn’t peer-reviewed, didn’t include data on the long-term stability of recorded neural signals nor the brain’s inflammatory response.
“That is utterly critical” before any device can advance to human trials, said Loren Frank, a University of California, San Francisco neuroscientist developing brain-computer interfaces.
Neuralink has said it is doing those experiments but isn’t ready to make the data public.
The device, in theory, was designed to also stimulate brain cells, but “we have not demonstrated these capabilities here,” according to the paper. Direct brain stimulation with implanted electrodes is a longstanding approach to treating movement disorders and epilepsy.
Most brain-computer interfaces are so-called open-looped systems that don’t adapt to a patient’s needs and experience. Neurosurgeons and technologists have pointed to that drawback as reason why brain stimulation hasn’t worked for treating mood disorders.
The advantage of a system like Neuralink envisions would be its ability to analyze recordings using machine learning and to adapt the type of stimulation it delivers to a patient’s brain, according to the company and other experts.
Because of Mr. Musk, Neuralink has perhaps the highest profile among startups developing brain-computer interface technology.
But in the past couple of years, nearly all startups in the sector have seen a considerable boost in investor and regulator interest. Since 2016, startups including Paradromics and CTRL Labs have collectively raised around $260 million from a mix of venture capital, grants and corporate investors.
In February, the FDA released guidelines for regulating brain-computer interface technology, in hopes of spurring faster development of devices.
Dolby Family Ventures managing director David Dolby, whose firm has backed Paradromics, said as regulators are involved and more startups emerge, the time is right for the private sector to push brain-computer interface technology into the next stages of commercialization.
“There are a multitude of applications for this technology. Through open market competition, I think we will learn a lot and benefit as a society,” said Mr. Dolby.
Not all neurotech investors are convinced implantable devices are the way forward. Lux Capital co-founder and managing partner Joshua Wolfe is in investor in CTRL Labs, which is developing sensor-based technology to decode nerve signals, but said he isn’t yet comfortable with invasive devices such as those developed by Neuralink and Paradromics.
“There is no way I’m thinking about technology that involves drilling holes behind ears right now,” said Mr. Wolfe.
Enke Bashllari, a neuroscientist by training who now heads venture-capital firm Arkitekt Ventures, agreed there are considerable safety measures that must be ensured with implantable technology and said she believes that noninvasive devices also have a valuable role to play in augmenting human movement or cognitive performance. But she said the highest unmet medical needs will require technology that goes inside.
“It has to allow for two main things—high spatial resolution and high bandwidth. It has to interface with millions of neurons at the same time and you need to know exactly which neuron is firing,” she said. “That can currently only be done invasively.”
Elon Musk’s AI Project To Replicate The Human Brain Receives $1 Billion From MIcrosoft
‘The creation of AGI will be the most important technological development in human history,’ says OpenAI boss.
Microsoft has invested $1 billion in the Elon Musk-founded artificial intelligence venture that plans to mimic the human brain using computers.
OpenAI said the investment would go towards its efforts of building artificial general intelligence (AGI) that can rival and surpass the cognitive capabilities of humans.
“The creation of AGI will be the most important technological development in human history, with the potential to shape the trajectory of humanity,” said OpenAI CEO Sam Altman.
“Our mission is to ensure that AGI technology benefits all of humanity, and we’re working with Microsoft to build the supercomputing foundation on which we’ll build AGI.”
The two firms will jointly build AI supercomputing technologies, which OpenAI plans to commercialise through Microsoft and its Azure cloud computing business.
OpenAI was founded in 2015 with the goal of developing AGI that can learn and master several disciplines, rather than the narrow abilities of most modern artificial intelligence systems.
The startup has already achieved a number of AI milestones, most notably beating the world’s best human players at the video game Dota 2, but hopes its technology can one day help address climate change and other major challenges facing the planet.
“An AGI working on a problem would be able to see connections across disciplines that no human could,” OpenAI CTO Greg Brockman wrote in a blog post announcing the investment.
“We want AGI to work with people to solve currently intractable multi-disciplinary problems, including global challenges such as climate change, affordable and high-quality healthcare, and personalised education.”
OpenAI also claims its technology will ultimately provide everyone with the economic freedom to pursue whatever they find most fulfilling, while creating “new opportunities for all our lives that are unimaginable today”.
Since co-founding OpenAI three years ago, Mr Musk has since stepped back from the AI startup but remains vocal about the risks artificial intelligence poses to humanity, claiming its development poses a greater risk than nuclear weapons.
In 2017, the SpaceX and Tesla CEO joined Microsoft researchers in signing an open letter outlining principles that will ensure the development of AI that is beneficial to humanity.
“We cannot predict what we might achieve when this intelligence is magnified by the tools AI may provide, but the eradication of disease and poverty are not unfathomable,” the letter stated.
“Because of the great potential of AI, it is important to research how to reap its benefits while avoiding potential pitfalls.”
Updated: 5-20-2021
Billionaire Peter Thiel Backs Fundraiser of Brain-Computer Firm
Billionaire investors Peter Thiel and Christian Angermayer have thrown their weight behind a technology startup that promises to help neurologically impaired patients regain lost skills with the aid of electrodes connecting the human brain with computers.
The Utah-based company, Blackrock Neurotech, was granted $10 million from the investors to expand its clinical studies and research capabilities, it said in a statement Wednesday. There’s no connection with BlackRock Inc., the fund manager.
Founded by German duo Marcus Gerhardt and Florian Solzbacher in 2008, the company makes so-called brain-computer interfaces, or BCI. The devices are akin to mini-computers equipped with electrodes that are implanted in different regions of the brain, where they pick up signals — similar to thoughts — and convert them into electrical signals.
The devices can help patients regain their eyesight or the ability to hear and talk again. In some of the most high-profile cases, the technology enabled fully paralyzed patients to either control robotic prosthesis directly with their brain or move their own limbs again.
In another breakthrough, the technology allows patients suffering from amyotrophic lateral sclerosis, or ALS, even in a fully locked-in state to communicate via an auditory speller controlled by their mind.
Blackrock Neurotech operates at the forefront of a sector that’s still in its infancy. Of the just 30 patients worldwide using the technology, 28 have worked with the Salt Lake City-based company. Elon Musk’s startup Neuralink recently unveiled a BCI device implanted in a monkey’s brain.
While the devices for these patients are all individualized, Blackrock Neurotech aims to use some of the funds to ramp up production and offer its tools to patients worldwide, Gerhardt said in an interview.
“Enabling people to walk, talk, see, hear and feel again is already a massive market, as more than 5 million people in the U.S. are living with some form of paralysis,” investor Angermayer said in a statement.
Forays into connecting human brains with computers have increasingly attracted funds in the past years. Bryan Johnson, founder of the payments service Braintree, has committed $100 million to a BCI startup called Kernel. Facebook Inc. is developing a skullcap it says will allow users to mentally type their thoughts at 100 words per minute.
Thiel and Angermayer have co-invested in a range of new frontier ventures, including psychedelic startup Atai Life Sciences AG or Lithium explorer Rock Tech Lithium Inc.
Updated: 7-22-2021
Neuralink Competitor Raises $20 Million For Brain Implants
Paradromics is one of several companies working on technology to help people interact with the outside world using their mind.
A competitor to Elon Musk’s Neuralink said it raised $20 million, enough to get it on track to introduce a new generation of powerful and tiny electrodes to the human brain.
Paradromics Inc., founded in 2015 about a year ahead of Neuralink, is building brain-computer interfaces that could help people with disorders ranging from paralysis to speech impediments.
“Once you start to realize that the best way to describe the brain is through data, you start to reframe a lot of classically hard-to-treat conditions,” said Matt Angle, chief executive officer of Paradromics. “What the brain really is—it’s a data system.”
For example, where a biological approach to blindness might be to attempt to regrow the retina, his approach would be to get visual data to the right part of the brain, even if it means using a computer.
The funding will help Paradromics hone its hardware, Connexus, tiny 8-millimeter square modules that get implanted under the skull, into the surface of the brain cortex.
Microwires cover the modules, penetrating an additional 1.5 millimeter into the cortex, and translate the brain’s bioelectric signals into digital ones that can be understood by a computer, and vice versa.
The modules transmit data to and from a fifth hub module planted in the skull, which in turn transmits the data to a sixth module just under the skin of the chest, and then wirelessly to a nearby computer, small enough to clip onto a wheelchair.
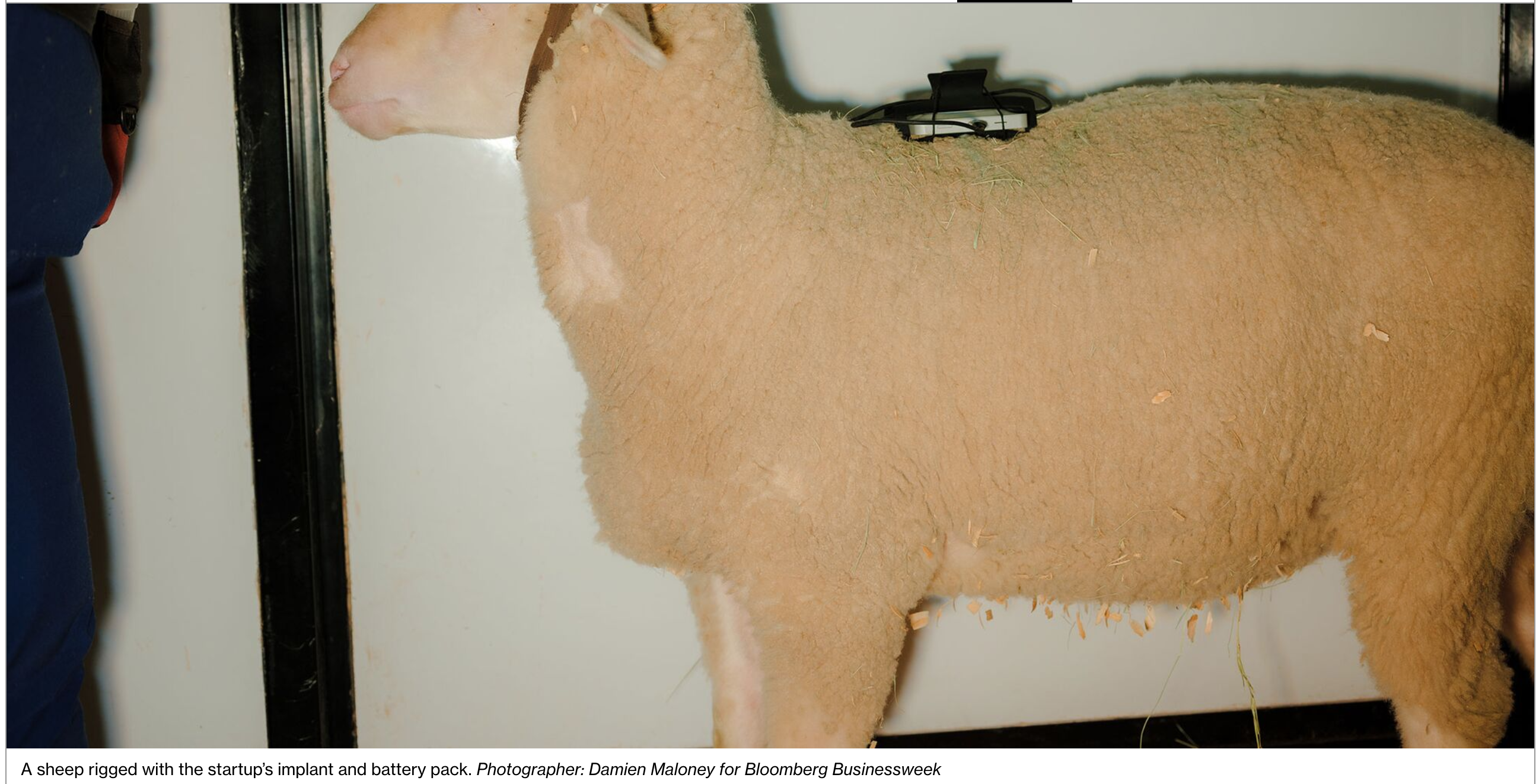
In that manner, the brain’s activity can be translated into an actionable command, such as the movement of a computer cursor. It’s still early days for the system, but so far, a predecessor technology has been tested successfully on sheep.
Paradromics, like other companies in its field, said it stays in touch with regulators to help smooth the process of applying for experimental use in humans. But first, it needs to fine-tune its hardware. Angle expects the devices to be ready to start the application process next year.
Paradromics’ technology builds on decades of research and work by initiatives such as BrainGate, and is one several companies in the field. So far this year, brain-machine interface startups have raised $132.8 million, according to PitchBook.
That’s already one-third more than the sector raised in all of last year, and by far the most since 2017, a year when Neuralink raised $107 million.
Paradromics said one of its advantages comes from the number of electrodes—400—that sit on each of its modules, more than on analogous devices. That will improve the quality and quantity of the data they can handle, the startup said.
“People are using the devices they have access to right now, and trying to push the envelope,” said Amy Kruse, a former program manager for neuroscience at the Defense Advanced Research Projects Agency who is now a partner at Prime Movers Lab, which led the investment. “How much data you can get out? Where Paradromics is headed, and in particular around those interfaces, is around high-bandwidth, high-throughput devices.”
Of course, any such device is risky, and brain surgery is an inherently fraught process. That said, several hundred thousand people globally already have devices implanted in their skulls, mostly to control seizures and tremors.
Kruse said Musk’s interest in neuroscience and in particular the work at Neuralink—which last year showed off pigs with devices implanted in their brains—has encouraged more venture capitalists to look at the field of brain-computer interfaces.
“What Elon has done, he’s normalized neurotech for a group of investors that otherwise wouldn’t have touched it,” Angle said.
Neuralink has signaled it will boost its presence in Austin, Paradromics’ home turf. But Angle said he’s not worried, and describes his relationship with Musk’s company as “collegial.”
In fact, Angle believes having two major brain-computer interface, or BCI, companies in close proximity will help establish the city as the center for similar start-ups.
“The markets in which we’re involved are hundreds of billions of dollars,” Angle said. “In the case of BCI, we don’t have enough BCI companies to service all of the possible use cases.”
The funding round includes participation from Westcott Investment Group, Dolby Family Ventures, Synergy Ventures and others. Prime Movers’ Kruse has taken a board seat.
Updated: 1-20-2022
Elon Musk’s Brain Implant Company Is Inching Toward Human Trials
Neuralink has posted job listings for a clinical trial director and coordinator.
Elon Musk’s brain implant company Neuralink is now hiring a clinical trial director, an indication that the company’s longstanding goal of implanting chips in human brains is coming closer.
The trial director position would oversee the startup’s long-promised human trials of its medical device, according to the listing. Neuralink’s brain implant — which Musk has said already allows monkeys to play video games with their thoughts alone — is intended to help treat a variety of neurological disorders, such as paralysis.
The job description for the position, based in Fremont, California, promises that the applicant will “work closely with some of the most innovative doctors and top engineers” as well as with “Neuralink’s first Clinical Trial participants.”
It also indicates that the job will mean leading and building “the team responsible for enabling Neuralink’s clinical research activities,” as well as adhering to regulations.
Last month, Musk told the Wall Street Journal that Neuralink hoped to implant its device in human brains sometime in 2022. However, he has made similar predictions in the past that proved overly optimistic — including in 2019, when he said during a presentation that the device would be in human skulls by the following year.
The first in-human test that medical device makers must go through on the path to U.S. Food and Drug Administration approval is known as a feasibility test. The next, after a submission, review and approval process with the FDA based on the feasibility test, is a pivotal device test.
It isn’t clear precisely where Neuralink is in the process. Representatives for Neuralink and the FDA didn’t respond to requests for comment. Generally, device makers hire trial directors early in their interaction with the FDA, to help design trials in a way that would maximize the chances of FDA approval.
Neuralink doesn’t appear to have human trials posted on clinicaltrials.gov, but postings there can lag recruitment of the first patients.
Last year, another company working on brain-machine interfaces, Synchron, said the FDA had approved its planned feasibility study. Synchron is currently in the recruiting stage, a spokeswoman said.
Neuralink is also advertising for a Fremont-based clinical trial coordinator, as well as several other positions.
Updated: 2-4-2022
Ex-Neuralink President Invests In Competing Brain Startup
Max Hodak co-founded Neuralink alongside Elon Musk.
New York-based Synchron Inc.—a competitor to Elon Musk’s brain-machine interface company, Neuralink—has won financial backing from Neuralink’s co-founder and former president.
Max Hodak, who left Neuralink last year, has invested in Synchron and joined its advisory board, he wrote in a blog post on Friday. He did not say how much he was investing.
“I jumped at the chance to work with them,” he wrote, saying the company’s technology could benefit tens of millions of patients. Synchron aims to treat conditions like paralysis by inserting its Stentrode implant in human brains.
That is similar to Neuralink, which also plans to use implanted devices to help treat paralysis and similar conditions.
Unlike Neuralink, whose device will be implanted by drilling into skulls, Synchron to implant its device via blood vessels, which Hodak called “a different, really quite elegant idea.”
Synchron has already tried the Stentrode in four patients and has received Food and Drug Administration permission to launch a clinical trial in the United States. That puts it far ahead of Neuralink, which is now recruiting a trial director.
Brain computer interfaces have become a hot investor area. Halfway through last year, startups in the field had already raised more cash than they did in all of 2020, according to PitchBook.
Neuralink, which raised $205 million last year, is the best-capitalized, but plenty of others exist, like Paradromics Inc. and Precision Neuroscience Corp., as well as Synchron.
Hodak became president of Neuralink in 2017. When he left, “it wasn’t my choice,” he wrote in a September blog post. “There was a lot left there I still wanted to do. But as usual, Elon was right: it was time for me to go.”
He raised at least $47 million for a new neuroscience venture, Science Corp., according to a July filing with the SEC.
“I really don’t want this to be construed as a knock on Neuralink,” Hodak said in an email, adding that he has not sold any of his Neuralink stock. “I’m sure they will also get into humans soon too,” he wrote, noting that Synchron is an older company. “There are many different ideas worth trying in this space.”
While it is unusual for an executive to leave one company and invest in a rival, a long history exists of startup executives forming or joining competing companies.
Silicon Valley was born after the so-called “Traitorous Eight” quit Shockley Semiconductor Laboratory to found Fairchild Semiconductor in 1957. Decades later with tech’s cutting edge dominated by software, marketing executive Whitney Wolfe Herd left Tinder and founded competing dating site Bumble in 2014.
Hodak gave an implicit acknowledgment of the rivalry toward the end of his blog post. “Engineering the brain is such a profoundly powerful concept that I expect there to be multiple trillion-dollar companies to emerge in this space as it matures,” he wrote.
Updated: 11-21-2022
Ex-President of Elon Musk’s Neuralink Has New Brain-Computer Startup
With $160 million, Science Corp. is second only to Musk’s startup in total funding.
At Elon Musk’s Neuralink Corp., Max Hodak helped lead development of a computing device designed to be inserted into a person’s brain. Now he has a competing startup with $160 million in funding that seeks to do something even more striking: manipulate the brain without any in-skull implants.
Hodak’s new company Science Corp. is working on a brain-computer interface that’s in some ways similar to Neuralink’s but using a physical science called photonics. The idea is to use light and the eye’s optic nerve as a pathway into the brain, rather than implant chips deep within it.
Scientists and companies have experimented with photonics for years in the hope of restoring patients’ vision, but efforts to make brain-machine interfaces using the optic nerve have fallen short.
Hodak, the former president of Neuralink, has been operating Science in stealth mode since last year. He envisions making a variety of brain-augmenting products beginning with vision. The startup built a prosthetic device, Science Eye, that it says works in rabbits. In the next year or two, Science hopes to try it in humans.
The machine is intended to treat retinitis pigmentosa, a disease affecting peripheral vision, and macular degeneration, which affects central vision.
“We’re starting in very disabled patient populations with serious unmet needs,” said Hodak, whose grandfather suffered from retinitis pigmentosa. “But if you refine that technology five or six generations, you get to replace glasses and [virtual reality] googles with just the tiny little implant in the eye.”
The philosophy is similar to one outlined by Musk. If the technology can address disabilities, then eventually healthy people may be willing to try it.
The future goal would be to create a new sort of brain operating system that could, for example, project turn-by-turn directions onto the eyeball or conjure more immersive video games.
Whether any of this would actually happen is unknown. The risk of opening one’s brain to hackers may be enough to scare away the general population.
For now, Hodak said he’s “focused on pragmatic, near-term things.” Science Eye is based on a 2-millimeter wide, very thin LED film that gets implanted on top of the retina, behind the eyelid.
The surgical procedure lasts two hours, but Science said it can eventually be done in half that time. The LED film processes patterns sent wirelessly from glasses embedded with tiny cameras, which convert images into a form that the optical nerve can read.
For the device to function, the cells of the optic nerve must become light sensitive. Using gene therapy, Science delivers an engineered protein via injection that alters the optic nerve cells.
By targeting individual cells rather than groups of cells, Science said it can achieve a much higher resolution compared with other cutting-edge approaches to treating blindness or eye disease.
But getting it to work in a rabbit is a far step from doing so in a person. The main way Science knows it is succeeding is by measuring activity in the rabbits’ visual cortexes, the part of the brain that handles sight.
One of the rabbits, Lela, sat patiently in her quarters when a reporter visited the Science headquarters in Alameda, Calif., this month. A small metal plate was attached on her forehead, under fluffy, white ears.
As the reporter approached, Lela and some of her bunny colleagues hopped to the front of their hutches to observe.
Science’s technology will eventually be able to read from and write to the brain at the same time but for now is focusing on the writing part. Hodak said he has discussed Science Eye in a preliminary conversation with the US Food and Drug Administration.
Although the company’s technology is unproven in humans, photonics would solve a major flaw of existing brain-computer interfaces. Widespread adoption of such a machine is seen as unlikely if users are required to have holes drilled in their skulls.
Finding a way to stimulate targeted sections of the brain in humans without implanting an electrode, though, has long eluded scientists. A practical application would require significant biological breakthroughs, Hodak acknowledged.
Still, he said he became convinced of the viability of photonics after working at Neuralink, which he left abruptly last year. He declined to comment on his time there or his departure from the company.
He is still a shareholder in Neuralink, he said, as well as another competing startup, Synchron. Both are pursuing electrode-based systems.
Hodak runs Science with his co-founders, the biologist Alan Mardinly, microfabrication director Yifan Kong, engineer Corey Wolin and software lead Emma Zhou. The startup’s funding haul of $160 million is second in the field only to Neuralink.
Among Science’s backers are founders of the crypto companies Ripple Labs, Protocol Labs and Paradigm as well as the former GV partner Blake Byers. Artis Ventures is also an investor.
Updated: 12-1-2022
Elon Musk Says Brain Implant Startup Neuralink Should Be Ready For Human Testing In Six Months
Neuroscience company also showcases ‘telepathic typing’ in monkey with brain device, other capabilities, in live-streamed update.
Elon Musk’s startup Neuralink Corp. should be ready to test its technology on humans in six months, the entrepreneur said Wednesday during a live-streamed update about progress the company has made with its brain-implant technology.
Neuralink has submitted most of its paperwork to the U.S. Food and Drug Administration, which oversees medical devices, including neural implants, Mr. Musk said. In 2019, he said the company planned to seek the FDA’s approval for human testing and predicted it could begin as soon as 2020.
“We are now confident that the Neuralink device is ready for humans, so timing is a function of working through the FDA approval process,” Mr. Musk said in a tweet during the event.
During a question-and-answer session, the company said it was working to address concerns from the FDA regarding overheating of the device and also toxic chemicals seeping into the brain from the implant, both of which could cause damage.
Mr. Musk said the implants are compact and that he would feel comfortable getting one now. He teased the possibility that he will eventually have an implant for a future demonstration.
“I could have a Neuralink device implanted right now and you wouldn’t even know, hypothetically,” he said.
The company wants to be able to help restore vision and enable people with severe disabilities to move and communicate by decoding brain activity.
Eventually, Neuralink wants to open clinics where patients could get a device implanted into their brains by their surgical robots, which the company also showcased at the Wednesday event.
The robot surgeon threads Neuralink’s tiny proprietary electrodes, or brain-signal recording wires, into the brain.
Mr. Musk showed a video of “telepathic typing” from a monkey that has a Neuralink brain implant. The animal wasn’t typing into a keyboard but was able to move a cursor to images of letters.
“He’s moving the cursor with his mind,” he said. “He can’t actually spell. I don’t want to oversell this thing.”
Neuralink has been testing its implant technology on nonhuman primates for several years, including in April 2021, when the company released a video showing that a monkey implanted with two Neuralink devices could play a videogame called Pong as the device translated its brain activity into commands with the help of machine-learning software.
Clinical testing that proves an implantable device is safe and effective long-term would be necessary before a brain-computer interface, like Neuralink’s, could be widely rolled out to patients, neurotechnology experts said.
“That’s a big challenge,” said Sumner Norman, chief neuroscientist at AE Studio, a development agency with a dedicated team of brain-computer interface engineers.
In the past year, Neuralink has been challenged with safety concerns after several of its monkeys had to be euthanized, including for suspected device-associated infections. It is standard practice in pharmaceutical and medical-device development to do preclinical testing on animals.
“We’re not cavalier about putting devices into animals,” Mr. Musk said Wednesday.
Other researchers have managed to use a brain-computer interface to enable monkeys to produce words on a computer screen.
Last year, University of California, San Francisco, researchers used an experimental brain implant to translate a patient’s brain signals into words on a screen.
Synchron Inc., a neurotech company developing a brain implant that is threaded into the brain’s blood vessels instead of interacting with brain tissue itself, has allowed five patients to type, according to Tom Oxley, the company’s chief executive.
Advancements in artificial intelligence, the miniaturization of electronics and new surgical techniques have sparked a flurry of investment and development into brain-computer interfaces, according to Michael Mager, chief executive of Precision Neuroscience Corp., which is working on a film-like sensor that sits on top of the brain and is inserted through a tiny slit in the skull.
Several companies are going after restoring sight, including Neuralink.
Science Corp.—founded by Max Hodak, formerly president of Neuralink—aims to use a combination of gene therapy, a thin retinal implant and smart glasses to help restore vision. Blackrock Neurotech recently announced it was developing an ultrathin, flexible implant called Neuralace for that same purpose, according to Florian Solzbacher, the company’s co-founder and president.
The technology would take electrodes, often called channels, and deliver into the visual system thousands of pulses of electricity to create the perception of seeing objects, Dr. Solzbacher said.
“For that, you need thousands of channels, and the more the better,” Dr. Solzbacher said.
So far, researchers have been able to re-create the perception of edges and other simple shapes—basically the equivalent of an electronic walking stick, Dr. Solzbacher added—but nothing that approximates what we would interpret as full vision. He said he hoped the device would be available to researchers in 2024.
“The challenge is building something that is both high-channel count and can last for a long time in the body,” said Matt Angle, chief executive of neurotech firm Paradromics Inc.
Most neural implants to date have been one-way highways of information, taking signals the brain generates and translating them into commands that an external device like a computer can then use to “type” or play videogames.
But developers hope the next generation of devices will be able to shuttle information into and out of the brain, for instance, to restore our senses and help with movement. Mr. Musk made several references to this “read and write” capability Wednesday.
Precision Neuroscience is working on a device that can both record from the brain and send signals into it. In January, the company said its implant could record from and stimulate areas of the brain involved in vision and the perception of touch.
During the presentation, Neuralink said it had implanted its device into the visual cortex of two monkeys already and used it to record brain activity and to create the perception of seeing tiny spots of light.
The ability of neural implants to “create the perception of touch or vision—that would be a huge win,” said Dr. Norman of AE Studio.
These next-generation devices augment some of the ethical concerns about brain implants. Some have called them the last frontier of human privacy.
“People identify their sense of self most closely with their brains,” said Nita Farahany, a Duke University neuroethicist. “Writing to the brain has the great peril to manipulate and override our choices and preferences.”
Updated: 5-6-2023
Elon Musk’s Neuralink Says It Has FDA Approval For Study Of Brain Implants In Humans
The startup tested its technology on primates for several years.
Elon Musk’s brain-implant startup Neuralink said it has been approved by the U.S. Food and Drug Administration to begin the company’s first human clinical study.
The company is working on a device that will allow people to control computer interfaces with their brains. Neuralink said the devices would help restore vision and enable people with severe disabilities to move and communicate by decoding brain activity.
Neuralink plans to open clinics where surgical robots will implant the devices into the brains of patients. The robot surgeon threads Neuralink’s proprietary electrodes, or brain-signal recording wires, into the brain.
“We are excited to share that we have received the FDA’s approval to launch our first-in-human clinical study,” Neuralink said in a tweet. “This is the result of incredible work by the Neuralink team in close collaboration with the FDA and represents an important first step that will one day allow our technology to help many people.”
The FDA acknowledged Neuralink’s announcement.
The company said it hasn’t opened recruitment yet for its trial but will announce more information soon.
Musk said in 2019 that the company planned to apply for FDA approval for human testing. At the time he predicted the approval would be in place as soon as 2020.
Neuralink has tested its implant technology on primates for several years. The company released a video in April 2021 showing a monkey implanted with two Neuralink devices could play a videogame called Pong using only his brain.
During a demonstration in December, Musk showed a video of what he called “telepathic typing” from a monkey with a Neuralink brain implant. The animal used his mind to move a cursor to images of letters during the demonstration.
Clinical testing proving the Neuralink device is safe and effective long-term would be necessary before a brain-computer interface could be introduced to patients, neurotechnology experts have said.
Musk said in December the company was working to address concerns from the FDA regarding overheating of the device and toxic chemicals seeping into the brain from the implant. Neuralink has euthanized several of its monkeys for suspected device-associated infections, the company said in February 2022.
Neuralink said device-associated infections are an inherent risk with any medical device that is implanted through the skin. The company later developed new surgical protocols.
Updated: 8-7-2023
Elon Musk’s Neuralink Raises $280 Million To Develop Brain Implants
The funding round is the latest cash influx for brain technology startups.
Neuralink Corp., Elon Musk’s brain-implant company, has raised $280 million in new funding from investors to develop its technology.
The startup announced the funding round in a post on Musk’s X social network, formerly known as Twitter. The deal was led by Founders Fund, a venture capital firm backed by billionaire Peter Thiel.
Neuralink is the best-known player in a growing field of brain technology companies. Partly spurred by Neuralink’s high profile, investors have backed dozens of other startups exploring similar technology.
Startups in the neurotechnology field raised $143 million this year through June 26, according to PitchBook data. That compares to $278.1 million in all of 2022 and $518.9 million in 2021. One rival, Synchron Inc., enrolled its first patient in a US clinical trial last year, beating Neuralink to that milestone.
Neuralink recently received approval from the US Food and Drug Administration to conduct human clinical trials. The company is developing a small device that uses electrode-laced wires to link the brain to a computer. Placing the device requires drilling into the skull.
Musk’s startup has previously raised concerns within animal rights groups for its testing on primates. Earlier this year, the US Transportation Department began an investigation into the company after an animal advocacy organization said it obtained emails suggesting the startup did not follow proper procedure when shipping possibly hazardous materials.
Updated: 11-6-2023
Elon Musk’s Brain Implant Startup Is Ready To Start Surgery
Neuralink has FDA clearance to start messing with people’s heads—and not just figuratively.
Elon Musk is preparing for the most consequential launch of his career. But this one isn’t rocket science—it’s brain surgery. Musk’s company Neuralink Corp. is seeking a volunteer for its first clinical trial, meaning it’s looking for someone willing to have a chunk of their skull removed by a surgeon so a large robot can insert a series of electrodes and superthin wires into their brain.
When the robot finishes, the missing piece of skull will have been replaced with a computer the size of a quarter that’s meant to stay there for years. Its job will be to read and analyze the person’s brain activity, then relay that information wirelessly to a nearby laptop or tablet.
For the purposes of the trial, an ideal candidate would be an adult under age 40 whose four limbs are paralyzed. Such a patient would likely have Neuralink’s implant inserted into what’s known as the hand knob area of their premotor cortex, which governs the hands, wrists and forearms.
The goal is to show that the device can safely collect useful data from that part of the patient’s brain, a key step in Neuralink’s efforts to convert a person’s thoughts into a range of commands a computer can understand.
Several companies and research teams have already created implants that can help patients perform basic tasks with their thoughts, such as clicking objects on a screen with a cursor. Neuralink, in familiar Musk fashion, has issued far wilder promises.
For the past four years, starting with the company’s first public demonstration, he has made it sound as if there would soon be ubiquitous clinics where anyone could go in for a 15-minute robosurgery and come out a human-machine hybrid.
These cyborgs would be able to download knowledge the way Keanu Reeves does in The Matrix or upload their thoughts into storage, even to other brains. “This is going to sound pretty weird, but ultimately we will achieve symbiosis with artificial intelligence,” Musk said at that first demo in 2019, when the company said human trials could begin in 2020.
Unrealistic timetables are one of Musk’s favorite management techniques. To his credit, he’s made several improbable dreams come true—eventually.
But while rockets and cars are serious business, neural implants require perfection on a whole other level. One does not rush a brain implant to market and hope for the best.
Two other companies, Synchron and Onward, have more than a year’s head start on human trials with brain implants and related technology.
Neuralink has, however, gotten dramatically more attention than the decades of incremental, largely academic research that preceded it, and not always to its credit.
Some neuroscientists have said Neuralink is hyping the technology. Animal-rights groups have accused it of cruelty to the monkeys, pigs and other mammals it’s tested implants on so far.
The through-line is Musk, whose increasingly manic and reactionary online persona has done little to suggest he stands as the ideal candidate to mass-produce mind-control devices.
All these concerns are valid. Yet Neuralink’s trial is exciting, too. The company appears to have turbocharged progress in this slow-and-steady field, and it’s now built the world’s most powerful and elegant brain implant.
If the product works as intended, later iterations could improve, in miraculous ways, the life of millions of people suffering from paralysis, stroke, Lou Gehrig’s disease, and hearing and vision loss.
In the meantime, its high profile already has investors hunting for the next Neuralink. Once again, Musk has reshaped an entire industry, and this one could be the most transformative of all.
In the past three years, I’ve made 10 trips to Neuralink’s facilities in Silicon Valley and its growing operations in Austin. In tandem with Musk’s impatient demands, I’ve seen his team profoundly advance their technology and ambitions.
As they prepare for the trial, the pressure to succeed is something even Musk hasn’t seen before.
Tesla Inc., after all, took many years to mass-produce its cars, and SpaceX’s first three rockets exploded. When it comes to brains, “We can’t blow up the first three,” says Shivon Zilis, Neuralink’s director of special projects. “That’s not an option here.”
The modern history of brain implants began with the technological advances of the 1990s. The science, simplifying tremendously: Thoughts cause neurons to fire in particular patterns, and these patterns have some degree of consistency across brains.
In fact, roughly the same neurons fire when a person thinks about moving their arms and fingers, whether they can physically move them or not.
Brains light up in similar, consistent ways when people want to move a mouse cursor to click somewhere on a computer screen, too.
The same is true for speech: If you can think of saying a letter or word, you’re making the same neurons fire as you would by physically speaking it.
Even if you can’t speak then, a well-trained computer should be able to discern your intent and, theoretically, speak for you.
The challenge is figuring out each entry in the neurons-to-English dictionary, which requires gathering and studying tons of data about the patterns of many people’s brain signals. To get the clearest signals, you want to place sensors as close to the neurons as possible.
Some researchers have tried to avoid surgery by keeping their devices outside a person’s skull, but the added distance and interference have yielded muted results. The most precise data usually comes from electrodes sitting right beside brain cells.
“The long-term goal is to have this available for billions of people and unlock human potential and go beyond our biological capabilities”
For most of the past 20 years, the Utah array has been the implant to beat. It’s a tiny, flat square of silicon that could fit atop a child’s fingernail. Wires protrude from its edge, and on one face of the chip is a bed of about 100 rigid spikes.
To implant the Utah array, a surgeon must perform a craniotomy, cutting a large hole in the patient’s skull, then gently hammer the tiny spikes into the brain.
The wires are positioned to connect to a metal port that visibly pokes out of the scalp after the opening is sutured shut. Post-op, to use the device, an ice-cube-size computer is attached to the patient’s head.
Researchers have made major advances with Utah arrays. They use them to read and translate the brain activity of people with paralysis and other conditions.
Software created with this information allows patients to communicate with caregivers and loved ones, or to manipulate robotic arms to pick up objects.
The catch is the hardware’s clunky design, which has remained largely unchanged over 20 years. The arrays also need a raft of computers and other equipment operated by trained personnel and lots of medical care, which has kept them confined mostly to research labs.
Musk co-founded Neuralink in 2016 with seven scientists and $100 million of his own money. The splashiness of his investment, and his grand promises about the underpinnings of the technology, proved irresistible to venture capitalists.
Neuralink has since raised more than $500 million, including $280 million this year, and the attention has helped draw investors to other brain-computer interface efforts, including long-standing university projects as well as newer startups.
Last year, 37 such companies raised more than $560 million, according to the research company PitchBook.
Most of these enterprises have the same primary objective: Build a brain-scanning device that can leave the lab behind. The ideal implant will have plenty of computing oomph to record and input lots of data and also to transmit the data via strong wireless signals.
This must all be done while using as little battery power as possible and without running too hot, which could irritate or injure a patient.
Beyond the hardware, the brain-computer interface companies also need serious machine-learning software skills and to perform thousands upon thousands of tests.
Neuralink’s implant sits invisibly beneath the scalp, flush with the skull. It’s also packed with enough computing horsepower to handle jobs well beyond think-and-click.
In the nearish future, the idea is to enable high-speed typing as well as seamless use of a cursor. Neuralink has also been working on a complementary spinal implant intended to restore movement and sensation in paralyzed people.
“The short-term goal of the company is to build a generalized brain interface and restore autonomy to those with debilitating neurological conditions and unmet medical needs,” says DJ Seo, a Neuralink co-founder and vice president for engineering.
“Then, really, the long-term goal is to have this available for billions of people and unlock human potential and go beyond our biological capabilities.”
Although some competitors have beaten Neuralink to human trials, the company’s raw technology is closest to being a general-purpose computer in the brain. The implant has more than 1,000 electrodes for gathering brain data, compared with 16 or so in rival devices.
The Neuralink hardware is a nesting doll of processing, communications and charging systems, including a battery and signal amplification.
Competitors, meanwhile, must still connect their implants via wires to bulky pacemaker-size battery and amplifier units that are often surgically implanted in a patient’s chest.
Neuralink’s battery lasts a few hours and can be recharged wirelessly in a couple of hours via a custom baseball cap.
Another favorite Musk move is bringing key manufacturing in-house, which adds financial risk, obviously, but saves time. Neuralink even makes its own semiconductors, an exceedingly rare step in the medical-device business. It tailors them specifically for its low-power, low-heat needs.
In Austin it’s turned a former ax-throwing bar into a 12,000-square-foot implant manufacturing line and testing center. Along with the usual mills, lathes and laser cutters, the shop includes more outlandish equipment, such as a refrigerator-size cabinet—filled with a type of synthetic brain fluid—that heats, cools and jostles implants to simulate years of wear and tear.
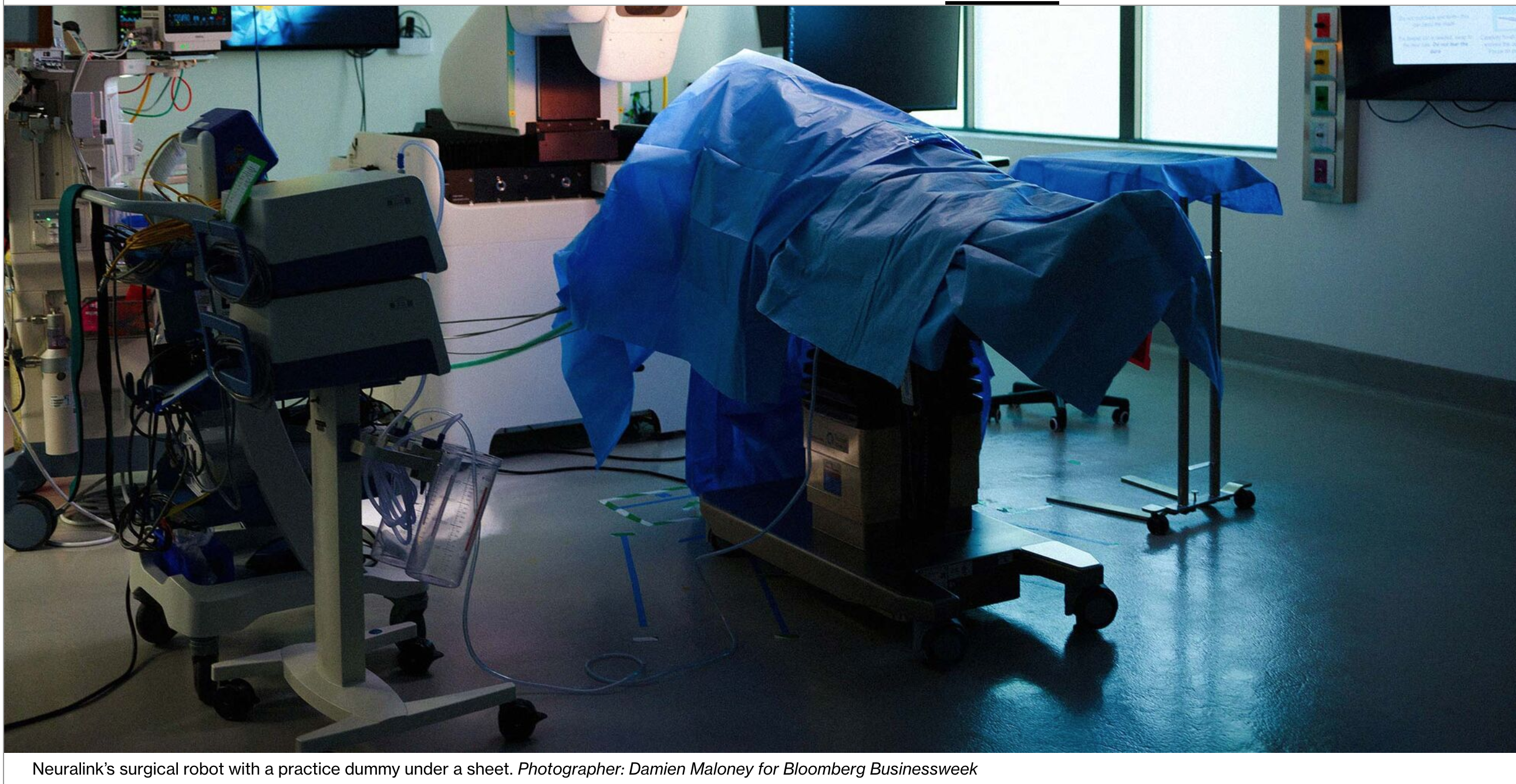
The priority during the surgery is to avoid creating any bleeding or scar tissue in a patient’s brain. To that end, Neuralink also built its own surgical robot. It’s a hulking, 7-foot-tall white machine with a steady, cubed base supporting a tower of electronics and equipment.
Once a human surgeon cuts a hole in a patient’s skull, the robot performs the ultra-delicate task of placing the electrode-laced wires, which Neuralink calls threads, into the brain.
The robot has cameras, sensors and a tiny laser-milled needle that it hooks into a loop at the end of each thread. One by one, the needle pushes the 64 threads, each lined with 16 electrodes, into the brain, all while carefully dodging blood vessels.
No human would be allowed to try this given that each thread is 5 microns thick, or about 1/14 the diameter of a strand of human hair.
To further avoid damaging tissue, the threads have been engineered to be a mix of thin, pliable and sturdy and have been coated with a special polymer to keep them from deteriorating over years.
Neuralink’s dozen or so robots performed 155 of these surgeries on sheep, pigs and monkeys in 2021 and 294 last year. With human subjects, the surgical prep and craniectomy are expected to take a couple of hours, followed by about 25 minutes for the actual implantation.
“The last two years have been all about focus on building a human-ready product,” Seo says. “It’s time to help an actual human being.”
During one of my visits, Musk pushed Seo and the rest of the engineers to do more, and faster. He wanted the robot to perform the surgery in less time and ideally without the help of a human surgeon.
He wanted semiconductor experts to forget what they learned in school and try out simpler manufacturing techniques. He wanted the implant to look sleeker and last longer and, well, maybe everyone needed to rethink just about everything.
I saw scientists wince as they considered the distance between their boss’s demands and the physical capabilities of their hardware. But I also saw Musk perform a type of pattern-matching for which he was especially suited, thinking ahead to how a range of design tweaks would affect mass production down the line.
His been-there, done-that attitude seemed to give the staff confidence that he was right—that he had some master plan he knew they could pull off.
Musk’s management style has its upside. It’s yielded the world’s most successful rocket and its most valuable car company. Of course, as anyone who’s Googled “Cybertruck window” can tell, his first drafts can be a mess. And, as he’s torn X, né Twitter, down around himself, he’s shown that the engineering shoe-banging doesn’t always lead to rational product choices.
“We need to get there before the AI takes over. We want to get there with a maniacal sense of urgency. Maniacal”
Seo and Musk are the only Neuralink founders still with the company. (The other six left of their own volition or because of disagreements about the company’s direction; a number of them have since started similar companies.) Seo runs development of both the implant and the surgical robot.
Jeremy Barenholtz, a computer scientist only two years out of Stanford University, has arisen as one of the lead executives and has managed the arduous US Food and Drug Administration approval process.
Musk tends to pop in and out, but he looms as the ultimate authority and chief executive officer. A July 2022 visit to the company’s headquarters in Fremont, California, showed how that works in practice.
Musk summoned top executives and engineers to catch him up on the latest progress toward human trials. The team gathered at a rectangular, Ikea-style table in the office’s main work area. It’s a large, open-plan space full of people at computers, robot prototypes and testing equipment.
Musk, in a black suit and holding a Red Bull, stood at the head of the table and started asking about competitors. As employees gave updates, he peppered them with technical questions. He was particularly focused on Synchron, which had already received regulatory approval to begin human trials.
Synchron’s pitch is technology that doesn’t require cutting into the skull. It makes a small, stentlike product called the stentrode that can be slid into a brain’s blood vessel through a patient’s vascular system.
The hardware can’t sit right against neurons for the best signals, but patients with paralysis were already using it to navigate web pages and send WhatsApp messages.
“We should be far exceeding the stentrode,” Musk said. “And they are currently kicking our ass. I want to be in dozens of people next year.”
His lieutenants grimaced in sync. Barenholtz had the unenviable task of explaining to Musk that the FDA wanted to wait at least a year after the company’s first surgery took place to try and implant more individuals.
“Unacceptable,” Musk said. “Generally, the way regulators work is that they’ll say one thing and that they really just kind of want to see what happens. If the patient is an advocate for the device and says it’s incredible and that we see no complications, they will approve the next one very quickly.”
It was like SpaceX getting federal approval for more rocket tests, he said: “If the thing is working well, and you get a letter-writing campaign to the FDA, I guarantee they’ll move.”
At another “update Elon” meeting a couple months later, Musk trained his sights more on Onward, which makes implants that nestle against the spine.
Its device sends electrical pulses that help reanimate muscles and have enabled people with paralysis to walk again. Their gait tends to be somewhat herky-jerky, but for people who’ve been paralyzed—and their loved ones—the act of standing and walking again can be similar to a religious experience.
Musk was also there to prepare for an important demo. Neuralink was planning to announce that it had started work on its own spinal implant, to be paired with its brain implant. This time, Musk seemed more agitated, and he pushed harder.
“In general, the company needs to make faster progress,” he said to Joseph O’Doherty, one of the assembled engineers. “The pace of progress is too slow.”
“OK,” O’Doherty said.
“Step it up.”
“OK.”
“Yeah.”
“OK.”
“I mean it.”
“Understood.”
“Step it up. Let’s go.”
None of this was playful. O’Doherty took it in stride and rolled right into a lengthy presentation on Neuralink’s progress with its early spinal technology, showing videos of an implant being used to stimulate the legs of pigs to make them walk on a treadmill.
The conversation covered different parts of the brain, parts of the spine, spike grids, joint angles, machine-learning models and more.
Musk, who has no formal medical training, kept up with it all. He made suggestions on how the implant could be tweaked to perhaps produce a less jerky motion in the animal’s gait.
His ideas were sometimes far afield of practicality, but then also often spot on. Following one visit, the semiconductor engineers retooled their process for bonding the threads to the company’s chips on Musk’s advice, and their manufacturing speeds went up 50% while defects went down, according to Zack Tedoff, the chip division’s head of brain interfaces.
The team working on the spinal implant went back to the drawing board to try to get their pigs to walk in a more true-to-life manner, and Barenholtz more or less started living at the office to address Musk’s every demand.
Musk turned out to be right about the FDA. Neuralink has received an outpouring of interest from thousands of prospective patients, and the agency recently gave it the green light to perform additional implant trials in 2024 without a yearlong evaluation period.
The company estimates that each implant surgery will run it about $10,500, including exams, parts and labor, and that it will charge insurers about $40,000.
It forecasts annual revenue as high as $100 million within five years. Neuralink says it plans to perform 11 surgeries in 2024, 27 in 2025 and 79 in 2026.
Then things really ramp up, going from 499 surgeries in 2027 to 22,204 by 2030, according to documents provided to investors.
Before that September 2022 meeting ended, Musk stressed speed on an entirely different level, “like the world is coming to an end.”
The reason the staff needed to work dramatically faster, he said, was to make sure hybrid human-implant brains stayed competitive with a theoretical artificial superintelligence that might otherwise wipe out humanity.
“We need to get there before the AI takes over,” he said. “We want to get there with a maniacal sense of urgency. Maniacal.”
There’s nothing pleasant about testing medical devices on animals. The practice is, on some level, a form of animal sacrifice on the altar of science, increasing their pain in the hope of decreasing it in humans.
Yet Neuralink has come in for special scrutiny regarding its treatment of animal test subjects, especially this year.
Reports in Wired, Reuters and other outlets have detailed surgical complications, behavioral side effects and prolonged suffering, particularly with Neuralink’s primate subjects.
Some implanted monkeys, the reports said, scratched and yanked at their heads until they drew blood, or acted despondent or agonized until they were euthanized.
Wired’s recent stories, which describe the monkeys’ deaths as “gruesome” and “grisly,” were based on public records it obtained and interviews with researchers.
Neuralink acknowledges that it’s made mistakes during exploratory surgeries, though it attributed them to human error rather than issues with its equipment. It stresses that the most troubling reports are drawn from its early years, before it built its own testing facility in Fremont, and that it has gone to great lengths to provide better living conditions there.
“I will always find a way to protect the animals in front of me,” says Autumn Sorrells, who manages Neuralink’s nonhuman test subjects and previously oversaw lab-animal welfare at the University of California at San Francisco.
“We get called ‘killers’ and ‘animal abusers’ and then have to come in to work and snuggle a sheep and make sure they have a great day. That’s f—ing hard.”
She says Neuralink’s animals have larger cages, more food and entertainment options, and much more socialization than she’s seen in other lab settings.
This squares with my reporting. I’ve seen the same group of rhesus macaque monkeys living at Fremont for three years now.
They’ve all had implants in their brain at various points. The devices can be removed, and a couple have been upgraded to newer models.
Seventeen of the monkeys are still active, healthy and feeding Neuralink brain data on-site; three retired to a sanctuary; and one was euthanized during a planned terminal procedure.
Before you can enter the animal facility, you have to don gloves, a gown, booties, a mask and protective eyewear. You also get a briefing on how to approach the animals: slowly, carefully, sans eye contact.
If you look a monkey in the eye, a signal of aggression, the monkey might freak out. Inside, spacious playpens are filled with toys, faux trees and playground equipment. Music is often playing throughout the facility, and TVs are on hand, showing mostly nature programs.
The monkeys’ primary role is to demonstrate that both the device and the surgical robot can work as intended. When they feel like it, they also contribute to the company’s thought translator by playing computer games.
Yes, Neuralink has a room full of monkeys that sit in front of computers and have their mind read, and it looks weirder than it sounds. For a couple of hours a day, they stare at laptop screens that are wheeled into position just outside the cages.
They can choose from games that involve joysticks and touchscreens (such as tracing letters and spelling words) or games that depend on brain-controlled clicking.
In one example, a 35-by-35 grid of small boxes appears on the screen, then one box suddenly lights up. The monkey’s goal is to move a cursor onto the lit box just by thinking it. The monkeys get faster at the tasks over time, and the expectation is that humans will, too.
It’s common for contract research centers to perform similar work by withholding food and water from the animals and pinning their skulls to metal rigs to hold their head in place.
Instead of experiencing such Clockwork Orange-style torments, Neuralink’s monkeys snack on fruit and smoothies while they do their jobs and stop when they want.
“Anytime they leave what we call the consent area, that means they’re done and that we need to back off,” Sorrells says. (The animals pick their food and soon their TV programs from an iPad.)
She’s unapologetic about Neuralink’s desire to move fast if that means a human gets a life-altering device sooner. “It’s unethical not to be hyperfocused on this,” she says.
The company is beginning to shift the balance of its animal testing and much of its operations from California to Texas, where there’s a newer, bigger campus on 37 acres of ranchland outside Austin.
This location has a surgery facility with multiple operating rooms, barns, a pathology building and a sci-fi-themed employee bar.
Neuralink says it plans to build an indoor-outdoor space for its primates. Today, it has dozens of sheep and pigs penned there. During a recent visit, the pigs were wearing little backpacks that held batteries and fed power to a patch atop their head to keep their implants charged.
The animals also have buttons in their pens they can press with their snouts to ask for food or a trip outside the barn.
It’s a long way from the snout button to 22,000 human implants. Just as with a rocket malfunction, a surgery gone wrong or an implant that leaks chemicals into someone’s brain would set the company back years.
And beyond basic safety, the device has to live up to its promise. Humans will be able to tell the world things about the implant that monkeys can’t, including where its limits are.
So far, downloading kung fu into your brain, let alone doing battle with an evil super-AI, remains sci-fi. Seo says that future implants will likely have 128 or more threads and that the next version of Neuralink’s custom chip should extend battery life to as long as 11 hours.
“The goal is to get to a full day,” he says, at which point patients will be able to recharge their implant overnight via a charging pad built into a pillow.
The Elon of it all makes a lot of things tougher. And yet, if his bluster pays off here, he’ll have played a central role in massively improving the life of a great many people.
He’ll deserve some of the credit even if it’s Synchron or Onward or somebody else that becomes the industry standard. If that thought makes your eyes roll, just try not to think about it too hard.
Related Article:
Our Brains Are Quantum Computers. Well, Maybe Not Yours (#GotBitcoin?)
Marrying Quantum Mechanics, the Human Brain, Consciousness and the Holographic Universe
The Intention Experiment The Largest Mind-Over Matter Experiment in History
Scientific Evidence for ESP A World Where There Are No More Secrets!
Quantum Computation In Brain Microtubules? The Penrose …
VW Nears Commercialization of ‘Quantum Routing’ Technique (#GotBitcoin?)
The Man Turning China Into A Quantum Superpower (#GotBitcoin?)










Leave a Reply
You must be logged in to post a comment.