Trump Get’s An “F” On Lowering Drug Prices (#GotBitcoin?)
Drugmakers have sharply boosted prices of some older, low-cost prescription medicines amid supply shortages and recalls—in some cases, by threefold and more. Trump Get’s An “F” On Lowering Drug Prices (#GotBitcoin?)
Patients and pharmacies grapple with sharply higher costs for certain generic drugs..
The increases are leading to higher costs for hospitals, pharmacies and patients on what are generally cheaper generic drugs, including some widely used medicines.

At least three sellers of a popular blood-pressure medication, valsartan, have lifted prices since a series of safety-related recalls of the drug by other manufacturers began last summer.
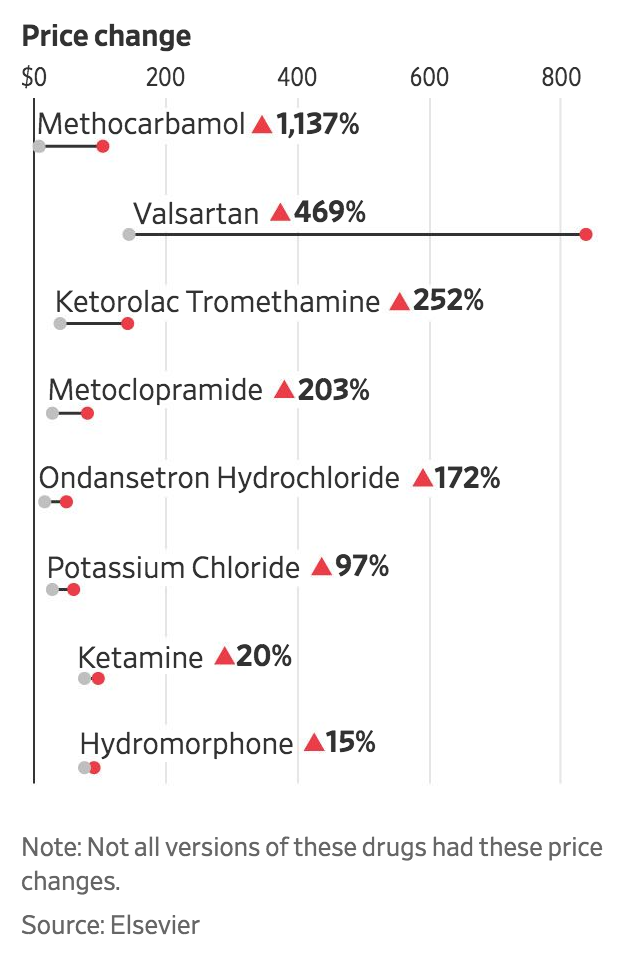
Virtus Pharmaceuticals in May raised the U.S. price of a bottle of a muscle relaxant that has been in short supply, methocarbamol, to $105 from $8.49. The 1,137% increase was the biggest for a prescription medicine in 2018, according to a new analysis of pricing data.
Of the nearly 120 drugs listed by the Food and Drug Administration as currently or recently in shortage, about one-third had price increases after the shortages started, according to a Wall Street Journal review of pricing data provided by RELX Group’s Elsevier health-information unit. Virtus didn’t respond to requests for comment.
Drugmakers say the price hikes reflect higher costs they have incurred to help fill supply voids that have become relatively common in recent years.
Supply And Demand
Drugmakers have raised prices for several older, low-cost medicines that face supply shortages or product recalls.
Several valsartan-based medications have been subject to recalls since last summer, after the FDA said an active ingredient from a supplier in China contained a potentially cancer-causing impurity. The agency restricted imports from the company, Zhejiang Huahai Pharmaceutical Co. Companies including Mylan NV and Teva Pharmaceutical Industries Ltd. have collectively recalled millions of bottles or cartons of valsartan.
The drug is still available from companies that haven’t been subject to recalls, and some of these have raised prices significantly.
Alembic Pharmaceuticals Ltd. of India raised the prices of several valsartan products by between 340% and 469% in July. Alembic’s price for a bottle of 90 valsartan 80-milligram tablets jumped to $155.11 from $29.98, according to Elsevier.
In August, India-based Macleods Pharmaceuticals Ltd. boosted valsartan prices by 216% to 306%. The same month, AmerisourceBergen Corp.’s American Health Packaging unit of Columbus, Ohio, raised prices for valsartan products by 57% to 63%, according to Elsevier.
A spokesman for AmerisourceBergen said its prices are affected by prices of bulk product that it purchases and repackages for health-system pharmacies. Macleods and Alembic didn’t respond to requests for comment.
In October, Alembic managing director Pranav Amin told analysts on a conference call that “one of our roles is to create a nimble supply chain that can react quickly to market opportunities,” according to a transcript. “So we could respond very fast to the valsartan opportunity, and we could ramp up our supplies and we could get on the market at a high price.”
The prices reflect so-called list prices, and drugmakers contend the ultimate price is often lower because of discounts and rebates negotiated by pharmacy-benefit managers and other purchasers. But the average price that U.S. community retail pharmacies pay for valsartan tablets has also risen—to 31 cents a tablet from 10 cents in July, according to a Wall Street Journal review of data from a Centers for Medicare and Medicaid Services survey of pharmacies.
And many patients with high deductibles often wind up paying full list price or a share of the cost derived from the list price.
David Behrman, a Houston retiree, said he used to pay about $13 out-of-pocket for a 90-day supply of valsartan to treat his high blood pressure, under a Medicare prescription plan. But he had to switch to a different company’s version because of recalls, and his cost has jumped to more than $108.
“It’s always been pretty reasonable until recently,” Mr. Behrman said. He said he emailed a complaint to the manufacturer of his new valsartan, Endo International PLC.
A spokeswoman for Endo said the Dublin-based company hasn’t increased the price of valsartan recently, and it is possible the previous supplier of Mr. Behrman’s valsartan was charging a lower price. Mr. Behrman couldn’t recall the supplier.
Overall, manufacturers of older generic drugs have come under heavy pricing pressure, as buyers including pharmacies and group-purchasing organizations consolidate and negotiate lower prices.
When shortages arise, health regulators such as the FDA sometimes ask other suppliers to boost production.
If a company incurs costs to boost production, it might choose to pass that along to customers, or “just have prices rise as a function of capitalism,” said Rena Conti, associate professor of markets, public policy and law at Boston University’s Questrom School of Business. “More limited supply meeting inelastic demand should give a producer pricing leverage should they choose to exert it.”
A study published last year in the journal Value in Health found that average prices rose about 14% for drugs in shortages lasting at least 18 months, and 6% during shortages of less than six months.
Another drug in shortage for more than a year has been hydromorphone hydrochloride, an injected opioid for pain relief, partly because one of the main suppliers, Pfizer Inc.’s Hospira unit, experienced production delays, according to the FDA.
The FDA asked another supplier, Fresenius Kabi AG, to increase production, Matt Kuhn, a spokesman for the German company, said. In late December, Fresenius boosted prices for various versions of hydromorphone, including a 15% increase for a box of 10 vials, to about $90.
Mr. Kuhn said the price increase reflected costs to boost production. “We have to make a decision to meet customer demand and shift our prioritization,” he said. Fresenius has reduced prices for other drugs, he added.
In October, Fresenius Chief Executive Stephan Sturm told analysts on a conference call, “Our various investments are meant to put us in pole position to fill any rising shortage situation.”
Mr. Kuhn said that while some price increases for generic drugs are large in percentage terms, they remain relatively inexpensive compared with brand-name drugs. This month, Pfizer, Johnson & Johnson , Allergan PLC and other drugmakers are raising list prices on many brand-name drugs that generally cost more than generics.
Updated: 12-18-2019
U.S. Advances Plan To Allow Imports Of Some Drugs In Bid To Cut Prices
Trump administration rule would let states develop plans to import certain prescription drugs from Canada.
The Trump administration is moving forward on a plan to allow the importation of cheaper drugs from Canada and other countries as part of its effort to reduce prescription costs.
The Food and Drug Administration will announce a rule letting states develop plans to import certain prescription drugs from Canada, federal officials said. About a dozen states, including Colorado and Florida, have pressed for permission to import drugs that meet U.S. quality standards.
In addition, draft guidance to be released Wednesday by the Department of Health and Human Services would enable certain medications made and sold in other countries to be imported to the U.S.
Opening up U.S. markets to imported drugs is a central plank of the administration’s effort to reduce drug prices, and federal officials say it would let Americans attain the same cost savings as consumers in other countries. It is also likely to be a major talking point for President Trump as the 2020 election kicks off.
Industry groups have argued the administration’s plan could threaten patient safety by letting in substandard or counterfeit drugs.
“Patients deserve access to the medicines they need with out-of-pocket costs they can afford,” said Jim Greenwood, chief executive of the Biotechnology Innovation Organization, which represents biotechnology companies. “Under no circumstance should we risk patient safety to achieve this goal, yet that’s exactly what this dangerous importation scheme will do.”
Mr. Azar, in a news conference Wednesday with Florida Gov. Ron DeSantis, said the government aims to make imported drugs as safe as any sold in Canada, and that there would be a controlled supply chain far different from that through which an American might buy what appear to be Canadian drugs online that in fact originate elsewhere.
Mr. Azar said there would be a 75-day public comment period for the rule and that the administration hopes to finalize it as quickly as possible.
Under the draft guidance, imported drugs would have to be tested for authenticity and safety, and be labeled for sale in the U.S., federal officials said. The drugs would also have to be FDA approved.
Importation of generic drugs isn’t part of the plan, but the Trump administration will seek comment from the public on possibly adding them, officials said.
Some industry groups in Canada have raised concerns about whether a change in U.S. policy might strain on supplies for the country’s own residents. Health Canada, the government department overseeing public health, has said it would take action if necessary to ensure Canadians’ access to drugs isn’t limited.
Drug prices are far lower in many countries compared with the U.S. Canada regulates the prices of many drugs, which keeps them relatively cheaper. In Europe, governments directly or indirectly control their costs through a negotiating process. In some cases, national health services buy drugs and set a price that manufacturers must meet.
U.S. prices are set on the open market, and through often less robust negotiation by insurers and some hospitals.
A number of state lawmakers have long sought to allow imported drugs from Canada. Mr. DeSantis, a Republican, has said the state’s plan to import drugs could mean cost savings of more than $150 million a year. State agencies spent almost $4.3 million in 2018 on a drug to treat human immunodeficiency virus that would have cost about $2 million in Canada, according to a Florida state report.
Colorado Gov. Jared Polis, a Democrat, signed legislation this year to eventually allow the importation of drugs from Canada, a plan that would require federal approval.
The proposals announced Wednesday build on a 15-year old federal law that allowed the commercial importation of drugs from Canada under certain circumstances. The law, which required HHS to ensure the imports would pose no consumer risk and save costs, was never implemented.
Seventy-nine percent of Americans said the cost of prescription drugs was unreasonable in a February poll by the Kaiser Family Foundation, and 78% said they supported importing drugs from Canada to lower costs.
The move by the Trump administration follows the House passage last week of a bill urged by Speaker Nancy Pelosi that would allow extensive negotiation of drug prices directly by the federal Medicare program, something that the administration doesn’t endorse.
Some Democratic presidential candidates, including former Vice President Joe Biden and Sen. Bernie Sanders of Vermont, have also called for the importation of drugs in their health proposals.
Consumer-oriented experts on drug prices say imports can mean lower prices for individual purchasers, but that drug companies and even foreign governments likely would limit the strategy for a broader population.
“People suffering from unconscionable drug prices are looking for relief, and for some people, with adequate safeguards, that may be found in the somewhat lower prices across the border,” said Peter Maybarduk of the consumer group Public Citizen.
But, he added, “Canada has about a 10th the population of the United States. Importing medicine is no national solution to our crisis of unaffordable medicine at home. Our government needs to challenge the power of drugmakers head-on, beginning by negotiating prices directly on behalf of Medicare, as candidate Donald Trump promised but President Trump has failed to do.”
Harvard Medical School professor Aaron S. Kesselheim, a researcher on drug issues, called the plan “another drug pricing-related proposal from the current administration that does not seem designed to actually try to reduce drug spending for patients or the government, but rather seems designed to create the appearance of activity to generate headlines.”
While federal officials said the proposals would help consumers with high prices, other initiatives by Mr. Trump to drive down drug prices haven’t taken off.
The White House in July dropped its plan to end drug rebates in Medicare over concerns it would be too costly, and that same month a federal judge blocked an administration rule that would have required drugmakers to put list prices in television ads.
Another proposal laid out in October 2018 to link some drug prices in Medicare to the lower prices paid overseas remains under review.
Updated: 1-2-2020
Drug Prices Climb by 5.8% on Average, Less Than Last Year
Pfizer led the way with increases of over 9% on more than 40 products, according to a new analysis
Pharmaceutical companies started 2020 by raising the prices of hundreds of drugs, according to a new analysis, though the average increase of 5.8% was lower than a year earlier amid growing scrutiny from patients, lawmakers and health plans.
Pfizer Inc. led the way, including increasing prices by over 9% on more than 40 products. The drug industry traditionally sets prices for its therapies at the start of the year and again in the middle of the year.
More than 60 drugmakers raised prices in the U.S. on Wednesday, according to an analysis from Rx Savings Solutions, which sells software to help employers and health plans choose the least-expensive medicines. The average increase was 5.8%, according to the analysis, including increases on different doses for the same drug.
The average is just below that of a year ago, when more than 50 companies raised the prices on hundreds of drugs by an average of more than 6%, according to the analysis.
Pfizer said that 27% of the drugs Pfizer sells in the U.S. will increase in price by an average of 5.6%. More than 90 of the New York-based company’s products rose in price, according to the Rx Savings Solutions analysis. Among them are Ibrance, which sold nearly $3.7 billion globally through the first nine months last year, and rheumatoid arthritis therapy Xeljanz.
A Pfizer spokeswoman said that nearly half of its drugs whose prices went up are sterile injectables, which are typically administered in hospitals, and the majority of those increases amount to less than $1 per product dose.
Pfizer’s largest percent increases, 15%, are on its heparin products, which are generic blood thinners typically administered in hospitals.
Pfizer said the heparin increases are to help offset a 50% increase in the cost of raw materials and expand capacity to meet market demand. The company said it is monitoring the global heparin supply, which has been challenged by the impact of African swine flu in China, as the drug is derived from pig products and disruption could lead to a shortage. Pfizer said that its U.S. heparin supply is not sourced from China.
Overall, the increases by drugmakers Wednesday affect “list prices,” which are set by manufacturers, although most patients don’t pay these prices, which don’t take into account rebates, discounts and insurance payments. Drugmakers have said prices are increased in conjunction with rebates they give to pharmacy-benefit managers, or PBMs, in order to be placed on the lists of covered drugs known as formularies.
In fact, drugmakers have said that their net prices have declined because of large rebates to PBMs, which negotiate prices in secret with their clients, such as employers and labor unions.
Pfizer said its price increases will be offset by higher rebates paid to insurers and middlemen. The company said the net effect on revenue growth in 2020 will be 0%, which is the same percentage expected for 2019. The company said the average net price of its drugs declined by 1% in 2018.
In 2018, Pfizer was assailed by President Trump after the company raised the prices on some 40 drugs. Pfizer temporarily rolled back the increases, but raised prices again later.
In Washington, Republicans and Democrats in the U.S. Congress have drawn up proposals for lowering drug costs, while the Trump administration recently introduced a plan for importing drugs from Canada.
“Prices go up but demand remains the same,” said Michael Rea, CEO of Rx Savings Solutions. Clients of the Overland Park, Kan., company include Target Corp. and Quest Diagnostics Inc. “Without the appropriate checks and balances in place, this is a runaway train. Consumers, employers and health plans ultimately pay the very steep price.”
While some increases in his firm’s analysis were steep, most product prices rose by less than 9%.
AbbVie Inc. raised the price of rheumatoid arthritis treatment Humira, the world’s top-selling drug, by 7.4%, according to the analysis. Through the first nine months of 2019, Humira sales totaled nearly $11 billion.
AbbVie didn’t respond to a request for comment.
GlaxoSmithKline PLC raised the prices on more than two dozen different therapies, although none by more than by 5%. That includes its shingles vaccine, Shingrix, which sold about $1.7 billion globally in the first nine months of 2019.
A Glaxo spokeswoman confirmed the increases and said net prices for its U.S. products fell about 3.4% on average annually the past five years.
Other major companies that raised prices included generic drugmaker Teva Pharmaceutical Industries Ltd. , which raised the price of more than two dozen products, but none by more than 6.4%, according to the analysis. Sanofi S.A. raised prices on some of their therapies, but none by more than 5%, while Biogen Inc. took increases that didn’t exceed 6%, including on multiple-sclerosis therapy Tecfidera.
A Teva spokeswoman said the company sets prices to help enable patient access, maintain a commitment to innovation and fulfill obligations to its stakeholders. A Sanofi spokeswoman confirmed the increases and said that the changes are consistent with its pledge to ensure price increases don’t exceed medical inflation. A Biogen spokesman confirmed the price changes and said adjustments are made to products for which it continues to invest in research, and otherwise increases follow inflation.
In addition to Pfizer’s increases on heparin, companies increased prices for several therapies by more than 10%, according to the analysis.
Cotempla XR-ODT, which is approved in the U.S. to treat attention-deficit hyperactivity disorder in children between 6 and 17 years old, increased by more than 13% to $420 for a month supply. The therapy is sold by Neos Therapeutics Inc., based in Grand Prairie, Texas.
Representatives for Neos didn’t respond to requests for comment.
Related Articles:
Trump Get’s An “F” On Lowering Drug Prices (#GotBitcoin?)
Drugmakers Sue To Block Federal Rule Requiring Drug Prices In TV Ads (#GotBitcoin?)
A Billionaire Pledges To Fight High Drug Prices, And The Industry Is Rattled (#GotBitcoin?)
What Do You Get When Legal Drug Dealers Peddle “Heroin-in-a-Pill” To It’s “Clientele”?
Freedom of Information Act Document Reveals Antidepressant Drugs Are Less Effective Than Placebos
Fentanyl’s New Foe: A Quick Test Strip That Can Prevent Overdoses (#GotBitcoin?)
Pharmaceuticals Found In Our Drinking Water!
Opiate-Addicted Babies Are The New Crack Babies of 2019
A Billionaire Pledges To Fight High Drug Prices, And The Industry Is Rattled (#GotBitcoin?)

Leave a Reply
You must be logged in to post a comment.