A Users Guide To Immortality
Aging In Humans Reversed With Hyperbaric Oxygen Therapy
Aging, which happens over time in humans, has been biologically reversed by hyperbaric oxygen therapy, according to new research published in the journal Aging. Aging In Humans Reversed With Hyperbaric Oxygen Therapy
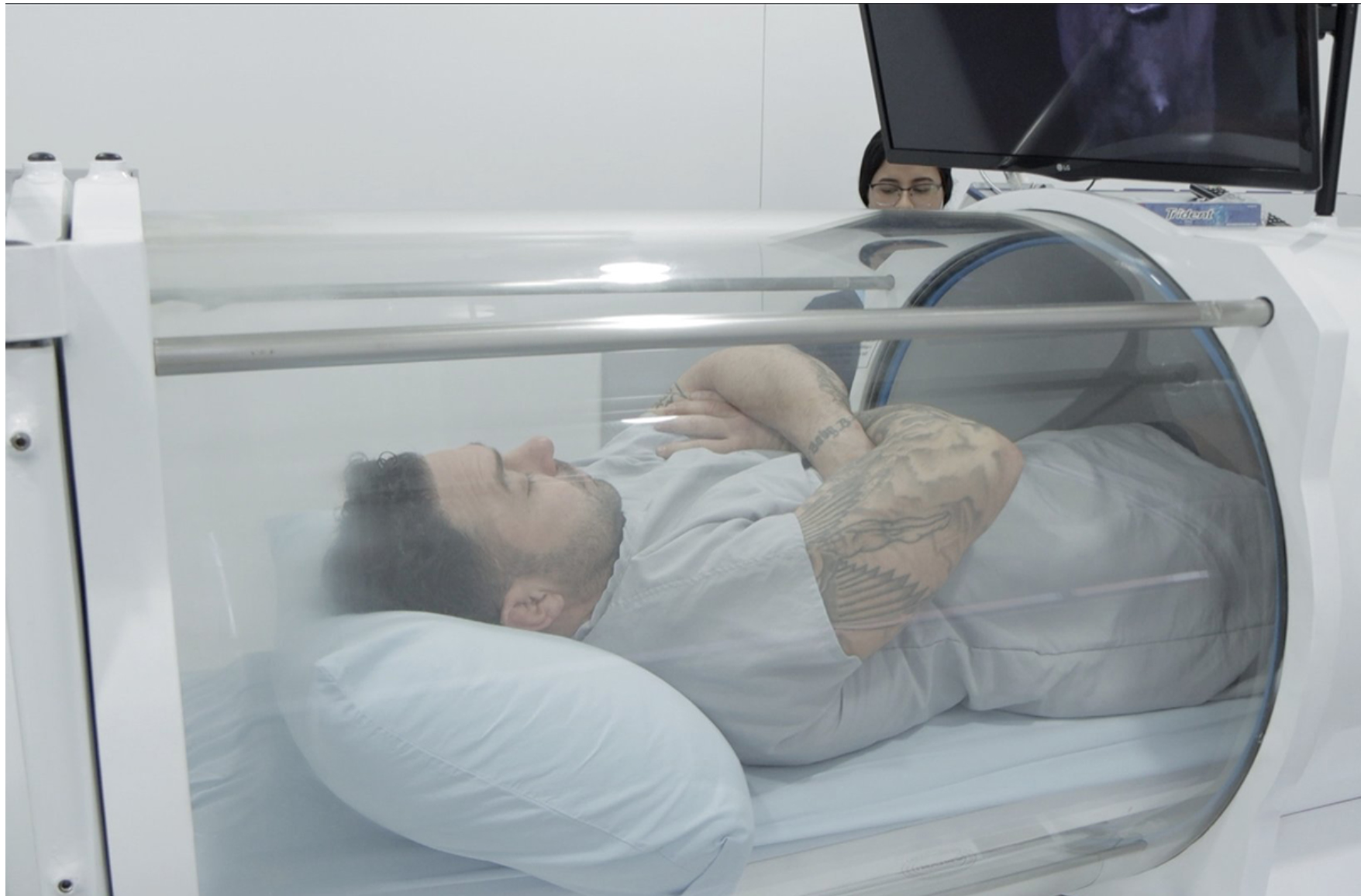
Scientists, led by Yafit Hachmo, from the Research and Development Unit, Shamir Medical Center, Tzrifin, Israel, turned back the human clock for 35 adults aged 64 and older by reversing two key areas of the body thought responsible for the frailty and ill-health that comes with growing older.
Protective caps at the ends of chromosomes, known as telomeres, shorten, causing DNA to become damaged and cells to stop replicating as people age. At the same time, senescent cells build up in the body, preventing regeneration.

Related:
Bitcoin Community Leaders Join Longevity Movement
Increasing telomere length and getting rid of senescent cells is the focus of anti-ageing research, with drugs being developed to target those areas, The Telegraph reported.
The 35 patients were repeatedly given pure oxygen in a hyperbaric chamber, which increased the length of their telomeres by 20%, a feat that has never been achieved before.
Scientists said the growth may mean that the telomeres of trial participants were now as long as they had been 25 years earlier.
The therapy also reduced senescent cells by up to 37%, enabling new healthy cells to regrow. Animal studies have shown that removing senescent cells extends remaining life by more than 33%.
“Since telomere shortening is considered the ‘Holy Grail’ of the biology of ageing, many pharmacological and environmental interventions are being extensively explored in the hopes of enabling telomere elongation,” said one of the scientists involved in the research, Professor Shai Efrati, Director of the Sagol Center at Shamir Medical Center.
“The significant improvement of telomere length shown during and after these unique protocols provides the scientific community with a new foundation of understanding that ageing can indeed be targeted and reversed at the basic cellular-biological level.”
Scientists believe ageing itself is responsible for diseases such as Alzheimer’s, Parkinson’s, arthritis, cancer, heart disease and diabetes.
It is known that obesity, smoking, lack of physical activity, vitamin deficiency and inflammation can speed up the shortening of telomeres, demonstrating that they have a major impact on the length of a life.
The 35 participants did not undergo any lifestyle, diet or medication adjustments. Each patient was placed in a hyperbaric chamber for 90 minutes for five days a week over three months while breathing 100% oxygen through a mask.
The pressurized chamber allows more oxygen to be dissolved into the tissues and mimics a state of hypoxia, or oxygen shortage, which is known to have regenerating effects.
Previous trials have shown that eating a healthy diet can preserve telomere length, while high-intensity training for six months has been proven to lengthen telomeres by up to 5%.
The Israeli team has also previously demonstrated that the pressurised oxygen therapy can improve cognitive decline.
“Until now, interventions such as lifestyle modifications and intense exercise were shown to have some inhibition effect on the expected telomere length shortening,” said Dr Amir Hadanny, Chief Medical Research Officer of the Sagol Center for Hyperbaric Medicine and Research.
“However, what is remarkable to note in our study is that, in just three months of therapy, we were able to achieve such significant telomere elongation – at rates far beyond any of the current available interventions or lifestyle modifications.”
About The Scientists Professor Shai Efrati (pictured above) is an Israeli physician. He is also an associate professor at the Sagol School of Neuroscience at Tel Aviv University.
Moreover, his title includes director of the The Sagol Center for Hyperbaric Medicine and Research at Shamir Medical Center at the Shamir Medical Center in Israel. Professor Efrati is also medical advisor and serves as Chair of Aviv Scientific’s Medical Advisory Board.
Amir Hadanny, MD is a senior physician, certified neurosurgeon and a hyperbaric physician and Chief Medical Research Officer at the Sagol Center for Hyperbaric Medicine and Research and Shamir Medical Center.
For the past decade, Dr. Hadanny has worked alongside Professor Shai Efrati on novel research focused on neuro-rehabilitation, neuro-plasticity and physiology. He has published more than 25 research papers focused on the effects of HBOT.
“With this pioneering study, we have opened a door for further research on the prolonged cellular impact of the therapy to reverse the ageing process. After dedicating our research to exploring its impact on the areas of brain functionality and age-related cognitive decline, we have now uncovered, for the first time in humans, biological effects at the cellular level in healthy ageing adults.”
The researchers pointed out that the study has several limitations and strengths to consider. First, the limited sample size has to be taken into account. Second, the lack of a control group.
“However, the study suggests impressive results on telomeres and senescent cell clearance, which weren’t observed in other interventions,” said the research paper.
Hope for your ageing brain and failing memory! @TelAvivUniversity & The Sagol Center for Hyperbaric Medicine & Research at Shamir Medical Center announce ground breaking anti ageing research
Full Interview: https://t.co/6A4Lb5wSdh @giselewaymes @neurology_live @JanineBester pic.twitter.com/LdD45drjSY— Bronwyn Nielsen (@bronwynnielsen) November 18, 2020
For The First-Time A Human Study Shows The Reversal In Biology Of Aging, With Hyperbaric Oxygen Therapy (HBOT)
TEL AVIV – November 18, 2020: In a scientifically verified approach, signalling an important breakthrough in the study of aging, Tel Aviv University and The Sagol Center for Hyperbaric Medicine and Research at Shamir Medical Center announced today that, for the first time in humans, two key biological hallmarks of aging, telomere length shortening, and accumulation of senescent cells, can be reversed.
The prospective clinical trial, published in peer-reviewed Journal Aging, utilizes Hyperbaric Oxygen Therapy protocols to demonstrate cellular level improvement in healthy aging adults.
Hyperbaric Oxygen Therapy Targets Aging As A Reversible Disease
The prospective clinical trial is part of a comprehensive aging research program taking place in Israel. It was conducted by Prof. Shai Efrati, MD, from the Faculty of Medicine and Sagol School of Neuroscience at Tel Aviv University, and Amir Hadanny, MD, Chief Medical Research Officer of The Sagol Center for Hyperbaric Medicine and Research and co-author of the study.
Using a specific protocol of hyperbaric oxygen therapy (HBOT), telomere length was significantly increased and senescent cells were reduced in a population of healthy aging subjects.
The study was published in the peer-reviewed journal Aging. Titled: Hyperbaric Oxygen Therapy Increases Telomere Length and Decreases Immunosenescence in Isolated Blood Cells: A Prospective Trial.
A Significant Breakthrough In The Study Of Aging
The biological deterioration of aging is cited as a major risk factor for cancer, cardiovascular diseases, diabetes, dementia, and Alzheimer’s disease. At the cellular level, two key hallmarks of the aging process are:
- The shortening of telomere length of approximately 20-40 bases per year, which is associated with a variety of serious life-threatening illnesses; and
- The accumulation of senescent cells, the so-called “old malfunctioning cells,” inhibit cell proliferation. The accumulation of senescence contributes to many age-associated conditions and illnesses, while the elimination of those cells can reverse them, as shown in previous animal studies.
The First Study To Evaluate Telomere Length And Senescence
This is the first study to evaluate whether hyperbaric oxygen therapy can affect telomere length and senescence using a specific HBOT protocol. The trial included 35 healthy independent adults aged 64 and older. They did not undergo any lifestyle, diet, or medication adjustments.
How Was The Study Conducted?
Each patient received 60 daily hyperbaric oxygen therapy sessions over the course of 90 days. Whole blood samples were collected prior to treatment, at the 30th and 60th session, and one to two weeks following the last HBOT session, to assess peripheral blood mononuclear cells (PMBCs) telomere length and senescence.
The Holy Grail Of The Biology Of Aging
“After dedicating our HBOT research to exploring its impact on the areas of brain functionality and age-related cognitive decline, we have now uncovered for the first time in humans hyperbaric oxygen therapy’s biological effects at the cellular level in healthy aging adults,” said Prof. Shai Efrati.
“Since telomere shortening is considered the ‘Holy Grail’ of the biology of aging, many pharmacological and environmental interventions are being extensively explored in the hopes of enabling telomere elongation.”
Significant Improvement Of Telomere Length
“The significant improvement of telomere length shown during and after these unique hyperbaric oxygen therapy protocols provides the scientific community with a new foundation of understanding that aging can, indeed, be targeted and reversed at the basic cellular-biological level.”
Improvement In Just Three Months
Results found that the telomere length of T helper, T cytotoxic, natural killer, and B cells increased significantly. They rose by over 20 percent, following HBOT. The most significant change was in B cells, which increased during the 30th session, 60th session, and post HBOT by:
- 25.68%±40.42 (p=0.007)
- 29.39%±23.39 (p=0.0001)
- 37.63%±52.73 (p=0.007)
In addition, there was a significant decrease in the number of senescent T helpers by -37.30%±33.04 post-HBOT (P<0.0001). T-cytotoxic senescent cell percentages decreased significantly by -10.96%±12.59 (p=0.0004) post-HBOT.
“Until now, interventions such as lifestyle modifications and intense exercise were shown to have some inhibition effect on the expected telomere length shortening,” explained Dr. Hadanny.
Reverse The Aging Process!
“However, what is remarkable to note in our study, is that in just three months of hyperbaric oxygen therapy we were able to achieve such significant telomere elongation – at rates far beyond any of the current available interventions or lifestyle modifications.
With this pioneering study, we have opened a door for further research on the prolonged cellular impact of HBOT to reverse the aging process.”
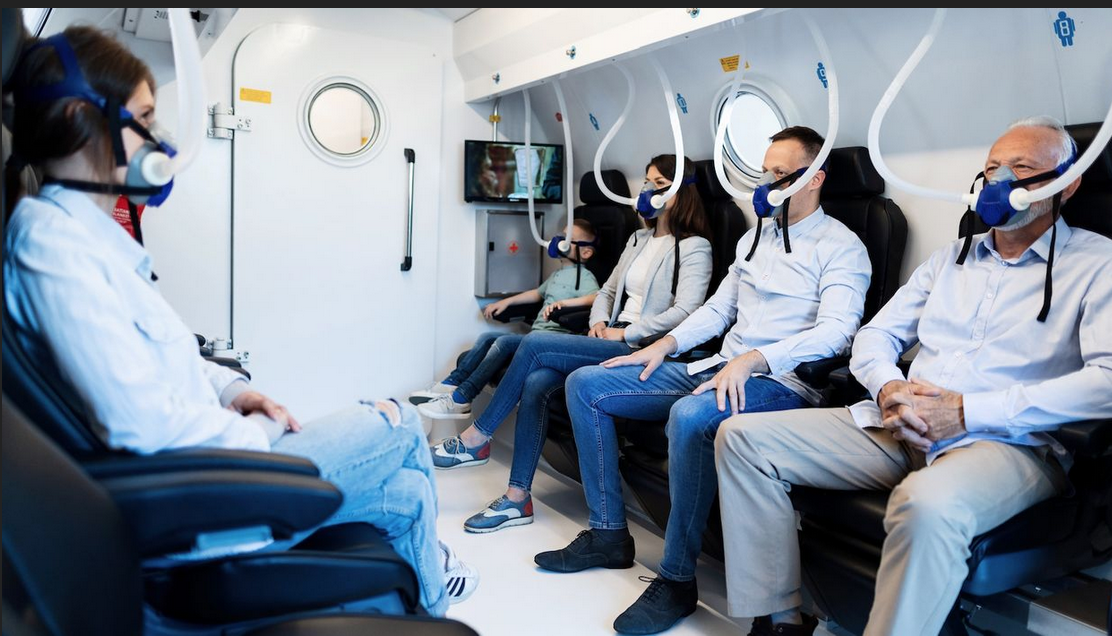
What Is A Hyperbaric Oxygen Chamber?
Hyperbaric oxygen has been used to treat several illnesses and injuries for which the FDA has approved. According to Johns Hopkins, hyperbaric oxygen therapy was first used in the U.S. in the early 20th century. “This was when Orville Cunningham used pure oxygen to successfully treat someone dying from the flu.
He developed a hyperbaric oxygen chamber but dismantled it after his use of therapy for other conditions failed. The therapy was tried again in the 1940s when the U.S. Navy used hyperbaric oxygen to treat deep-sea divers who had decompression sickness. By the 1960s, the therapy was also used to combat carbon monoxide poisoning.
Today, it’s still used to treat sick scuba divers and people suffering from carbon monoxide poisoning, including firefighters and miners. It has also been approved for more than a dozen conditions ranging from wound care, burns to bone disease:”
How Does It Work?
Firstly, there is a notable difference between portable hyperbaric oxygen therapy chambers or pods you can buy on the internet and even those you may use at walk-in health centres.
This is not to be confused with medically designed, approved hyperbaric oxygen therapy suites referred to in this scientific study.
According to the Mayo Clinic, hyperbaric oxygen therapy is generally a safe procedure. Complications are rare. But this treatment does carry some risk.
Nobel Prize For Medicine
Research into the revitalizing powers of oxygen has been in the spotlight recently. In October 2019, the Nobel Prize for Medicine was awarded to three researchers for their discovery of how living cells sense and react to changes in oxygen levels.
Maximize Rejuvenation
Our body senses fluctuations in oxygen and that rejuvenation processes can actually be triggered by these oxygen fluctuations.
This is one of the main principles behind the hyperbaric oxygen therapy medical protocol. The treatment involves the inhalation of 100% pure oxygen while sitting in a large HBOT suite.
The chamber is pressurized with air to above atmospheric levels. During HBOT, oxygen levels in the body’s tissues rise 10-15 times over that of normal conditions.
First, it increases the oxygen levels in the body’s tissues. It then fluctuates the level of oxygen in the body to enhance the body’s natural processes of rejuvenation. By controlling and combining these two processes, the medical team can maximize the rejuvenation potential of a person’s body and brain.
How Long Is The Treatment?
The treatment begins with three days of intensive assessments, identifying a person’s baseline cognitive and physical performance. Hyperbaric oxygen therapy sessions will take approximately two hours per day, five days a week, for 12 weeks.
Two to three times a week, at a designated clinic and increased to 2.5 hours to allow the person to participate in additional on-site training if needed.
How Long Do The Results Last?
The longevity of results varies from one individual to the next. They also depend on the person’s individual biology and lifestyle – mainly nutrition and physical activity.
In terms of the study, in just three months of HBOT, the scientists were able to achieve a significant telomere elongation. This is at a rate far beyond any of the currently available interventions or lifestyle modifications.
Can Pure Oxygen Reverse Effects Of Ageing? People Placed In Pressurised Chambers Gain Significant Benefits From Breathing Clean Air, Research Shows
* Breathing Pure Oxygen May Help To Reverse The Ageing Process, Scientists Claim
* Found Those Put In Pressurised Oxygen Chambers Gained Significant Benefits
* Sessions Lasted 90 Minutes Each And Took Place For Five Days A Week Over Three Months
Breathing pure oxygen may help to reverse the ageing process, the scientists behind a breakthrough study claim.
They found that subjects put in pressurised oxygen chambers gained significant benefits.
Firstly, it enabled telomeres – the protective caps on chromosomes – to regrow by more than 20 per cent.
Telomeres naturally become shorter with age, leading to illnesses including cancer, Alzheimer’s and Parkinson’s.
But in the experiment they grew back to the length of people 25 years younger.
The study also found that the number of senescent cells – which prevent regeneration as they build up in the body – was cut by up to 37 per cent. Removing the so-called zombie cells can extend life.
Professor Shai Efrati of the Faculty of Medicine at Tel Aviv University said: ‘Since telomere shortening is considered the ‘Holy Grail’ of the biology of ageing, many pharmacological and environmental interventions are being extensively explored in the hopes of enabling telomere elongation.
‘The significant improvement of telomere length shown during and after these unique protocols provides the scientific community with a new foundation of understanding that ageing can indeed be targeted and reversed at the basic cellular-biological level.’
In the study, published in the journal Ageing, 35 healthy adults aged 64 and older breathed in 100 per cent oxygen through a mask whilst sitting in pressurised chambers.
The sessions lasted 90 minutes each and took place for five days a week over three months.
The pressurised chamber mimics a state of ‘hypoxia’, or oxygen shortage, enabling tissues to dissolve more oxygen which has well-known regenerative effects.
Previous trials revealed that healthy eating and high-intensity exercise can also preserve telomere length – but this experiment far outdid natural interventions, the researches explained.
Study researcher Dr Amir Hadanny, of Tel Aviv’s Sagol Center for Hyperbaric Medicine and Research, said: ‘Until now, interventions such as lifestyle modifications and intense exercise were shown to have some inhibition effect on the expected telomere length shortening.
‘However, what is remarkable to note in our study is that, in just three months of therapy, we were able to achieve such significant telomere elongation – at rates far beyond any of the current available interventions or lifestyle modifications.’
Inquiry into the evolution of aging aims to explain why almost all living things weaken and die with age. A Users Guide To Immortality
There is not yet agreement in the scientific community on a single answer. The evolutionary origin of senescence remains a fundamental unsolved problem in biology.
Historically, aging was first likened to “wear and tear”: living bodies get weaker just as with use a knife’s edge becomes dulled or with exposure to air and moisture iron objects rust.
Prospects For Extending Healthy Life – A Lot
But this idea was discredited in the 19th century when the second law of thermodynamics was formalized.
Entropy (disorder) must increase inevitably within a closed system , but living beings are not closed systems . It is a defining feature of life that it takes in free energy from the environment and unloads its entropy as waste.
Living systems can even build themselves up from seed, and routinely repair themselves. There is no thermodynamic necessity for senescence. In addition, generic damage or “wear and tear” theories could not explain why biologically similar organisms (e.g. mammals ) exhibited such dramatically different life spans.
Furthermore, this initial theory failed to explain why most organisms maintain themselves so efficiently until adulthood and then, after reproductive maturity, begin to succumb to age-related damage.
THE IMMORTALISTS – A Short Film By Jason Silva
Aging has been slowed and healthy lifespan prolonged in many disparate animal models (C. elegans, Drosophila, Ames dwarf mice, etc.). Thus, assuming there are common fundamental mechanisms, it should also be possible to slow aging in humans.
Greater knowledge about aging should bring better management of the debilitating pathologies associated with aging, such as cancer, cardiovascular disease, type II diabetes, and Alzheimer’s.
Therapies targeted at the fundamental mechanisms of aging will be instrumental in counteracting these age-related pathologies.
Eliminating Cancer With Nanotechnology
Therefore, this blog is a call to action for greater funding and research into both the underlying mechanisms of aging and methods for its postponement. Such research may yield dividends far greater than equal efforts to combat the age-related diseases themselves.
Personalized Medicine Drugs Tailored To Your Genetic Makeup
Personalized Medicine: Using an individual’s own genetic information to guide better treatment and prevention of diseases–is one of the most talked-about areas in healthcare.
To understand how personalized medicine may play out in real life, consider a frequent traveler whose business takes him or her to Asia, South America or Africa.
In all of those places, mosquitoes spread dengue fever–a rapidly-growing, infectious tropical disease for which there is no vaccine.
People who contract dengue fever can have a wide range of reactions. A fortunate few develop no symptoms at all. Others experience a week of flu-like symptoms–high fever, vomiting, headaches, muscle pain or a measles-like rash.
However, a small number of people develop a life-threatening variety known as dengue hemorrhagic fever. Diagnosing and treating the disease quickly, especially the more severe variety, has always been challenging for doctors.
How does this relate to personalized medicine? Allan Brasier, director of the Institute for Translational Sciences at the University of Texas Medical Branch (UTMB) , led a team that identified protein markers that may be able to predict a predisposition toward developing dengue fever and dengue hemorrhagic fever.
In the future, these markers could guide physicians to take earlier steps with those who show symptoms and are at high risk for the more serious strain of the disease. They could receive transfusions and other early intervention strategies that could save more lives.
This has been the goal of personalized medicine since the human genome was first sequenced in 1993. “Personalized medicine could eliminate the trial-and-error approach of giving every patient with the same disease the identical drugs or treatment ,” Brasier says.
“We can identify subgroups that have the same disease and can be targeted for different treatments based on their genetic information.” The goal is to avoid wasting time and money on potentially ineffective treatments, which expose many patients to harmful side effects.
Genomics and Disease
Clay Marsh, M.D., executive director of the Center for Personalized Health Care at The Ohio State University Medical Center , explains the leading uses for personalized medicine so far have been treatments for cancer and infectious diseases, along with better targeting of pharmaceuticals.
“Cancer is the most clinically applicable domain of genomics in medicine today. A cancer cell is clearly identifiable as the problematic cause of the disease and genetic profiling has identified key cellular pathways to target with specific drugs.
Similarly, infectious disease cells can also be genetically fingerprinted for a specific disease, and this is the next exciting application of genomics. Other diseases are proving to be more complex to fingerprint.”
Currently, there are an estimated 300 Phase II or higher oncology drugs that are being developed which have the potential for testing against a genetic biomarker . Right now, molecular testing is helping identify which patients with breast cancer and colon cancer are likely to benefit from different treatments.
For example, a gene expression test has been developed that can help determine which patients with breast cancer might benefit from chemotherapy. Joseph Sparano, M.D., associate chairman of the department of oncology at Montefiore Einstein Center for Cancer Care in New York , says the test measures the activity level of a panel of genes within the tumor sample, and the result correlates with the likelihood of having breast cancer recurrence.
Because of this information, doctors can identify a subset of patients who are likely to be cured with surgery and hormonal therapy alone, sparing them the need to undergo chemotherapy after surgery.
Clinical trials are underway to help guide treatment of the 25 to 50 percent of patients who fall into the “gray” area–the intermediate risk category–for which the best course of action is unclear.

Personalized Prescriptions
, the science of how a person’s genetics affects how they respond to certain medications, is another key area of personalized medicine. Variations in DNA affect how an individual’s body absorbs, metabolizes and uses drugs.
Michael Christman, Ph.D., president and chief executive officer of the Coriell Institute for Medical Research , a nonprofit biomedical research institution in Camden, N.J., points to clopidogrel , a medication that is prescribed after someone has a heart attack or stent placement.
“Up to 30 percent of people prescribed the medication do not activate the drug, and may as well take sugar pills,” he says. “However, there is an alternative FDA-approved drug, and if genetic testing were performed for the patient prior to dosing, the best drug could be selected first .”
This is hardly an isolated example. Personalized medicine has the potential to assist the large number of people who are prescribed medication that provides them no benefit because of their individual genetic response.
“Right now, we know that one-third of the people who receive a drug get a positive response, one-third get no response, and one-third get a toxic response,” says Jonathan S. Dordick, Ph.D., director of the Center for Biotechnology & Interdisciplinary Studies at the Rensselaer Polytechnic Institute . “By tailoring drugs to the physiologic of the person, we can change the number of people who get a beneficial response from one-third to two-thirds. And we can reduce the negative reaction from one-third to one-sixth.”
That means fewer trial-and-error prescriptions and a steep drop in the number of adverse drug reactions, which cause more than 770,000 injuries and deaths a year in the U.S .
Faster Time To Market
Genetic information could also provide benefits in how new drugs are developed. Dordick points out that pharmaceutical companies now focus on developing drugs that have a large enough potential to generate $1.5 billion to $2 billion in revenues. That’s primarily because the complex drug approval and clinical trial process is so costly.
The promise of personalized medicine is that instead of developing, say, one drug for asthma, a pharmaceutical company could develop five different versions of the drug designed for different populations based on their genetics.
However, one issue is that each of these five versions, under the current drug development framework, would need to undergo the expensive drug approval and testing process. That may not be cost effective since the testing costs would increase fivefold, but the amount of revenue the five drugs generate in total would not be five times that of a single version of the drug.
Dordick envisions new testing techniques that would make it more viable to develop a series of drugs for genetic subgroups of patients.
For one thing, he suggests eliminating the initial step of testing a drug’s efficacy on rats or dogs–the idea is that if human genetics are so dissimilar that not every drug will work with every human, there is no reason to spend time and money testing the drugs on biologically dissimilar creatures.
Instead, researchers could leverage new technologies that test the toxicity and effectiveness of new drug molecules using individual human cell cultures. In essence, at some point, a doctor could test how effectively a drug would work for you by testing your own cells.
If such tools were integrated into the drug approval process, Dordick says they could speed the process, reduce the costs, and weed out unsafe or ineffective drugs early in the process.
“If you are giving a drug to a specific set of the patient population, you can get very high quality candidates for testing and shave a tremendous number of years off the development,” he says. ” Because the limited number of patients can be more easily classified, you will be able to use hundreds of patients in the clinical trials rather than thousands of patients .”
Inventing The Rules
At the same time, medical practitioners say that safeguards need to be put in place for how to use the evolving information about genetics in treatment. The Coriell Institute uses a scientific advisory panel composed of physicians, scientists and ethicists called the Informed Cohort Oversight Board (ICOB) to help determine what genetic variants will be used in guiding treatment for different diseases.
“We generate and present risk information to the ICOB panel that independently judges its validity. We abide by the panel’s expert decisions, even when they disagree with our recommendations,” Christman says. For example, research done by the Institute identified a genetic risk variant for breast cancer.
The researchers thought the variant could be useful in determining which women should receive a mammogram at an early age. However, an oncologist on the advisory panel noted the increased radiation risk from the earlier mammograms outweighed the predictive value of the genetic information.
As all this suggests, there are many questions about how to best integrate genetic information into the treatment of patients. Personalized medicine has huge promise, but it also brings up issues such as healthcare, payer and physician incentives, medical record privacy, and the ethics of clinical trials that will need to be worked through.
“This has happened so quickly,” says Brenda Finucane, a certified genetic counselor and president of the National Society of Genetic Counselors (NSGC) . “We have the genetic technology before we have evidence-based models on how to use the technology.
This is very different than other developments in medicine, where you had time to think about it for a while and developed evidence-based medicine. We don’t have time, so we are developing practice guidelines on the fly. Patients and healthcare providers will all be learning together as this gets rolled out.”
As the mechanisms of aging are increasingly understood, increasingly effective interventions can be developed that will help prolong the healthy and productive lifespans of a great many people .
Transending Human Capabilities
Transhumanism is an international intellectual and cultural movement that affirms the possibility and desirability of fundamentally transforming the human condition by developing and making widely available technologies to eliminate aging and to greatly enhance human intellectual, physical, and psychological capacities. Transhumanist thinkers study the potential benefits and dangers of emerging technologies that could overcome fundamental human limitations, as well as study the ethical matters involved in developing and using such technologies. They predict that human beings may eventually be able to transform themselves into beings with such greatly expanded abilities as to merit the label “posthuman”.
Bone Marrow Stem Cells
You have within you a powerful set of tools to repair your body and keep you healthy. The future of medicine is NOT better drugs but better use and application of your body’s own stem cells . As of now stem cell -rich tissue can be extracted from your hip with virtually no discomfort and used to help restore your body.
This opens up an exciting new horizon in terms of preventing and treating disease and tackling the symptoms of aging – if not aging itself. Already, patients are returning to Dr. Steenblock for additional bone marrow treatments because they are seeing that their gray or white hair is turning back to its original color. Their skin not infrequently looks younger too and they report having more energy and less arthritic aches and pains!
In regard to its anti-aging effects, the bone marrow contains primitive progenitor cells that are associated with the early development of the fetus. These primitive cells reside dormant deep inside your bones and sport a genetic profile from your early development.
When these primitive cells are released into your system, there can be a revitalization of your body that physiologically “sets the clock back” in-a-way. Several patients have reported that the bone marrow transplants have also improved their sexual performance. This side effect is thought to be the result of stem and progenitor cells repairing sex organs as well as other tissues.
What does this mean for you? Your bone marrow stem cells have the potential to repair damaged tissues and organs. Whether you want an ” anti-aging “ treatment or you need the procedure to repair damage in your joints, liver, kidneys, heart or brain, a bone marrow transplant is an efficient and sure way to flood your body with stem cells.
Simple Test s Determine How Long You’ll Live
The Power of Knowing
Telomere length is one of the best biomarkers of overall health status. It is a major “integrator” of current and lifelong factors that impact health, including genetics, diet, fitness, toxins, and chronic stress. Knowing your telomere length (and monitoring changes over time) can provide valuable information on your disease risk – or even the rate at which you are aging. With this information, you have the knowledge to change the quality of your life and health status at a cellular level.
The Power of Change
Telomeres are the only “changeable” part of the genome, and positive lifestyle choices can increase telomere length and promote individual wellness. Your Cells are Your Guide to Personalized Solutions for Optimal Health Monitoring your telomere length over time can provide insights about potential disease risk and your rate of physiological aging. This knowledge can help to inform your lifestyle and, eventually, as research reveals more specific applications, it may also help inform therapeutic or prophylactic drug choices and decisions.
“Knowing whether our telomeres are a normal length or not for a given chronological age will give us an indication of our health status and of our physiological ‘age’ even before diseases appear,” says Maria A. Blasco , who heads the Telomeres and Telomerase Group at the Spanish National Cancer Research Center and who co-founded the company Life Length .
Telomere research pioneer Calvin B. Harley, who co-founded Telome Health last spring with Nobel laureate Elizabeth H. Blackburn , considers telomere length ” probably the best single measure of our integrated genetics, previous lifestyle and environmental exposures .”
Soon the companies will offer telomere-measurement tests to research centers and companies studying the role of telomeres in aging and disease; the general public may have access soon after through doctors and laboratories, perhaps even directly.
When Human Brain Cells Meet Silicon Chips
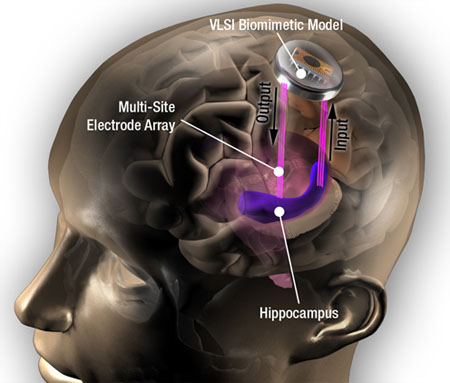
Direct interfaces between small networks of nerve cells and synthetic devices promise to advance our understanding of neuronal function and may yield a new generation of hybrid devices that exploit the computational capacities of biological neural networks. There are several research teams in the U.S. and Europe that are currently working on so-called neural-silicon hybrid chips.
One of the most celebrated researchers in the field is Ted Berger at the Center for Neural Engineering at University of Southern California in Los Angeles . Berger is also a key player in the newly established National Science Foundation Engineering Research Center devoted to biomimetic microelectronics.
Berger has set his sights on building artificial neural cells, initially to act as a cortical prosthesis for individuals who have lost brain cells to neurological diseases such as Alzheimer’s. But eventually, his lab’s efforts may usher in a new era in biologically inspired computing and information processing.
Berger’s strategy in building artificial neurons has been an empirical one. Rather than attempt to determine every aspect of how neurons communicate, he’s chosen to emulate their behavior, bombarding live neurons from rat hippocampus tissue with every conceivable type of electrical input, and observe what output emerges from the cell.
His team at USC then built a silicon microcircuit that behaves accordingly, at least in terms of spatio-temporal patterns of electrical inputs and outputs. The USC team has built circuits that model 100 neurons; their goal is to construct a 10,000-neuron chip model for implantation in primate hippocampus.
The Max Planck Institute in Germany is another center of research on neural-silicon hybrids. Recently, RA Kaul and P. Fromhertz from the Institute and NI Syed from the University of Calgary reported in Physical Review Letters on direct interfacing between a silicon chip and a biological excitatory synapse.
The team constructed a silicon-neuron hybrid circuit by culturing a presynaptic nerve cell atop a capacitor and transistor gate and a postsynaptic nerve cell atop a second transistor gate.
They applied a voltage to the capacitor, which excited the presynaptic neuron, and this activity was recorded with the first transistor. When the presynaptic neuron fired, it generated excitation of the postsynaptic neuron, presumably via an excitatory synapses, and the activity in the postsynaptic neuron was recorded with the second transistor. Further, short trains of activity in the presynaptic neuron appeared to increase the strength of the excitatory synapse between the cells, creating a memory trace within the circuit.
These results demonstrate the ability to use integrated capacitors and transistors to stimulate and record from cultured neurons. The neuron-silicon hybrid provides a tool to study formation and plasticity within small neural circuits and may lead to novel computational devices.
Fasting 101: Rebooting The Body’s Hard Drive: Rejuvenating, Life-Extending And Removes Deadly Toxins.
When Diet Is Wrong Medicine Is of No Use.
When Diet Is Correct Medicine Is of No Need.
~Ayurvedic Proverb
Updated: 2-15-2022
Centenarian-Sourced Probiotic Mixed With Dietary Fiber Has Anti-Aging Effects
A probiotic strain isolated from centenarians’ feces combined with dietary fiber showed beneficial effects on brain and gut function and has potential as an aging treatment for the elderly.
Highlights
* A probiotic isolated from centenarians called LTL1361 mixed with dietary fiber alleviates age-related cognitive impairment and protects the brain of healthy aging mice.
* This probiotic and dietary fiber combination also improves gut function and reduces inflammation in the blood and colon.
* These findings suggest the LTL1361 and dietary fiber mixture as a novel and promising anti-aging agent in humans.
Accumulating evidence indicates that gut microbiota regulates host health. This microorganism population can be manipulated in multiple ways: with probiotics to change the variety and with dietary nutrients or fiber to supply specific compounds.
While studies have focused on the anti-aging effects within a particular disease, the evidence is limited for the anti-aging effects of centenarian-sourced probiotics combined with a dietary fiber complex.
Researchers from Guangxi University in China reveal the anti-aging effects of an isolated probiotic strain from healthy centenarians’ feces mixed with dietary fiber.
With three months of treatment, this combination improved learning and memory ability, antioxidant capacity, and inflammation markers in aged mice.
Published in the journal Nutrients, the researchers suggest that probiotics and dietary fiber combination might be used as a novel and promising anti-aging synbiotic agent in humans.
Fighting Aging With Feces-Sourced Probiotics And Fiber
As aging is inevitable and its relative negative symptoms are complicated, we tend to pursue anti-aging treatments, ranging from diets to drugs. Accumulating evidence has indicated that prebiotics and probiotics could affect our health by regulating our gut microbiota.
Prebiotics — a form of dietary fiber that feeds the “friendly” bacteria in your gut — are resistant to digestion and absorption during passage through the stomach and small intestine but can be fermented in the large intestine by gut bacteria.
This dietary fiber can influence our gut bacteria to produce beneficial metabolites, such as short-chain fatty acids (SCFAs). But data indicate that daily dietary fiber intake for the elderly is roughly 40% below the recommended adequate intake.
For example, concentrations of SCFAs, which may promote weight loss and provide various health benefits, are less optimal in the elderly.
Live microorganisms called probiotics — like the ones advertised in cultured dairy products (e.g., yogurt and kefir) — administered in adequate amounts exert health benefits to the host.
Probiotic strains derived from the elderly exhibit excellent antioxidant, cholesterol-lowering, and immune-regulating activities.
Lactobacillus is the largest probiotic group with a high possibility of developing functional food. However, individual bacterial strains always exhibit unique bioactivities that require experimental confirmation.
Several Lactobacillus strains have been reported to have anti-aging effects due to their radical-scavenging activity and oxidation stress-attenuating ability.
Centenarian-Sourced Probiotic And Dietary Fiber Improve Aging In Mice
Previously, the Guangxi University research team isolated a strain of Lactobacillus — which they named LTL1361 — from the feces of healthy centenarians living in Bama, China, and demonstrated its potential probiotic properties in preliminary research in cultured cells.
In this study, Ren and colleagues examined the anti-aging effects of the LTL1361 strain and dietary fiber on learning and memory ability, antioxidant capacity, inflammation markers, and SCFAs in natural aging mice.
Using the Morris water maze test of spatial memory, the Guangxi University researchers evaluated the effects of dietary fiber and LTL1361 on cognitive abilities in aged mice. Researchers used the Morris water maze to study spatial memory and learning.
In this test, animals are repeatedly placed in an opaque pool of water surrounded by environmental cues, where they are tracked as they learn to find a hidden escape platform.
The researchers observed a reduction in the time it took aged mice treated with dietary fiber or LTL136 to find the platform. The results revealed that the combination of dietary fiber and centenarian-sourced LTL1361 significantly improved learning and memory ability.
In addition to improving learning and memory, Ren and colleagues showed that the combination of dietary fiber and Lactobacillus LTL1361 protected intestinal wall function and areas of the brain associated with cognition (pyramidal neurons) and learning/memory (hippocampus).
Furthermore, LTL1361 and dietary fiber decreased oxidative stress and inflammation and increased the availability of SCFAs in the small intestine.
Can Probiotics And Fiber Improve Human Aging?
The combination of dietary fiber and Lactobacillus LTL1361, which is a synbiotic (mixture of probiotics and prebiotics) seems like a promising treatment for cognitive decline in the elderly.
Nevertheless, further studies are required to clarify the biological connection between our gut, the microorganisms living within our gut, and how they affect our brain during aging.
Most of the latest clinical research has focused on applying commercial probiotics (one strain or a cocktail) to healthy older people.
The results indicate that probiotic consumption may positively impact by increasing the levels of specific beneficial gut microbe populations or modifying subpopulations of Lactobacillus.
These studies show that probiotics can enhance the immune response and improve bowel movements, among other beneficial effects.
Other studies have shown that the health benefits of probiotics are related to their ability to revert age-related increases of opportunistic pathogens, such as Clostridium difficile, which are involved in antibiotic-associated diarrhea that impact nutrition and inflammatory status.
In the elderly, C. difficile-associated diarrhea was linked to a reduction in the number of Bifidobacteria. For this reason, therapies based on the use of probiotics to correct the microbiota imbalance seem promising.
However, current guidelines do not recommend their administration. Notwithstanding the promising results, other studies reported controversial ones, most of them having no significant effects.
Source:
Ren M, Li H, Fu Z, Li Q. Centenarian-Sourced Lactobacillus casei Combined with Dietary Fiber Complex Ameliorates Brain and Gut Function in Aged Mice. Nutrients. 2022 Jan 13;14(2):324. doi: 10.3390/nu14020324. PMID: 35057509; PMCID: PMC8781173.
Updated: 4-10-2022
This Is What The World’s Oldest Person Ate Every Day
A recent study by UK-based CBD company Eden’s Gate sought to uncover the secrets to living a long life by studying some of the world’s oldest people (via Eat This, Not That!).
To get at the core of what gives individuals health and longevity, researchers examined the lifestyles of six people who lived to be older than 100.
Their results indicated that four main factors determine longevity — movement, socioeconomic status, stress levels, and diet. Of those categories, diet may be the one that people have the most control over.
There are plenty of foods that science says will help you live longer. For example, nuts, berries, and fish are known to provide essential nutrients that contribute to a long life by boosting immunity and improving heart health.
The Eden’s Gate study took a direct approach to gathering information by examining the diet of the oldest person from their sample of research subjects. At 122 years old, Jeanne Louise Calment is the only person to ever live past 120.

The Three Key Ingredients Of A Long Life
As Eat This, Not That! reports, CBD company Eden’s Gate studied the diet of the only person to ever live to 122 years old. In doing so, they identified three foods that may help you live longer.
Jeanne Louise Calment, who died in 1977, reportedly said olive oil was one of her secrets to a long healthy life.
According to Healthline, olive oil is rich in healthy fats, antioxidants, and anti-inflammatory properties. It also may protect against heart disease and can decrease one’s chances of having a stroke.
Another key element in Calment’s diet was red wine, which the Mayo Clinic reports is heart healthy and flush with antioxidants. Experts recommend only consuming red wine in moderation, however.
Finally, chocolate-lovers can rejoice in the fact that their favorite sweet treat can add years to your life, according to both Calment and Hopkins Medicine. Like red wine, chocolate has heart-healthy qualities and can boost immunity.
It can also help your athletic performance by boosting brain function and supporting circulation in the blood. So, pour a glass of wine, drizzle some olive oil on your food, and follow it up with some chocolate for dessert. It’s all in the name of longevity.
Longevity Guidebook
Chapter Five: Peter’s Meds, Supplements, & Therapeutics “It’s likely that we’re just another ten to twelve years away from the point that the general public will hit longevity escape velocity.” – Ray Kurzweil
Peter’s Legal Disclosure: I am an educator, entrepreneur, and scientist. I’m not a clinician and cannot make clinical recommendations for prevention or treatment of any disease. In providing the details below, I am sharing the list of supplements and medications I’m taking based on my physician’s recommendation.
No one should start taking any supplement without first checking with his or her personal physician. Some supplements can be dangerous for people with certain pre-existing medical conditions and supplements can interfere with some prescription drugs.
Supplements can also affect different people differently. The FDA has reviewed these supplements to determine whether their listed ingredients are safe to consume but no US regulatory authority has reviewed their ability to address cognition, dementia, Alzheimer’s disease, or human brain health generally.
The evidence of benefit for most of these supplements comes from laboratory experiments, and/or from epidemiology data, not from human clinical trials. Supplements should only be purchased from trusted retailers and brands; testing has shown that many supplements are tainted with unlisted ingredients and/or do not contain the amount of the supplement listed on their label.
My Prescription Medications
Note: The prescription medications listed below are specific to my medical objectives, based upon my blood biomarker testing, and the recommendations of my Fountain Life physician.
You will need to determine what is appropriate for you in consultation with your physician. My goal here is only to provide you with a full disclosure of my current regimen for informational purposes.
My Cholesterol Medications
One of my highest risks from family history and genetics is heart disease secondary to hypercholesterolemia. As a result, following are the cocktail of medications I take to give an ideal cholesterol blood panel. These work for me and keep my ApoB levels at an optimal range.
Nexlizet (180 / 10 mg): This is a combination medication used to treat high levels of cholesterol and other harmful types of cholesterol in the body. It contains bempedoic acid and ezetimibe, which work by inhibiting cholesterol synthesis and reducing the amount of cholesterol your body absorbs from your diet. This medication is taken orally, daily.
Crestor (5 mg): I take Crestor (rosuvastatin), a low-dose statin, for a number of reasons beyond its impact on lowering LDL (bad cholesterol) and triglycerides. It also has anti-inflammatory effects (established in various acute and chronic inflammatory models) as well as antiviral and antioxidant properties. It also helps to stabilize any vulnerable plaque.
Repatha (PCSK9 biologic): This is a biologic medication (a monoclonal antibody) that is used to lower LDL cholesterol levels. It works by blocking a protein called PCSK9, which inhibits the liver’s ability to remove LDL cholesterol from the blood. This is injected (with an autoinjector) every two weeks.
My “Longevity-related” Medications
Rapamycin (6 mg) – taken 1x per week, on Sunday evening: Rapamycin is a compound discovered on Easter Island, it was initially used as an immune-suppressant. It acts by inhibiting mTOR, a protein involved in cell growth and proliferation.
For longevity, its benefits may come from the modulation of cellular processes such as autophagy (cellular “clean-up” mechanism) and metabolism. It has been found to extend lifespan in multiple organisms.
It is FDA approved as an immunosuppressant for use in organ transplants when taken daily. Taken once a week it seems to be an immune modulator and have beneficial effects though its use in humans for longevity is still experimental. [Rapamycin Dosing: 0.1 mg / kg once weekly to be cycled for three months on and one month off, TruAge testing before and six months after].
Metformin ER (1 gm) – Taken in the evening (after dinner): Metformin, primarily a medication for type 2 diabetes, is being investigated for its potential longevity benefits due to its ability to decrease insulin resistance, reduce inflammation, and promote healthy aging.
Its mechanisms are not fully understood, but they may involve changes in metabolic and cellular processes such as activation of AMPK which promotes autophagy and cleanup of cellular debris that helps to delay the onset of age-related diseases.
CJC / Ipamorelin – Peptide to boost IGF-1/Growth Hormone, taken in a specific on and off cycle: These peptides stimulate the release of growth hormone (GH) from the pituitary gland, which in turn promotes the production of insulin-like growth factor 1 (IGF-1) in the liver. GH and IGF-1 can contribute to tissue repair, muscle growth, bone density, and improved metabolism. However, their role in longevity is complex as both low and high levels of IGF-1 have been associated with lifespan in various studies.
Testosterone Optimization – 2x Per Week: Testosterone optimization, often used as part of hormone replacement therapy, may have several benefits related to longevity. Adequate testosterone levels are associated with increased muscle mass, bone density, mood stability, cognitive function, and cardiovascular health.
However, maintaining an optimal balance is crucial, as both low and high levels can have potential negative effects on health and longevity. Based upon my lab levels, I take a 0.15 ml injection twice per week.
My Nootropic Medication
I’m often flying around the world, and as much as I don’t like anything less than eight hours of sleep, many times if I’m across the continent or the globe and need to go on stage, I will sometimes use a nootropic to focus my mind.
Nootropics, often referred to as “smart drugs” or “cognitive enhancers,” are substances that aim to improve cognitive function, particularly executive functions like memory, creativity, or motivation, in healthy individuals.
While caffeine is a commonly used nootropic, I’ve reduced my intake to one cup every two or three days. What I have found works very well for me is a prescription medication known as Modafinil (or Provigil). My typical dose is 100 mg.
Modafinil is a medication that promotes wakefulness. It’s most commonly used to treat sleep disorders such as narcolepsy, obstructive sleep apnea, and shift work sleep disorder. Despite its primary use, Modafinil has also gained popularity as a nootropic due to its potential to enhance cognition, particularly in terms of increasing alertness and reducing fatigue.
The exact mechanism of action of Modafinil is not fully understood. It’s known to affect various neurotransmitters in the brain: chemicals that neurons use to communicate with each other. Some of the key neurotransmitters affected by Modafinil include dopamine, norepinephrine, histamine, and orexin (hypocretin).
Modafinil is thought to increase the availability of these neurotransmitters in parts of the brain that control wakefulness and alertness. For instance, it’s known to inhibit the reuptake of dopamine, which increases the amount of dopamine available in the brain. This is a similar mechanism to some stimulants, but Modafinil doesn’t tend to cause the same level of overstimulation or potential for addiction.
While Modafinil can be beneficial, it’s not without risks and potential side effects. Some of the common side effects include headache, nausea, anxiety and insomnia.
Personally, I haven’t experienced any of these side effects, except for insomnia if the Modafinil is taken within eight hours of sleep. Modafinil should not be used when consuming alcohol. It’s also important to remember that Modafinil doesn’t replace the need for sleep.
My Skin Care
My favorite skin product is called OneSkin, a product I use every day, twice per day. OneSkin’s product OS-1 addresses the root cause of skin aging, specifically senescent cells, which are damaged cells that build up in the body, contributing to aging and age-related disease. As senescent cells accumulate in our skin, they create wrinkles and sagging, produce inflammation, and also make us more susceptible to skin cancer.
OS-1 is a 20 amino acid peptide. OneSkin’s experiments have shown that this proprietary peptide can significantly decrease the level of senescent cells, reducing the age of the skin by several years at a molecular level.
My Supplements
In addition to Lifeforce supplements/products listed above, following are supplements that I take as part of my Longevity Practices, at various times depending on my medical testing. You can evaluate which are appropriate for you, and at what dosages.
Creatine (Morning of Workouts): Creatine helps to generate ATP in skeletal muscles which is the body’s fuel source, it thus has been shown to enhance muscle strength, power, endurance and exercise performance which can contribute to maintaining overall physical function and mobility as we age, thereby promoting longevity. Since your body uses a lot of resources to make creatine, it can be helpful to take it to free up some of those resources.
Alpha Lipoic Acid: Alpha lipoic acid is a potent antioxidant that helps protect cells from oxidative stress. By neutralizing free radicals and supporting cellular, especially mitochondrial health, it may contribute to longevity by reducing the risk of age-related damage.
Selenomethionine: Selenomethionine is a form of selenium, a trace mineral that acts as an antioxidant and supports immune function. Adequate selenium levels have been associated with reduced risk of certain chronic diseases, potentially promoting longevity. You can usually get selenium from your food, but since many agricultural soils are deficient in selenium it can be important to supplement.
Lion’s Mane: Lion’s Mane is a mushroom with potential neuroprotective effects. It may support brain health by promoting the production of nerve growth factors and enhancing cognitive function, which could contribute to maintaining mental acuity and longevity.
Vitamin D w/K2: Vitamin D plays a crucial role in bone health and immune function. When combined with vitamin K2, it helps ensure calcium is properly utilized and deposited in bones, potentially reducing the risk of fractures and supporting overall health as we age. Vitamin K2 has also been shown to reduce arterial stiffness, by decreasing microcalcifications and lower the incidence of diabetes and cardiovascular disease.
Seed / Probiotic (30 Billion Bacteria): A healthy gut microbiome is essential for various aspects of health. A high-quality seed / probiotic supplement with a diverse range of beneficial bacteria can help support digestion, immune function, and potentially improve overall health and longevity.
CoQ10 w/PQQ: Coenzyme Q10 (CoQ10) and Pyrroloquinoline quinone (PQQ) are both involved in cellular energy production and have antioxidant properties. These compounds may help protect mitochondria, the energy factories of cells, and support overall cellular health, potentially promoting longevity. Since statin drugs inhibit the enzyme that makes CoQ10 in our bodies, it is important to supplement with CoQ10 whenever taking a statin.
Quercetin: Quercetin is a flavonoid with antioxidant and anti-inflammatory properties. It may help reduce oxidative stress, inflammation, and support cardiovascular health, which can contribute to overall longevity.
Xymogen (Methyl Protect): Xymogen’s Methyl Protect is a supplement containing B vitamins that is designed to support methylation, a key biochemical process involved in gene expression and cellular function. By optimizing methylation, it may help promote overall health and potentially contribute to longevity.
Magnesium Threonate & Magnesium Glycinate: Magnesium is an essential mineral involved in numerous physiological processes. Magnesium threonate can get into the brain and thus has been suggested to enhance brain function and support cognitive health, while magnesium glycinate may help with relaxation and sleep. By promoting brain and overall well-being, they may indirectly contribute to longevity.
ProdromeNeuro & ProdromeGlia: ProdromeNeuro and ProdromeGlia are supplements designed to support brain health and protect against neurodegenerative conditions. By providing nutrients and compounds that promote brain function and reduce oxidative stress, they may help maintain cognitive vitality and potentially contribute to longevity.
Arterosil: Arterosil is a supplement that has been shown to regenerate the endothelial glycocalyx which is the protective lining inside all of our blood vessels. It thus supports endothelial function, which is important for maintaining healthy blood vessels. By promoting proper blood flow and reducing the risk of cardiovascular disease, Arterosil may contribute to overall longevity by supporting heart health.
Taurine: Taurine is amazing for mitochondrial health, mitochondrial proteostasis, nfr2 upregulation and MTP pore control. I take 1,000 mg per day, five days per week.
NMN (Nicotinamide mononucleotide): NMN is a critical precursor to NAD+ (more below). I take 1 gram of NMN per day. I also take a product called Nuchido TIME+ which activates the NAD+ recycling pathway by activating NAMPT.
MORE ABOUT NMN: NMN is a molecule that plays a crucial role in our body’s energy production process. It’s a precursor to NAD+ (nicotinamide adenine dinucleotide), a vital coenzyme present in every cell of our body, that plays a critical role in energy metabolism and maintaining proper cell functioning.
Research has shown a connection between NMN, NAD+ levels, and aging. As we age, the levels of NAD+ in our bodies tend to decrease, and this decrease is believed to be linked to various aspects of aging and age-related health issues. Here’s why NMN is important in the context of aging and longevity:
1. Cellular Energy Production: NMN helps produce NAD+, which is critical in the conversion of nutrients into usable energy. As we age, lower NAD+ levels can contribute to fatigue and other age-related health concerns.
2. DNA Repair: NAD+ assists in the repair of broken DNA strands, a process that becomes less efficient as we age. Therefore, maintaining NAD+ levels through NMN supplementation could potentially support DNA repair.
Sirtuin Activation: Sirtuins are a group of proteins believed to play a role in aging by controlling cellular health. They are dependent on NAD+, so more NMN might mean better functioning of these proteins.
4. Mitochondrial Health: NAD+ is important for mitochondrial function, and mitochondria are the powerhouses of cells, producing the energy necessary for cellular functions. Aging is associated with a decrease in mitochondrial function, so maintaining NAD+ levels with NMN could help maintain mitochondrial health.
5. Neuroprotection: Some research suggests that NAD+ can help protect neurons from age-related decline, potentially slowing the onset of neurodegenerative diseases.
It’s important to note that while these supplements have shown potential benefits, individual results may vary, and it’s always recommended to consult with a healthcare professional before starting any new supplement regimen.
My Therapeutics (I’m Considering)
Following are the therapeutic protocols that I am either participating in, or investigating under the Fountain Life Edge Membership. Note some of these are still in development.
Peptide Protocols: I’m looking at various peptide therapies aimed to improve a range of physiological processes including metabolism, growth and development, immune function, and tissue repair. (See the following pages for more details.)
Senolytics Protocol: I’m evaluating senolytic protocols aimed at eliminating senescent cells from the body and reducing the harmful effects they can have on tissues and organs.
Stem Cell Treatment:sd Stem cells are the precursor to all other cells in your body, they have the potential to repair, restore, replace, and regenerate cells, and could be used to treat many medical conditions and diseases.
Today, the use of allogeneic stem cells (meaning stem cells from another individual, such as an umbilical cord derived stem cell treatment) are not approved by the FDA and are therefore not legal in the United States. Fountain Life has evaluated multiple centers outside the U.S. including in Mexico, Panama, the Caribbean and Costa Rica.
Currently we advise our Fountain Members interested in Stem Cells to work with the Regenerative Medicine Institute (RMI) in Costa Rica (https://rmi-international.com/). Fountain Life is currently in development of a number of FDA Protocols that will be delivered to our members in our Orlando HQ under an “Investigational New Drug” protocol once approved in late 2023.
Exosomes: Exosomes are small vesicles containing growth and signaling factors produced by stem cells, used to communicate and stimulate repair and growth. Treatment with exosomes is a versatile and precisely targeted therapy that can be used to address conditions such as chronic pain, osteoarthritis, and musculoskeletal injuries. Currently, exosomes are not approved by the FDA and their use is investigational.
I have used exosomes both locally, i.e. injection at sites of inflammation, and in my shoulder post-surgical repair of my rotator cuff. I’ve also used it to stimulate follicular growth on my scalp. Exosomes have also been used for an overall regenerative therapeutic when given intravenously. Today, exosomes are likely to be provided through Fountain Life under an IRB approved clinical trial.
Total Plasma Exchange (TPE): Total plasma exchange (TPE) is a medical procedure that removes and replaces the plasma in your blood. Plasma is the liquid part of blood that contains proteins, antibodies, and other substances. TPE is used to treat a variety of diseases, including: autoimmune diseases, neurological disorders, hematological disorders and poisoning (wherein TPE can remove toxins that have been ingested or inhaled).
TPE is not a cure for any disease, but it can be used to improve symptoms and prolong life. There is some evidence that TPE may have pro-longevity benefits. It’s also suggested that with aging, our blood accumulates harmful substances, which could possibly be removed with plasma exchange, potentially slowing down the aging process. For example, one study found that TPE increased the lifespan of mice by 30%. However, more research is needed to confirm these findings in humans.
The Typical TPE Protocol Proposed By Dr. Dobri Kiprov (Who Pioneered Much Of This Work) For Longevity Benefits Is As Follows:
* Frequency: The protocol calls for TPE to be performed once a week for a total of 12 weeks. * Volume: Each TPE session removes 1.5 liters of plasma. * Replacement Fluid: The replacement fluid is a mixture of 5% human albumin and saline. * Indication: The protocol is indicated for people who are interested in improving their healthspan and longevity.
Dr. Kiprov believes that TPE can help to slow down the aging process by removing harmful proteins and other substances from the blood. He has conducted several studies that have shown that TPE can improve cognitive function, reduce inflammation, and increase lifespan in animals. Here are some of the potential benefits of TPE for longevity:
Reduced inflammation: Inflammation is a major factor in aging and age-related diseases. TPE can help to reduce inflammation by removing harmful proteins and other substances from the blood.
Improved Cognitive Function: TPE has been shown to improve cognitive function in animals and humans. This is likely due to the removal of harmful proteins that can damage brain cells.
Increased Lifespan: As mentioned above, several studies have shown that TPE can increase lifespan in animals. However, this has not yet been proven in humans.
Stem Cell Banking: Partnering with private, state-of-the art facilities, Fountain Life is facilitating its members in the collection, processing, and storing of their own “fat derived” and “bone-marrow” derived stem cells.
Biobanking: Biobanking of serum, plasma, whole cells (WBC), microbiome, saliva and urine. Partnering with a private, state-of-the-art biobank, we will facilitate the process of collecting, processing, storing, and distributing various biological samples for future purposes. Peptide Protocols (More Details)
Under guidance from the Fountain Life Medical team, I’m considering and sometimes using peptides. Peptides are short chains of amino acids, the building blocks of proteins. In medicine, peptides can be used as therapies for a variety of conditions. They are typically injected into the body, although other administration methods are also used.
Peptides can serve a range of functions. Some act like hormones and others like neurotransmitters. Many control and influence how our bodies react to diet and physical exercise.
Following Are The Peptides I’ve Used And / Or Am Considering:
Peptide: BPC 157, composed of 15 amino acids, is a partial sequence of body protection compound (BPC) that is discovered in and isolated from human gastric juice. Experimentally it has been demonstrated to accelerate the healing of many different wounds, including tendons, muscles, nervous system and superior healing of damaged ligaments. Those who suffer from discomfort due to muscle sprains, tears and damage may benefit from treatment with this peptide.
It can also help aid skin burns to heal at a faster rate and increase blood flow to damaged tissues. Animal studies suggest that BPC-157 has strong anti-inflammatory activity.
Common Formulas & Protocols:
BPC-157 (10 mg) Lyophilized Vial – 300 mcg SQ injection nightly for thirty days BPC-157 (500 mcg) Capsule – One capsule daily for thirty days. BPC-157 (500 mcg) Sublingual Troche – One troche daily for thirty days.
Peptide: Selank
Selank is a synthetic peptide that was developed by the Institute of Molecular Genetics of the Russian Academy of Sciences. It’s based on a naturally occurring immunomodulatory peptide called Tuftsin, which plays an important role in immune system responses.
Selank is known for its potential nootropic and anxiolytic (anti-anxiety) effects. It has been studied for its potential to improve anxiety, enhance cognitive functions, and modulate the immune system. Here are some specific areas of research:
Anxiety and depression: Selank has been studied for its potential to reduce anxiety and symptoms of depression. It appears to modulate the expression of GABA, which is one of the main inhibitory neurotransmitters in the human brain.
Cognitive Function: Some research has suggested that Selank could improve cognitive function. It’s thought that it may enhance memory and learning ability, though more research is needed in this area.
Immune modulation: Selank is derived from the peptide Tuftsin, which is known to modulate the immune system. As such, Selank may also have immune-modulating properties.
Common Formulas & Protocols:
Selank (7500 mcg/ml) Nasal Spray 3ml Bottle – 750 mcg (1 spray) each nostril daily.
Peptide: Semax
Semax increases brain-derived neurotrophic factor (BDNF) levels. BDNF is among the most active neurotrophins, which are chemicals that help to stimulate and control neurogenesis, the birth of new neurons in the brain. BDNF has been shown to play a role in neuroplasticity, which allows nerve cells in the brain to compensate for injury and adapt to new situations or changes in the environment.
Basically, BDNF helps to support the survival of existing neurons and encourages the growth, regeneration, and creation of new neurons and synapses.
The understood benefits of Semax Include: improving of cognitive function; improving long and short-term memory; helping to manage depression; improving non-proliferative diabetic neuropathy recovery from stroke/hypoxia; improving glaucoma optic neuropathy. Semax may also be helpful in the treatment of PTSD & ADHD neuroprotection.
Common Formulas & Protocols: Semax (7500 mcg/ml) Nasal Spray 3ml Bottle – 750 mcg (1 spray) each nostril daily.
Peptide: CJC-1295 / Ipamorelin:
CJC 1295 is often prescribed by physicians as a growth hormone releasing hormone (GHRH) analog. CJC 1295 has been shown to increase growth hormone as well as IGF-I secretion and it has been able to do so in very large amounts.
CJC 1295 stimulates growth hormone secretion and will keep a steady increase of HGH and IGF-1 with no increase in prolactin, leading to fat loss, and increased protein synthesis thereby promoting growth.
We suggest using the CJC 1295 in combination with Ipamorelin as it provides a synergistic effect, generating five times the benefits of using the CJC 1295 or Ipamorelin alone.
The combination allows for maximized release of GH because the CJC 1295 and Ipamorelin have different mechanisms of action and work on different receptors (GHRH-R & Ghrelin-R).
Subcutaneous administration of CJC 1295 resulted in sustained, dose-dependent increases in GH and IGF-I levels in healthy adults and was safe and relatively well tolerated, particularly at doses of 30 or 60 ug/ kg. There was evidence of a cumulative effect after multiple doses.
Related Articles:
The Complete Guide To The Science Of Circadian Rhythms (#GotBitcoin?)
Billionaire is Turning Heads With Novel Approach To Fighting Cancer (#GotBitcoin?)
Fighting Cancer By Releasing The Brakes On The Immune System (#GotBitcoin?)
Over-Diagnosis And Over-Treatment Of Cancer In America Reaches Crisis Levels (#GotBitcoin?)
Cancer Super-Survivors Use Their Own Bodies To Fight The Disease (#GotBitcoin?)
The Benefits of Grounding or Earthing For Improved Health
Microbiome Live News
@metagenomics










Leave a Reply
You must be logged in to post a comment.