Related Articles:
Bitcoin Information & Resources (#GotBitcoin?)
Ultimate Resource For News, Breakthroughs And Innovations In Healthcare
Slow-Twitch vs. Fast-Twitch Muscle Fibers
Chrono-Pharmacology Reveals That “When” You Take Your Medication Can Make A Life-Saving Difference
Jarlsberg Cheese Offers Significant Bone & Heart-Health Benefits Thanks To Vitamin K2, Says Study
Ultimate Resource For Cat Lovers
New Evidence For The Vibration Theory Of Smell
Why You Shouldn’t Buy A Low-End Home Security Camera
Ultimate Resource On Commodity Futures Trading Commission (CFTC) And It’s Impact On Bitcoin
Top Institutions And Individuals Trying To Destroy Bitcoin
Ultimate Resource On Online Streaming Services
Large Chinese Bank Protest Put Down With Violence (#GotBitcoin)
The NASA Engineer Who Made The James Webb Space Telescope Work
Ultimate Resource On Brittney Griner Being Held In Russian Jail
Workplace Hazards: What Are The Risks Surrounding Electricians?
“Better Days Ahead With Crypto Deleveraging Coming To An End” — Joker
What Happens When You Put Regular Gas In a Premium Car?
Amazon Prime Rewards “5% Back” Visa Signature Card Review
When World’s Central Banks Get It Wrong, Guess Who Pays The Price???????????? (#GotBitcoin)
Crypto Market Is Closer To A Bottom Than Stocks (#GotBitcoin)
Axon Ditches Plans For Weaponized Taser Drones As Majority Of Ethics Board Resigns
Israeli Tech Firm Rolls Out Tracking Devices The Size Of Postage Stamps
Gas Station Owner In Massachusetts Shuts Pumps To Protest Prices
Fed Money Printer Goes Into Reverse (Quantitative Tightening): What Does It Mean For Crypto?
Mocked As ‘Rubble’ By Biden, Russia’s Ruble Roars Back
Anti-ESG Movement Reveals How Blackrock Pulls-off World’s Largest Ponzi Scheme
$35 TRILLION Global Stock Market Meltdown!!! (#GotBitcoin)
Citi Trader Is Scapegoat For Flash Crash In Europe Stocks (#GotBitcoin)
Building And Running Businesses In The ‘Spirit Of Bitcoin’
Will Community Group Buying Work In The US?
Sri Lanka’s And Lebanon’s Defaults Could Be The First Of Many (#GotBitcoin)
Ultimate Resource On Bitcoin Price Manipulation By Wall Street
Ultimate Resource On Russians Oligarchs And The Impact Of Sanctions On Them
Ultimate Resource For The Crisis Taking Place In The Nickel Market
Does Your Doctor or Hospital Have A Financial Relationship With Big Pharma?
Former World Bank Chief Didn’t Act On Warnings Of Sexual Harassment
AI Experts Warn of Potential Cyberwar Facing Banking Sector vs Bitcoin Which Can Be Stored Off-line
Archaeologists Uncover Five Tombs In Egypt’s Saqqara Necropolis
What The Fed’s Rate Hike Means For Inflation, Housing, Crypto And Stocks
Unusual Side Hustles You May Not Have Thought Of
Happy International Women’s Day! Leaders Share Their Experiences In Crypto
Russia’s Independent Journalists Including Those Who Revealed The Pandora Papers Need Your Help
The Miracle Of Blockchain’s Triple Entry Accounting
Catawba, Native-American Tribe Approves First Digital Economic Zone In The United States
Ultimate Resource On Music And NFTs And The Implications For The Entertainment Industry
Traders Prefer Gold, Fiat Safe Havens Over Bitcoin As Russia Goes To War
Ultimate Resource On Music Catalog Deals
Why We Should Welcome Another Crypto Winter
Canada’s Major Banks Go Offline In Mysterious (Bank Run?) Hours-Long Outage (#GotBitcoin)
The Ultimate Resource For The Bitcoin Miner And The Mining Industry (Page#2) #GotBitcoin
Investors Pool Funds To Help Poor People Sue Big Companies
100 Million Americans Can Legally Bet on the Super Bowl. A Spot Bitcoin ETF? Forget About it!
‘I Cry Every Day’: Olympic Athletes Slam Food, COVID Tests And Conditions In Beijing
Ultimate Resource On Myanmar’s Involvement With Crypto-Currencies
Bitcoin For Corporations | Michael Saylor | Bitcoin Corporate Strategy
Japan’s $1 Trillion Crypto Market May Ease Onerous Listing Rules
How Bitcoin Contributions Funded A $1.4M Solar Installation In Zimbabwe
BREAKING: Arizona State Senator Introduces Bill To Make Bitcoin Legal Tender
Vast Troves of Classified Info Undermine National Security, Spy Chief Says
Inflation And A Tale of Cantillionaires
America COMPETES Act Would Be Disastrous For Bitcoin Cryptocurrency And More
Petition Calling For Resignation Of U.S. Securities/Exchange Commission Chair Gary Gensler
McDonald’s Jumps On Bitcoin Memewagon, Crypto Twitter Responds
Smart Money Is Buying Bitcoin Dip. Stocks, Not So Much
Henrietta Lacks And Her Remarkable Cells Will Finally See Some Payback
Stealing The Blood Of The Young May Make You More Youthful
Doctors Show Implicit Bias Towards Black Patients
Indexing Is Coming To Crypto Funds Via Decentralized Exchanges
Imagine There’s No Bitcoin. If You Can, Then We Haven’t Done Our Jobs
US Stocks Historically Deliver Strong Gains In Fed Hike Cycles (GotBitcoin)
Federal Regulator Says Credit Unions Can Partner With Crypto Providers
Jack Dorsey Announces Bitcoin Legal Defense Fund
Four Ways Black Families Can Fight Against Rising Inflation (#GotBitcoin)
Bitcoin’s Dominance of Crypto Payments Is Starting To Erode
Walmart Filings Reveal Plans To Create Cryptocurrency, NFTs
Ultimate Resource On Duke of York’s Prince Andrew And His Sex Scandal
FDA Approves First-Ever Arthritis Pain Management Drug For Cats
Is Art Therapy The Path To Mental Well-Being?
Arkansas Tries A New Strategy To Lure Tech Workers: Free Bitcoin
Wordle Is The New Lingo Turning Fans Into Argumentative Strategy Nerds
Nas Selling Rights To Two Songs Via Crypto Music Startup Royal
Teen Cyber Prodigy Stumbled Onto Flaw Letting Him Hijack Teslas
Joe Rogan: I Have A Lot Of Hope For Bitcoin
How Black Businesses Can Prosper From Targeting A Trillion-Dollar Black Culture Market (#GotBitcoin)
‘Yellowstone’ Is A Huge Hit That Started With Small-Town Fans
Sidney Poitier, Actor Who Made Oscars History, Dies At 94
How Jessica Simpson Almost Lost Her Name And Her Billion Dollar Empire
Ultimate Resource On Solana Outages And DDoS Attacks
Ultimate Resource On Kazakhstan As Second In Bitcoin Mining Hash Rate In The World After US
Nasdaq-Listed Blockchain Firm BTCS To Offer Dividend In Bitcoin; Shares Surge
Bitcoin Enthusiast And CEO Brian Armstrong Buys Los Angeles Home For $133 Million
Ultimate Resource On A Strong Dollar’s Impact On Bitcoin
Ultimate Resource On Bitcoin Unit Bias
Yosemite Is Forcing Native American Homeowners To Leave Without Compensation. Here’s Why
Raoul Pal Believes Institutions Have Finished Taking Profits As Year Winds Up
10 Women Who Used Crypto To Make A Difference In 2021
Disposable Masks That Don’t Pollute
Move Over, Tennis And Golf. Networks And Brands Are Cashing In On Pickleball
Athletes Unlimited Signs Big Sponsor In Boost For Women’s Sports
Grounded By The Pandemic? Donate Your Unused Points And Miles
Crypto And Its Many Fees: What To Know About The Hidden Costs Of Digital Currency
Deputizing Blockchain To Fight Corrupt Governments. Also America Blows It’s Tokenization Advantage
Crypto Attracts More Money In 2021 Than All Previous Years Combined
The US Shouldn’t Be Afraid of China
Ultimate Resource On Donald J. Trump
Ubiquitous Surveillance And Security
Finding Your Superpower In A Higher-Priced World With On-The-Spot Comparison Shopping
How remanufacturing is combating global warming
Shoppers Lined Up At Dawn For The ‘Open Run’ On $9,500 Chanel Bags
How Banks Win When Interest Rates Rise (#GotBitcoin)
Crypto, NFTs And Tungsten Cubes: A Guide To Giving Cash In 2021
Tech Giants Apple, Microsoft, Amazon And Others Warn of Widespread Software Flaw
Ultimate Resource On Web3 And Crypto’s Attempt To Reinvent The Internet
Bitcoin Core Developer Samuel Dobson Decides It Is ‘Time To Go’
The Biden Economy Is Actually Pretty Good
Live: House Memo Details Congress’ Priorities Ahead Of Crypto CEO Hearing
Verizon is Tracking iPhone Users by Default And There’s Nothing Apple Can Do. How to Turn It Off
A Plant-based Pet Food Frenzy Is On The Horizon
What Is Cryptocurrency, And How Does It Work?
One Of The Weirdest Reports: Investors React To 12-3-2021 Jobs Data (#GotBitcoin)
Ultimate Resource On China’s ‘Common Prosperity’ Drive How It Plans To Redistribute The Wealth
Note To Brands: Crypto Isn’t Funny Money. It’s Community
How Crypto Vigilantes Are Hunting Scams In A $100 Billion Market
6 Questions For Lyn Alden Schwartzer Of Lyn Alden Investment Strategy
Bitcoin Offers Little Refuge From Covid Market Rout
The Anti-Work Brigade Targets Amazon On Black Friday
Reddit’s Latest Money-Making Obsession Is An Obscure Fed Facility (Reverse Repo or RRP)
Federal Reserve To Taper Money Printing That Fueled Bitcoin Rally
Apple Sues NSO Group To Curb The Abuse Of State-Sponsored Spyware
Did You Buy Your Cat or Dog A Christmas Present? Join The Club
Gen Z Has $360 Billion To Spend, Trick Is Getting Them To Buy (#GotBitcoin)
10 Fundamental Rights For All Crypto Users Including Responsibilities For The Industry
Texas Plans To Become The Bitcoin Capital, Vulnerable Power Grid And All
Bitcoin Caught Between Longer-Term Buyers, Leveraged Speculators
Outcry Grows As China Breaks Silence On Missing Tennis Star
China Left In Shock Following Brutal Killing Of Corgi During Covid-19 Disinfection
Biden Orders Feds To Tackle ‘Epidemic’ Of Missing Or Murdered Indigenous People
An Antidote To Inflation? ‘Buy Nothing’ Groups Gain Popularity
Ultimate Resource For Cooks, Chefs And The Latest Food Trends
Open Enrollment Gameplan: How To Compare Your Partner’s Health Insurance With Yours
Millions Of Americans Are Skipping The Dentist. Lenders See A Financing Niche
Matt Damon To Promote Crypto.Com In Race To Lure New Users
US General Likens China’s Hypersonic Missile Test To A ‘Sputnik Moment’
The Key To Tracking Corona And Other Any Virus Could Be Your Poo
How To Evade A Chinese Bitcoin Ban
US Government Failed To Spot Pedophile At Indian Health Service Hospitals
Netflix Defends Dave Chappelle While Seeing “Squid Game” Become It’s Biggest Hit Ever
Ultimate Resource On Various Countries Adopting Bitcoin
Ultimate Resource On Bitcoin Billionaires
Stripe Re-Enters Processing Payments Using Cryptos 3 Years After Dropping Bitcoin
Why Is My Cat Rubbing His Face In Ants?
Pets Score Company Perks As The ‘New Dependents’
What Pet Owners Should Know About Chronic Kidney Disease In Dogs And Cats
The Facebook Whistleblower, Frances Haugen, Says She Wants To Fix The Company, Not Harm It
Walk-in Cryptocurrency Exchanges Emerge Amid Bitcoin Boom
Europe’s Giant Job-Saving Experiment Pays Off In Pandemic
To Survive The Pandemic, Entrepreneurs Might Try Learning From Nature
Rise of The FinFluencer And How They Target Young And Inexperienced Investors
Hyperinflation Concerns Top The Worry List For UBS Clients
Sotheby’s Opens First-Ever Exhibition of Black Jewelry Designers
America And Europe Face Bleak Winter As Energy Prices Surge To Record Levels
Ultimate Resource On Vaccine Boosters
Unvaccinated COVID-19 Hospitalizations Cost The U.S. Health System Billions Of Dollars
Wallets Are Over. Your Phone Is Your Everything Now
Crypto Mining Demand Soars In Vietnam Amid Bitcoin Rally
How The Supreme Court Texas Abortion Ruling Spurred A Wave Of ‘Rage Giving’
Ultimate Resource On Global Inflation And Rising Interest Rates (#GotBitcoin)
Presearch Decentralized Crypto-Powered Search Engine
How To Make Vietnam A Powerful Trade Ally For The U.S.
Banking Could Go The Way of News Publishing (#GotBitcoin)
Operation “Choke Point”: An Aggressive FTC And The Response of The Payment Systems Industry
Bezos-Backed Fusion Startup Picks U.K. To Build First Plant
TikTok Is The Place To Go For Financial Advice If You’re A Young Adult
More Companies Weigh Penalizing Employees Without Covid-19 Vaccinations
Crypto Firms Want Fed Payment Systems Access—And Banks Are Resisting
Travel Expert Oneika Raymond’s Favorite Movies And Shows
How To Travel Luxuriously In The Summer Of Covid-19, From Private Jets To Hotel Buyouts
Nurses Travel From Coronavirus Hot Spot To Hot Spot, From New York To Texas
Travel Is Bouncing Back From Coronavirus, But Tourists Told To Stick Close To Home
Tricks For Making A Vacation Feel Longer—And More Fulfilling
Who Is A Perpetual Traveler (AKA Digital Nomad) Under The US Tax Code
Four Stories Of How People Traveled During Covid
Director Barry Jenkins Is The Travel Nerd’s Travel Nerd
Why Is My Cat Rubbing His Face In Ants?
Natural Cure For Hyperthyroidism In Cats Including How To Switch Him/Her To A Raw Food Diet
A Paycheck In Crypto? There Might Be Some Headaches
About 46 Million Americans Or 17% Now Own Bitcoin vs 50% Who Own Stocks
Josephine Baker Is 1st Black Woman Given Paris Burial Honor
Cryptocurrency’s Surge Leaves Global Watchdogs Trying To Catch Up
It Could Just Be The U.S. Is Not The Center Of The Crypto Universe
Blue Turmeric Is The Latest Super Spice To Shake Up Pantry Shelves
Vice President Kamala Harris To Focus On Countering China On Southeast Asia Trip
The Vaccinated Are Worried And Scientists Don’t Have Answers
Silver Lining of Coronavirus, Return of Animals, Clear Skies, Quiet Streets And Tranquil Shores
Cocoa Cartel Stirs Up Global Chocolate Market With Increased Use Of Child Labor
Killer Whale Dies Suddenly At SeaWorld San Diego
Does Getting Stoned Help You Get Toned? Gym Rats Embrace Marijuana
Hope Wiseman Is The Youngest Black Woman Dispensary Owner In The United States
What Sex Workers Want To Do With Bitcoin
How A Video Résumé Can Get You Hired In The Covid-19 Job Market
US Lawmakers Urge CFTC And SEC To Form Joint Working Group On Digital Assets
Bitcoin’s Surge Lacks Extreme Leverage That Powered Past Rallies
JP Morgan Says, “Proof-Of-Stake Will Eat Proof-Of-Work For Breakfast — Here’s Why
Ranking The Currencies That Could Unseat The Dollar (#GotBitcoin)
The Botanist Daring To Ask: What If Plants Have Intelligence?
After A Year Without Rowdy Tourists, European Cities Want To Keep It That Way
Prospering In The Pandemic, Some Feel Financial Guilt And Gratitude
The Secret Club For Billionaires Who Care About Climate Change
A List Of Relief Funds For Restaurants, Bars, And Food Service Workers
Bill And Melinda Gates Welcome The Philanthropists Of The Future
CO2-Capture Plan Using Old Oil Reservoirs In Denmark Moves Ahead
Could This Be The Digital Nose Of The Future?
Bitcoin Fans Are Suddenly A Political Force
Walmart Seeks Crypto Product Lead To Drive Digital Currency Strategy
Music Distributor DistroKid Raises Money At $1.3 Billion Valuation
Books To Read, Foods To Eat, Movies To Watch, Exercises To Do And More During Covid19 Lockdown
Hacker Claims To Steal Data Of 100 Million T-Mobile Customers
Is The Cryptocurrency Epicenter Moving Away From East Asia?
Vietnam Leads Crypto Adoption In Finder’s 27-Country Survey
Bitcoin’s Latest Surge Lacks Extreme Leverage That Powered Past Rallies
Creatine Supplementation And Brain Health
Nigerian State Says 337 Students Missing After Gunmen Attack
America’s 690 Mile-Long Yard Sale Entices A Nation of Deal Hunters
Russian Economy Grew (10.3% GDP) Fastest Since 2000 On Lockdown Rebound
US Troops Going Hungry (Food Insecurity) Is A National Disgrace
Overheated, Unprepared And Under-protected: Climate Change Is Killing People, Pets And Crops
Accenture Confirms Hack After LockBit Ransomware Data Leak Threats
Climate Change Prompts These Six Pests To Come And Eat Your Crops
Leaked EU Plan To Green Its Timber Industry Sparks Firestorm
Students Take To The Streets For Day Of Action On Climate Change
Living In Puerto Rico, Where The Taxes Are Low And Crypto Thrives
Bitcoin Community Leaders Join Longevity Movement
Some Climate Change Effects May Be Irreversible, U.N. Panel Says
Israel’s Mossad Intelligence Agency Is Seeking To Hire A Crypto Expert
What You Should Know About ‘529’ Education-Savings Accounts
Bitcoin Doesn’t Need Presidents, But Presidents Need Bitcoin
Plant-Based Fish Is Rattling The Multibillion-Dollar Seafood Industry
The Scientific Thrill Of The Charcoal Grill
Escaping The Efficiency Trap—and Finding Some Peace of Mind
Sotheby’s Selling Exhibition Celebrates 21 Black Jewelry Designers
Marketers Plan Giveaways For Covid-19 Vaccine Recipients
Haiti’s President Moise Assassinated In Night Attack On His Home
The Argentine River That Carries Soybeans To World Is Drying Up
A Harvard Deal Tries To Break The Charmed Circle Of White Wealth
China Three-Child Policy Aims To Rejuvenate Aging Population
California Wants Its Salton Sea Located In The Imperial Valley To Be ‘Lithium Valley’
Who Will Win The Metaverse? Not Mark Zuckerberg or Facebook
The Biggest Challenge For Crypto Exchanges Is Global Price Fragmentation
London Block Exchange – LBX Buy, Sell & Trade Cryptocurrencies?
Chicago World’s Fair Of Money To Unveil A Private Coin Collection Worth Millions of Dollars
Bitcoin Dominance On The Rise Once Again As Crypto Market Rallies
Uruguayan Senator Introduces Bill To Enable Use Of Crypto For Payments
Talen Energy Investors Await Update From The Top After Pivot To Crypto
Ultimate Resource On Hydrogen And Green Hydrogen As Alternative Energy
Covid Made The Chief Medical Officer A C-Suite Must
When Will Stocks Drop? Watch Profit Margins (And Get Nervous)
Behind The Rise Of U.S. Solar Power, A Mountain of Chinese Coal
‘Buy Now, Pay Later’ Installment Plans Are Having A Moment AgainUS Crypto Traders Evade Offshore Exchange Bans
5 Easy Ways Crypto Investors Can Make Money Without Needing To Trade
What’s A ‘Pingdemic’ And Why Is The U.K. Having One?
“Crypto-Property:” Ohio Court Says Crypto-Currency Is Personal Property Under Homeowners’ Policy
Retinal And/Or Brain Photon Emissions
Antibiotic Makers Concede In The War On Superbugs (#GotColloidalSilver)
What It Takes To Reconnect Black Communities Torn Apart by Highways
Those Probiotics May Actually Be Hurting Your ‘Gut Health’ We’re Not Prepared To Live In This Surveillance Society
How To Create NFTs On The Bitcoin Blockchain
World Health Organization Forced Valium Into Israeli And Palestinian Water Supply
Blockchain Fail-Safes In Space: Spacechain, Blockstream And Cryptosat
What Is A Digital Nomad And How Do You Become One?
Flying Private Is Cheaper Than You Think — Here Are 6 Airlines To Consider For Your Next Flight
What Hackers Can Learn About You From Your Social-Media Profile
How To Protect Your Online Privacy While Working From Home
Rising Diaper Prices Prompt States To Get Behind Push To Pay
Want To Invest In Cybersecurity? Here Are Some ETFs To Consider
US Drops Visa Fraud Cases Against Five Chinese Researchers
Risks To Great Barrier Reef Could Thwart Tycoon’s Coal Plans
The Super Rich Are Choosing Singapore As The World’s Safest Haven
Jack Dorsey Advocates Ending Police Brutality In Nigeria Through Bitcoin
Psychedelics Replace Pot As The New Favorite Edgy Investment
Who Gets How Much: Big Questions About Reparations For Slavery
US City To Pay Reparations To African-American Community With Tax On Marijuana Sales
Crypto-Friendly Investment Search Engine Vincent Raises $6M
Trading Firm Of Richest Crypto Billionaire Reveals Buying ‘A Lot More’ Bitcoin Below $30K
Bitcoin Security Still A Concern For Some Institutional Investors
Weaponizing Blockchain — Vast Potential, But Projects Are Kept Secret
China Is Pumping Money Out Of The US With Bitcoin
Tennessee City Wants To Accept Property Tax Payments In Bitcoin
Currency Experts Say Cryptonotes, Smart Banknotes And Cryptobanknotes Are In Our Future
Housing Insecurity Is Now A Concern In Addition To Food Insecurity
Food Insecurity Driven By Climate Change Has Central Americans Fleeing To The U.S.
Eco Wave Power Global (“EWPG”) Is A Leading Onshore Wave Energy Technology Company
How And Why To Stimulate Your Vagus Nerve!
Green Finance Isn’t Going Where It’s Needed
Shedding Some Light On The Murky World Of ESG Metrics
SEC Targets Greenwashers To Bring Law And Order To ESG
Spike Lee’s TV Ad For Crypto Touts It As New Money For A Diverse World
Bitcoin Network Node Count Sets New All-Time High
Tesla Needs The Bitcoin Lightning Network For Its Autonomous ‘Robotaxi’ Fleet
How To Buy Bitcoin: A Guide To Investing In The Cryptocurrency
How Crypto is Primed To Transform Movie Financing
Paraguayan Lawmakers To Present Bitcoin Bill On July 14
Bitcoin’s Biggest Hack In History: 184.4 Billion Bitcoin From Thin Air
Paul Sztorc On Measuring Decentralization Of Nodes And Blind Merge Mining
Reality Show Is Casting Crypto Users Locked Out Of Their Wallets
EA, Other Videogame Companies Target Mobile Gaming As Pandemic Wanes
Strike To Offer ‘No Fee’ Bitcoin Trading, Taking Aim At Coinbase And Square
Coinbase Reveals Plans For Crypto App Store Amid Global Refocus
Mexico May Not Be Following El Salvador’s Example On Bitcoin… Yet
What The Crypto Crowd Doesn’t Understand About Economics
My Local Crypto Space Just Got Raided By The Feds. You Know The Feds Scared Of Crypto
My Local Crypto Space Just Got Raided By The Feds. You Know The Feds Scared Of Crypto
Bitcoin Slumps Toward Another ‘Crypto Winter’
NYC’s Mayoral Frontrunner Pledges To Turn City Into Bitcoin Hub
Lyn Alden On Bitcoin, Inflation And The Potential Coming Energy Shock
$71B In Crypto Has Reportedly Passed Through ‘Blockchain Island’ Malta Since 2017
Startups Race Microsoft To Find Better Ways To Cool Data Centers
Why PCs Are Turning Into Giant Phones
Panama To Present Crypto-Related Bill In July
Hawaii Had Largest Increase In Demand For Crypto Out Of US States This Year
What To Expect From Bitcoin As A Legal Tender
Petition: Satoshi Nakamoto Should Receive The Nobel Peace Prize
Can Bitcoin Turbo-Charge The Asset Management Industry?
Bitcoin Interest Drops In China Amid Crackdown On Social Media And Miners
Multi-trillion Asset Manager State Street Launches Digital Currency Division
Wall Street’s Crypto Embrace Shows In Crowd At Miami Conference
MIT Bitcoin Experiment Nets 13,000% Windfall For Students Who Held On
Petition: Let’s Make Bitcoin Legal Tender For United States of America
El Salvador Plans Bill To Adopt Bitcoin As Legal Tender
What Is Dollar Cost Averaging Bitcoin?
Paxful Launches Tool Allowing Businesses To Receive Payment In Bitcoin
CEOs Of Top Russian Banks Sberbank And VTB Blast Bitcoin
President of El Salvador Says He’s Submitting Bill To Make Bitcoin Legal Tender
Bitcoin Falls As Weibo Appears To Suspend Some Crypto Accounts
Israel-Gaza Conflict Spurs Bitcoin Donations To Hamas
Bitcoin Bond Launch Brings Digital Currency Step Closer To ‘World Of High Finance’
Worst Month For BTC Price In 10 Years: 5 Things To Watch In Bitcoin
Bitcoin Card Game Bitopoly Launches
Carbon-Neutral Bitcoin Funds Gain Traction As Investors Seek Greener Crypto
Ultimate Resource On The Bitcoin Mining Council
Libertarian Activists Launch Bitcoin Embassy In New Hampshire
Why The Bitcoin Crash Was A Big Win For Cryptocurrencies
Treasury Calls For Crypto Transfers Over $10,000 To Be Reported To IRS
Crypto Traders Can Automate Legal Requests With New DoNotPay Services
Bitcoin Marches Away From Crypto Pack In Show of Resiliency
NBA Top Shot Lawsuit Says Dapper’s NFTs Need SEC Clampdown
Maximalists At The Movies: Bitcoiners Crowdfunding Anti-FUD Documentary Film
Caitlin Long Reveals The ‘Real Reason’ People Are Selling Crypto
Microsoft Quietly Closing Down Azure Blockchain In September
How Much Energy Does Bitcoin Actually Consume?
Bitcoin Should Be Priced In Sats And How Do We Deliver This Message
Bitcoin Loses 6% In An Hour After Tesla Drops Payments Over Carbon Concerns
Crypto Twitter Decodes Why Zuck Really Named His Goats ‘Max’ And ‘Bitcoin’
Bitcoin Pullback Risk Rises As Whales Resume Selling
Thiel-Backed Block.one Injects Billions In Crypto Exchange
Sequoia, Tiger Global Boost Crypto Bet With Start-up Lender Babel
Here’s How To Tell The Difference Between Bitcoin And Ethereum
In Crypto, Sometimes The Best Thing You Can Do Is Nothing
Crypto Community Remembers Hal Finney’s Contributions To Blockchain On His 65Th Birthday
DJ Khaled ft. Nas, JAY-Z & James Fauntleroy And Harmonies Rap Bitcoin Wealth
The Two Big Themes In The Crypto Market Right Now
Crypto Could Still Be In Its Infancy, Says T. Rowe Price’s CEO
Governing Body Of Louisiana Gives Bitcoin Its Nod Of Approval
Sports Athletes Getting Rich From Bitcoin
Behind Bitcoin’s Recent Slide: Imploding Bets And Forced Liquidations
Bad Omen? US Dollar And Bitcoin Are Both Slumping In A Rare Trend
Wall Street Starts To See Weakness Emerge In Bitcoin Charts
Crypto For The Long Term: What’s The Outlook?
Mix of Old, Wrong And Dubious ‘News/FUD’ Scares Rookie Investors, Fuels Crypto Selloff
Wall Street Pays Attention As Bitcoin Market Cap Nears The Valuation Of Google
Bitcoin Price Drops To $52K, Liquidating Almost $10B In Over-Leveraged Longs
Bitcoin Funding Rates Crash To Lowest Levels In 7 Months, Peak Fear?
Investors’ On-Chain Activity Hints At Bitcoin Price Cycle Top Above $166,000
This Vegan Billionaire Disrupted The Crypto Markets. Now He Wants To Tokenize Stocks
Black Americans Are Embracing Bitcoin To Make Up For Stolen Time
Rap Icon Nas Could Net $100M When Coinbase Lists on Nasdaq
The First Truly Native Cross-Chain DEX Is About To Go Live
Reminiscing On Past ‘Bitcoin Faucet’ Website That Gave Away 19,700 BTC For Free
3X As Many Crypto Figures Make It Onto Forbes 2021 Billionaires List As Last Year
Bubble Or A Drop In The Ocean? Putting Bitcoin’s $1 Trillion Milestone Into Perspective
Pension Funds And Insurance Firms Alive To Bitcoin Investment Proposal
Here’s Why April May Be The Best Month Yet For Bitcoin Price
Blockchain-Based Renewable Energy Marketplaces Gain Traction In 2021
Crypto Firms Got More Funding Last Quarter Than In All of 2020
Government-Backed Bitcoin Hash Wars Will Be The New Space Race
Lars Wood On Enhanced SAT Solvers Based Hashing Method For Bitcoin Mining
Morgan Stanley Adds Bitcoin Exposure To 12 Investment Funds
One BTC Will Be Worth A Lambo By 2022, And A Bugatti By 2023: Kraken CEO
Rocketing Bitcoin Price Provides Refuge For The Brave
Bitcoin Is 3rd Largest World Currency
Does BlockFi’s Risk Justify The Reward?
Crypto Media Runs With The Bulls As New Entrants Compete Against Established Brands
Bitcoin’s Never-Ending Bubble And Other Mysteries
The Last Dip Is The Deepest As Wife Leaves Husband For Buying More Bitcoin
Blockchain.com Raises $300 Million As Investors Find Other Ways Into Bitcoin
What Is BitClout? The Social Media Experiment Sparking Controversy On Twitter
Bitcoin Searches In Turkey Spike 566% After Turkish Lira Drops 14%
Crypto Is Banned In Morocco, But Bitcoin Purchases Are Soaring
Bitcoin Can Be Sent With A Tweet As Bottlepay Twitter App Goes Live
Rise of Crypto Market’s Quiet Giants Has Big Market Implications
Canadian Property Firm Buys Bitcoin In Hopes Of Eventually Scrapping Condo Fees
Bitcoin Price Gets Fed Boost But Bond Yields Could Play Spoilsport: Analysts
Bank of America Claims It Costs Just $93 Million To Move Bitcoin’s Price By 1%
Would A US Wealth Tax Push Millionaires To Bitcoin Adoption?
NYDIG Head Says Major Firms Will Announce Bitcoin ‘Milestones’ Next Week
Signal Encrypted Messenger Now Accepts Donations In Bitcoin
Bitcoin Is Now Worth More Than Visa And Mastercard Combined
Retail Bitcoin Customers Rival Wall Street Buyers As Mania Builds
Crypto’s Rising. So Are The Stakes For Governments Everywhere
Bitcoin Falls After Weekend Rally Pushes Token To Fresh Record
Oakland A’s Major League Baseball Team Now Accepts Bitcoin For Suites
Students In Georgia Set To Be Taught About Crypto At High School
What You Need To Know About Bitcoin Now
Bitcoin Winning Streak Now At 7 Days As Fresh Stimulus Keeps Inflation Bet Alive
Bitcoin Intraday Trading Pattern Emerges As Institutions Pile In
If 60/40 Recipe Sours, Maybe Stir In Some Bitcoin
Explaining Bitcoin’s Speculative Attack On The Dollar
VIX-Like Gauge For Bitcoin Sees Its First-Ever Options Trade
A Utopian Vision Gets A Blockchain Twist In Nevada
Crypto Influencers Scramble To Recover Twitter Accounts After Suspensions
Bitcoin Breaks Through $57,000 As Risk Appetite Revives
Analyzing Bitcoin’s Network Effect
US Government To Sell 0.7501 Bitcoin Worth $38,000 At Current Prices
Pro Traders Avoid Bitcoin Longs While Cautiously Watching DXY Strengthen
Bitcoin Hits Highest Level In Two Weeks As Big-Money Bets Flow
OG Status In Crypto Is A Liability
Bridging The Bitcoin Gender Gap: Crypto Lets Everyone Access Wealth
HODLing Early Leads To Relationship Troubles? Redditors Share Their Stories
Want To Be Rich? Bitcoin’s Limited Supply Cap Means You Only Need 0.01 BTC
You Can Earn 6%, 8%, Even 12% On A Bitcoin ‘Savings Account’—Yeah, Right
Egyptians Are Buying Bitcoin Despite Prohibitive New Banking Laws
Is March Historically A Bad Month For Bitcoin?
Bitcoin Falls 4% As Fed’s Powell Sees ‘Concern’ Over Rising Bond Yields
US Retailers See Millions In Lost Sales Due To Port Congestion, Shortage Of Containers
Pandemic-Relief Aid Boosts Household Income Which Causes Artificial Economic Stimulus
YouTube Suspends CoinDesk’s Channel Over Unspecified Violations
It’s Gates Versus Musk As World’s Richest Spar Over Bitcoin
Charlie Munger Is Sure Bitcoin Will Fail To Become A Global Medium Of Exchange
Bitcoin Is Minting Thousands Of Crypto ‘Diamond Hands’ Millionaires Complete W/Laser Eyes
Dubai’s IBC Group Pledges 100,000 Bitcoin ($4.8 Billion) 20% Of All Bitcoin, Largest So Far
Bitcoin’s Value Is All In The Eye Of The ‘Bithodler’
Bitcoin Is Hitting Record Highs. Why It’s Not Too Late To Dig For Digital Gold
$56.3K Bitcoin Price And $1Trillion Market Cap Signal BTC Is Here To Stay
Christie’s Auction House Will Now Accept Cryptocurrency
Why A Chinese New Year Bitcoin Sell-Off Did Not Happen This Year
The US Federal Reserve Will Adopt Bitcoin As A Reserve Asset
Motley Fool Adding $5M In Bitcoin To Its ‘10X Portfolio’ — Has A $500K Price Target
German Cannabis Company Hedges With Bitcoin In Case Euro Crashes
Bitcoin: What To Know Before Investing
China’s Cryptocurrency Stocks Left Behind In Bitcoin Frenzy
Bitcoin’s Epic Run Is Winning More Attention On Wall Street
Bitcoin Jumps To $50,000 As Record-Breaking Rally Accelerates
Bitcoin’s Volatility Should Burn Investors. It Hasn’t
Bitcoin’s Latest Record Run Is Less Volatile Than The 2017 Boom
Blockchain As A Replacement To The MERS (Mortgage Electronic Registration System)
The Ultimate Resource On “PriFi” Or Private Finance
Deutsche Bank To Offer Bitcoin Custody Services
BeanCoin Currency Casts Lifeline To Closed New Orleans Bars
Bitcoin Could Enter ‘Supercycle’ As Fed Balance Sheet Hits New Record High
Crypto Mogul Bets On ‘Meme Investing’ With Millions In GameStop
Iran’s Central Banks Acquires Bitcoin Even Though Lagarde Says Central Banks Will Not Hold Bitcoin
Bitcoin To Come To America’s Oldest Bank, BNY Mellon
Tesla’s Bitcoin-Equals-Cash View Isn’t Shared By All Crypto Owners
How A Lawsuit Against The IRS Is Trying To Expand Privacy For Crypto Users
Apple Should Launch Own Crypto Exchange, RBC Analyst Says
Bitcoin Hits $43K All-Time High As Tesla Invests $1.5 Billion In BTC
Bitcoin Bounces Off Top of Recent Price Range
Top Fiat Currencies By Market Capitalization VS Bitcoin
Bitcoin Eyes $50K Less Than A Month After BTC Price Broke Its 2017 All-Time High
Investors Piling Into Overvalued Crypto Funds Risk A Painful Exit
Parents Should Be Aware Of Their Children’s Crypto Tax Liabilities
Miami Mayor Says City Employees Should Be Able To Take Their Salaries In Bitcoin
Bitcoiners Get Last Laugh As IBM’s “Blockchain Not Bitcoin” Effort Goes Belly-up
Bitcoin Accounts Offer 3-12% Rates In A Low-Interest World
Analyst Says Bitcoin Price Sell-Off May Occur As Chinese New Year Approaches
Why The Crypto World Needs To Build An Amazon Of Its Own
Tor Project’s Crypto Donations Increased 23% In 2020
Social Trading Platform eToro Ended 2020 With $600M In Revenue
Bitcoin Billionaire Set To Run For California Governor
GameStop Investing Craze ‘Proof of Concept’ For Bitcoin Success
Bitcoin Entrepreneurs Install Mining Rigs In Cars. Will Trucks And Tractor Trailers Be Next?
Harvard, Yale, Brown Endowments Have Been Buying Bitcoin For At Least A Year
Bitcoin Return To $40,000 In Doubt As Flows To Key Fund Slow
Ultimate Resource For Leading Non-Profits Focused On Policy Issues Facing Cryptocurrencies
Regulate Cryptocurrencies? Not Yet
Check Out These Cryptocurrency Clubs And Bitcoin Groups!
Blockchain Brings Unicorns To Millennials
Crypto-Industry Prepares For Onslaught Of Public Listings
Bitcoin Core Lead Maintainer Steps Back, Encourages Decentralization
Here Are Very Bitcoiny Ways To Get Bitcoin Exposure
To Understand Bitcoin, Just Think of It As A Faith-Based Asset
Cryptos Won’t Work As Actual Currencies, UBS Economist Says
Older Investors Are Getting Into Crypto, New Survey Finds
Access Denied: Banks Seem Prone To Cryptophobia Despite Growing Adoption
Pro Traders Buy The Dip As Bulls Address A Trifecta Of FUD News Announcements
Andreas Antonopoulos And Others Debunk Bitcoin Double-Spend FUD
New Bitcoin Investors Explain Why They’re Buying At Record Prices
When Crypto And Traditional Investors Forget Fundamentals, The Market Is Broken
First Hyperledger-based Cryptocurrency Explodes 486% Overnight On Bittrex BTC Listing
Bitcoin Steady As Analysts Say Getting Back To $40,000 Is Key
Coinbase, MEVP Invest In Crypto-Asset Startup Rain
Synthetic Dreams: Wrapped Crypto Assets Gain Traction Amid Surging Market
Secure Bitcoin Self-Custody: Balancing Safety And Ease Of Use
UBS (A Totally Corrupt And Criminal Bank) Warns Clients Crypto Prices Can Actually Go To Zero
Bitcoin Swings Undermine CFO Case For Converting Cash To Crypto
CoinLab Cuts Deal With Mt. Gox Trustee Over Bitcoin Claims
Bitcoin Slides Under $35K Despite Biden Unveiling $1.9 Trillion Stimulus
Bitcoin Refuses To ‘Die’ As BTC Price Hits $40K Just Three Days After Crash
Ex-Ripple CTO Can’t Remember Password To Access $240M In Bitcoin
Financial Advisers Are Betting On Bitcoin As A Hedge
ECB President Christine Lagarde (French Convict) Says, Bitcoin Enables “Funny Business.”
German Police Shut Down Darknet Marketplace That Traded Bitcoin
Bitcoin Miner That’s Risen 1,400% Says More Regulation Is Needed
Bitcoin Rebounds While Leaving Everyone In Dark On True Worth
UK Treasury Calls For Feedback On Approach To Cryptocurrency And Stablecoin Regulation
What Crypto Users Need Know About Changes At The SEC
Where Does This 28% Bitcoin Price Drop Rank In History? Not Even In The Top 5
Seven Times That US Regulators Stepped Into Crypto In 2020
Retail Has Arrived As Paypal Clears $242M In Crypto Sales Nearly Double The Previous Record
Bitcoin’s Slide Dents Price Momentum That Dwarfed Everything
Does Bitcoin Boom Mean ‘Better Gold’ Or Bigger Bubble?
Bitcoin Whales Are Profiting As ‘Weak Hands’ Sell BTC After Price Correction
Crypto User Recovers Long-Lost Private Keys To Access $4M In Bitcoin
The Case For And Against Investing In Bitcoin
Bitcoin’s Wild Weekends Turn Efficient Market Theory Inside Out
Bitcoin Price Briefly Surpasses Market Cap Of Tencent
Broker Touts Exotic Bitcoin Bet To Squeeze Income From Crypto
Broker Touts Exotic Bitcoin Bet To Squeeze Income From Crypto
Tesla’s Crypto-Friendly CEO Is Now The Richest Man In The World
Crypto Market Cap Breaks $1 Trillion Following Jaw-Dropping Rally
Gamblers Could Use Bitcoin At Slot Machines With New Patent
Crypto Users Donate $400K To Julian Assange Defense As Mexico Proposes Asylum
Grayscale Ethereum Trust Fell 22% Despite Rally In Holdings
Bitcoin’s Bulls Should Fear Its Other Scarcity Problem
Ether Follows Bitcoin To Record High Amid Dizzying Crypto Rally
Retail Investors Are Largely Uninvolved As Bitcoin Price Chases $40K
Bitcoin Breaches $34,000 As Rally Extends Into New Year
Social Media Interest In Bitcoin Hits All-Time High
Bitcoin Price Quickly Climbs To $31K, Liquidating $100M Of Shorts
How Massive Bitcoin Buyer Activity On Coinbase Propelled BTC Price Past $32K
FinCEN Wants US Citizens To Disclose Offshore Crypto Holdings of $10K+
Governments Will Start To Hodl Bitcoin In 2021
Crypto-Linked Stocks Extend Rally That Produced 400% Gains
‘Bitcoin Liquidity Crisis’ — BTC Is Becoming Harder To Buy On Exchanges, Data Shows
Bitcoin Looks To Gain Traction In Payments
BTC Market Cap Now Over Half A Trillion Dollars. Major Weekly Candle Closed!!
Elon Musk And Satoshi Nakamoto Making Millionaires At Record Pace
Binance Enables SegWit Support For Bitcoin Deposits As Adoption Grows
Santoshi Nakamoto Delivers $24.5K Christmas Gift With Another New All-Time High
Bitcoin’s Rally Has Already Outlasted 2017’s Epic Run
Gifting Crypto To Loved Ones This Holiday? Educate Them First
Scaramucci’s SkyBridge Files With SEC To Launch Bitcoin Fund
Samsung Integrates Bitcoin Wallets And Exchange Into Galaxy Phones
HTC Smartphone Will Run A Full Bitcoin Node (#GotBitcoin?)
HTC’s New 5G Router Can Host A Full Bitcoin Node
Bitcoin Miners Are Heating Homes Free of Charge
Bitcoin Miners Will Someday Be Incorporated Into Household Appliances
Musk Inquires About Moving ‘Large Transactions’ To Bitcoin
How To Invest In Bitcoin: It Can Be Easy, But Watch Out For Fees
Megan Thee Stallion Gives Away $1 Million In Bitcoin
CoinFLEX Sets Up Short-Term Lending Facility For Crypto Traders
Wall Street Quants Pounce On Crytpo Industry And Some Are Not Sure What To Make Of It
Bitcoin Shortage As Wall Street FOMO Turns BTC Whales Into ‘Plankton’
Bitcoin Tops $22,000 And Strategists Say Rally Has Further To Go
Why Bitcoin Is Overpriced by More Than 50%
Kraken Exchange Will Integrate Bitcoin’s Lightning Network In 2021
New To Bitcoin? Stay Safe And Avoid These Common Scams
Andreas M. Antonopoulos And Simon Dixon Say Don’t Buy Bitcoin!
Famous Former Bitcoin Critics Who Conceded In 2020
Jim Cramer Bought Bitcoin While ‘Off Nicely From The Top’ In $17,000S
The Wealthy Are Jumping Into Bitcoin As Stigma Around Crypto Fades
WordPress Adds Official Ethereum Ad Plugin
France Moves To Ban Anonymous Crypto Accounts To Prevent Money Laundering
10 Predictions For 2021: China, Bitcoin, Taxes, Stablecoins And More
Movie Based On Darknet Market Silk Road Premiering In February
Crypto Funds Have Seen Record Investment Inflow In Recent Weeks
US Gov Is Bitcoin’s Last Remaining Adversary, Says Messari Founder
$1,200 US Stimulus Check Is Now Worth Almost $4,000 If Invested In Bitcoin
German Bank Launches Crypto Fund Covering Portfolio Of Digital Assets
World Governments Agree On Importance Of Crypto Regulation At G-7 Meeting
Why Some Investors Get Bitcoin So Wrong, And What That Says About Its Strengths
It’s Not About Data Ownership, It’s About Data Control, EFF Director Says
‘It Will Send BTC’ — On-Chain Analyst Says Bitcoin Hodlers Are Only Getting Stronger
Bitcoin Arrives On Wall Street: S&P Dow Jones Launching Crypto Indexes In 2021
Audio Streaming Giant Spotify Is Looking Into Crypto Payments
BlackRock (Assets Under Management $7.4 Trillion) CEO: Bitcoin Has Caught Our Attention
Bitcoin Moves $500K Around The Globe Every Second, Says Samson Mow
Pomp Talks Shark Tank’s Kevin O’leary Into Buying ‘A Little More’ Bitcoin
Bitcoin Is The Tulipmania That Refuses To Die
Ultimate Resource On Ethereum 2.0
Biden Should Integrate Bitcoin Into Us Financial System, Says Niall Ferguson
Bitcoin Is Winning The Monetary Revolution
Cash Is Trash, Dump Gold, Buy Bitcoin!
Bitcoin Price Sets New Record High Above $19,783
You Call That A Record? Bitcoin’s November Gains Are 3x Stock Market’s
Bitcoin Fights Back With Power, Speed and Millions of Users
Exchanges Outdo Auctions For Governments Cashing In Criminal Crypto, Says Exec
Coinbase CEO: Trump Administration May ‘Rush Out’ Burdensome Crypto Wallet Rules
Bitcoin Plunges Along With Other Coins Providing For A Major Black Friday Sale Opportunity
The Most Bullish Bitcoin Arguments For Your Thanksgiving Table
‘Bitcoin Tuesday’ To Become One Of The Largest-Ever Crypto Donation Events
World’s First 24/7 Crypto Call-In Station!!!
Bitcoin Trades Again Near Record, Driven By New Group Of Buyers
Friendliest Of Them All? These Could Be The Best Countries For Crypto
Bitcoin Price Doubles Since The Halving, With Just 3.4M Bitcoin Left For Buyers
First Company-Sponsored Bitcoin Retirement Plans Launched In US
Poker Players Are Enhancing Winnings By Cashing Out In Bitcoin
Crypto-Friendly Brooks Gets Nod To Serve 5-Year Term Leading Bank Regulator
The Bitcoin Comeback: Is Crypto Finally Going Mainstream?
The Dark Future Where Payments Are Politicized And Bitcoin Wins
Mexico’s 3rd Richest Man Reveals BTC Holdings As Bitcoin Breaches $18,000
Ultimate Resource On Mike Novogratz And Galaxy Digital’s Bitcoin News
Bitcoin’s Gunning For A Record And No One’s Talking About It
Simple Steps To Keep Your Crypto Safe
US Company Now Lets Travelers Pay For Passports With Bitcoin
Billionaire Hedge Fund Investor Stanley Druckenmiller Says He Owns Bitcoin In CNBC Interview
China’s UnionPay And Korea’s Danal To Launch Crypto-Supporting Digital Card #GotBitcoin
Bitcoin Is Back Trading Near Three-Year Highs
Bitcoin Transaction Fees Rise To 28-Month High As Hashrate Drops Amid Price Rally
Market Is Proving Bitcoin Is ‘Ultimate Safe Haven’ — Anthony Pompliano
3 Reasons Why Bitcoin Price Suddenly Dropping Below $13,000 Isn’t Bearish
Bitcoin Resurgence Leaves Institutional Acceptance Unanswered
WordPress Content Can Now Be Timestamped On Ethereum
PayPal To Offer Crypto Payments Starting In 2021 (A-Z) (#GotBitcoin?)
As Bitcoin Approaches $13,000 It Breaks Correlation With Equities
Crypto M&A Surges Past 2019 Total As Rest of World Eclipses U.S. (#GotBitcoin?)
How HBCUs Are Prepping Black Students For Blockchain Careers
Why Every US Congressman Just Got Sent Some ‘American’ Bitcoin
CME Sounding Out Crypto Traders To Gauge Market Demand For Ether Futures, Options
Caitlin Long On Bitcoin, Blockchain And Rehypothecation (#GotBitcoin?)
Bitcoin Drops To $10,446.83 As CFTC Charges BitMex With Illegally Operating Derivatives Exchange
BitcoinACKs Lets You Track Bitcoin Development And Pay Coders For Their Work
One Of Hal Finney’s Lost Contributions To Bitcoin Core To Be ‘Resurrected’ (#GotBitcoin?)
Cross-chain Money Markets, Latest Attempt To Bring Liquidity To DeFi
Memes Mean Mad Money. Those Silly Defi Memes, They’re Really Important (#GotBitcoin?)
Bennie Overton’s Story About Our Corrupt U.S. Judicial, Global Financial Monetary System And Bitcoin
Stop Fucking Around With Public Token Airdrops In The United States (#GotBitcoin?)
Mad Money’s Jim Cramer Will Invest 1% Of Net Worth In Bitcoin Says, “Gold Is Dangerous”
State-by-state Licensing For Crypto And Payments Firms In The Us Just Got Much Easier (#GotBitcoin?)
Bitcoin (BTC) Ranks As World 6Th Largest Currency
Pomp Claims He Convinced Jim Cramer To Buy Bitcoin
Traditional Investors View Bitcoin As If It Were A Technology Stock
Mastercard Releases Platform Enabling Central Banks To Test Digital Currencies (#GotBitcoin?)
Being Black On Wall Street. Top Black Executives Speak Out About Racism (#GotBitcoin?)
Tesla And Bitcoin Are The Most Popular Assets On TradingView (#GotBitcoin?)
From COVID Generation To Crypto Generation (#GotBitcoin?)
Right-Winger Tucker Carlson Causes Grayscale Investments To Pull Bitcoin Ads
Bitcoin Has Lost Its Way: Here’s How To Return To Crypto’s Subversive Roots
Cross Chain Is Here: NEO, ONT, Cosmos And NEAR Launch Interoperability Protocols (#GotBitcoin?)
Crypto Trading Products Enter The Mainstream With A Number Of Inherent Advantages (#GotBitcoin?)
Crypto Goes Mainstream With TV, Newspaper Ads (#GotBitcoin?)
A Guarded Generation: How Millennials View Money And Investing (#GotBitcoin?)
Blockchain-Backed Social Media Brings More Choice For Users
California Moves Forward With Digital Asset Bill (#GotBitcoin?)
Walmart Adds Crypto Cashback Through Shopping Loyalty Platform StormX (#GotBitcoin?)
Congressman Tom Emmer To Lead First-Ever Crypto Town Hall (#GotBitcoin?)
Why It’s Time To Pay Attention To Mexico’s Booming Crypto Market (#GotBitcoin?)
The Assets That Matter Most In Crypto (#GotBitcoin?)
Ultimate Resource On Non-Fungible Tokens
Bitcoin Community Highlights Double-Standard Applied Deutsche Bank Epstein Scandal
Blockchain Makes Strides In Diversity. However, Traditional Tech Industry Not-S0-Much (#GotBitcoin?)
An Israeli Blockchain Startup Claims It’s Invented An ‘Undo’ Button For BTC Transactions
After Years of Resistance, BitPay Adopts SegWit For Cheaper Bitcoin Transactions
US Appeals Court Allows Warrantless Search of Blockchain, Exchange Data
Central Bank Rate Cuts Mean ‘World Has Gone Zimbabwe’
This Researcher Says Bitcoin’s Elliptic Curve Could Have A Secret Backdoor
China Discovers 4% Of Its Reserves Or 83 Tons Of It’s Gold Bars Are Fake (#GotBitcoin?)
Former Legg Mason Star Bill Miller And Bloomberg Are Optimistic About Bitcoin’s Future
Yield Chasers Are Yield Farming In Crypto-Currencies (#GotBitcoin?)
Australia Post Office Now Lets Customers Buy Bitcoin At Over 3,500 Outlets
Anomaly On Bitcoin Sidechain Results In Brief Security Lapse
SEC And DOJ Charges Lobbying Kingpin Jack Abramoff And Associate For Money Laundering
Veteran Commodities Trader Chris Hehmeyer Goes All In On Crypto (#GotBitcoin?)
Activists Document Police Misconduct Using Decentralized Protocol (#GotBitcoin?)
Supposedly, PayPal, Venmo To Roll Out Crypto Buying And Selling (#GotBitcoin?)
Industry Leaders Launch PayID, The Universal ID For Payments (#GotBitcoin?)
Crypto Quant Fund Debuts With $23M In Assets, $2.3B In Trades (#GotBitcoin?)
The Queens Politician Who Wants To Give New Yorkers Their Own Crypto
Why Does The SEC Want To Run Bitcoin And Ethereum Nodes?
US Drug Agency Failed To Properly Supervise Agent Who Stole $700,000 In Bitcoin In 2015
Layer 2 Will Make Bitcoin As Easy To Use As The Dollar, Says Kraken CEO
Bootstrapping Mobile Mesh Networks With Bitcoin Lightning
Nevermind Coinbase — Big Brother Is Already Watching Your Coins (#GotBitcoin?)
BitPay’s Prepaid Mastercard Launches In US to Make Crypto Accessible (#GotBitcoin?)
Germany’s Deutsche Borse Exchange To List New Bitcoin Exchange-Traded Product
‘Bitcoin Billionaires’ Movie To Tell Winklevoss Bros’ Crypto Story
US Pentagon Created A War Game To Fight The Establishment With BTC (#GotBitcoin?)
JPMorgan Provides Banking Services To Crypto Exchanges Coinbase And Gemini (#GotBitcoin?)
Bitcoin Advocates Cry Foul As US Fed Buying ETFs For The First Time
Final Block Mined Before Halving Contained Reminder of BTC’s Origins (#GotBitcoin?)
Meet Brian Klein, Crypto’s Own ‘High-Stakes’ Trial Attorney (#GotBitcoin?)
3 Reasons For The Bitcoin Price ‘Halving Dump’ From $10K To $8.1K
Bitcoin Outlives And Outlasts Naysayers And First Website That Declared It Dead Back In 2010
Hedge Fund Pioneer Turns Bullish On Bitcoin Amid ‘Unprecedented’ Monetary Inflation
Antonopoulos: Chainalysis Is Helping World’s Worst Dictators & Regimes (#GotBitcoin?)
Survey Shows Many BTC Holders Use Hardware Wallet, Have Backup Keys (#GotBitcoin?)
Iran Ditches The Rial Amid Hyperinflation As Localbitcoins Seem To Trade Near $35K
Buffett ‘Killed His Reputation’ by Being Stupid About BTC, Says Max Keiser (#GotBitcoin?)
Blockfolio Quietly Patches Years-Old Security Hole That Exposed Source Code (#GotBitcoin?)
Bitcoin Won As Store of Value In Coronavirus Crisis — Hedge Fund CEO
Decentralized VPN Gaining Steam At 100,000 Users Worldwide (#GotBitcoin?)
Crypto Exchange Offers Credit Lines so Institutions Can Trade Now, Pay Later (#GotBitcoin?)
Zoom Develops A Cryptocurrency Paywall To Reward Creators Video Conferencing Sessions (#GotBitcoin?)
Bitcoin Startup Purse.io And Major Bitcoin Cash Partner To Shut Down After 6-Year Run
Open Interest In CME Bitcoin Futures Rises 70% As Institutions Return To Market
Square’s Users Can Route Stimulus Payments To BTC-Friendly Cash App
$1.1 Billion BTC Transaction For Only $0.68 Demonstrates Bitcoin’s Advantage Over Banks
Bitcoin Could Become Like ‘Prison Cigarettes’ Amid Deepening Financial Crisis
Bitcoin Holds Value As US Debt Reaches An Unfathomable $24 Trillion
How To Get Money (Crypto-currency) To People In An Emergency, Fast
Bitcoin Miner Manufacturers Mark Down Prices Ahead of Halving
Privacy-Oriented Browsers Gain Traction (#GotBitcoin?)
‘Breakthrough’ As Lightning Uses Web’s Forgotten Payment Code (#GotBitcoin?)
Bitcoin Starts Quarter With Price Down Just 10% YTD vs U.S. Stock’s Worst Quarter Since 2008
Bitcoin Enthusiasts, Liberal Lawmakers Cheer A Fed-Backed Digital Dollar
Crypto-Friendly Bank Revolut Launches In The US (#GotBitcoin?)
The CFTC Just Defined What ‘Actual Delivery’ of Crypto Should Look Like (#GotBitcoin?)
Crypto CEO Compares US Dollar To Onecoin Scam As Fed Keeps Printing (#GotBitcoin?)
Stuck In Quarantine? Become A Blockchain Expert With These Online Courses (#GotBitcoin?)
Bitcoin, Not Governments Will Save the World After Crisis, Tim Draper Says
Crypto Analyst Accused of Photoshopping Trade Screenshots (#GotBitcoin?)
QE4 Begins: Fed Cuts Rates, Buys $700B In Bonds; Bitcoin Rallies 7.7%
Mike Novogratz And Andreas Antonopoulos On The Bitcoin Crash
Amid Market Downturn, Number of People Owning 1 BTC Hits New Record (#GotBitcoin?)
Fatburger And Others Feed $30 Million Into Ethereum For New Bond Offering (#GotBitcoin?)
Pornhub Will Integrate PumaPay Recurring Subscription Crypto Payments (#GotBitcoin?)
Intel SGX Vulnerability Discovered, Cryptocurrency Keys Threatened
Bitcoin’s Plunge Due To Manipulation, Traditional Markets Falling or PlusToken Dumping?
Countries That First Outlawed Crypto But Then Embraced It (#GotBitcoin?)
Bitcoin Maintains Gains As Global Equities Slide, US Yield Hits Record Lows
HTC’s New 5G Router Can Host A Full Bitcoin Node
India Supreme Court Lifts RBI Ban On Banks Servicing Crypto Firms (#GotBitcoin?)
Analyst Claims 98% of Mining Rigs Fail to Verify Transactions (#GotBitcoin?)
Blockchain Storage Offers Security, Data Transparency And immutability. Get Over it!
Black Americans & Crypto (#GotBitcoin?)
Coinbase Wallet Now Allows To Send Crypto Through Usernames (#GotBitcoin)
New ‘Simpsons’ Episode Features Jim Parsons Giving A Crypto Explainer For The Masses (#GotBitcoin?)
Crypto-currency Founder Met With Warren Buffett For Charity Lunch (#GotBitcoin?)
Bitcoin’s Potential To Benefit The African And African-American Community
Coinbase Becomes Direct Visa Card Issuer With Principal Membership
Bitcoin Achieves Major Milestone With Half A Billion Transactions Confirmed
Jill Carlson, Meltem Demirors Back $3.3M Round For Non-Custodial Settlement Protocol Arwen
Crypto Companies Adopt Features Similar To Banks (Only Better) To Drive Growth (#GotBitcoin?)
Top Graphics Cards That Will Turn A Crypto Mining Profit (#GotBitcoin?)
Bitcoin Usage Among Merchants Is Up, According To Data From Coinbase And BitPay
Top 10 Books Recommended by Crypto (#Bitcoin) Thought Leaders
Twitter Adds Bitcoin Emoji, Jack Dorsey Suggests Unicode Does The Same
Bitcoiners Are Now Into Fasting. Read This Article To Find Out Why
You Can Now Donate Bitcoin Or Fiat To Show Your Support For All Of Our Valuable Content
2019’s Top 10 Institutional Actors In Crypto (#GotBitcoin?)
What Does Twitter’s New Decentralized Initiative Mean? (#GotBitcoin?)
Crypto-Friendly Silvergate Bank Goes Public On New York Stock Exchange (#GotBitcoin?)
Bitcoin’s Best Q1 Since 2013 To ‘Escalate’ If $9.5K Is Broken
Billionaire Investor Tim Draper: If You’re a Millennial, Buy Bitcoin
What Are Lightning Wallets Doing To Help Onboard New Users? (#GotBitcoin?)
If You Missed Out On Investing In Amazon, Bitcoin Might Be A Second Chance For You (#GotBitcoin?)
2020 And Beyond: Bitcoin’s Potential Protocol (Privacy And Scalability) Upgrades (#GotBitcoin?)
US Deficit Will Be At Least 6 Times Bitcoin Market Cap — Every Year (#GotBitcoin?)
Central Banks Warm To Issuing Digital Currencies (#GotBitcoin?)
Meet The Crypto Angel Investor Running For Congress In Nevada (#GotBitcoin?)
Introducing BTCPay Vault – Use Any Hardware Wallet With BTCPay And Its Full Node (#GotBitcoin?)
How Not To Lose Your Coins In 2020: Alternative Recovery Methods (#GotBitcoin?)
H.R.5635 – Virtual Currency Tax Fairness Act of 2020 ($200.00 Limit) 116th Congress (2019-2020)
Adam Back On Satoshi Emails, Privacy Concerns And Bitcoin’s Early Days
The Prospect of Using Bitcoin To Build A New International Monetary System Is Getting Real
How To Raise Funds For Australia Wildfire Relief Efforts (Using Bitcoin And/Or Fiat )
Former Regulator Known As ‘Crypto Dad’ To Launch Digital-Dollar Think Tank (#GotBitcoin?)
Currency ‘Cold War’ Takes Center Stage At Pre-Davos Crypto Confab (#GotBitcoin?)
A Blockchain-Secured Home Security Camera Won Innovation Awards At CES 2020 Las Vegas
Bitcoin’s Had A Sensational 11 Years (#GotBitcoin?)
Sergey Nazarov And The Creation Of A Decentralized Network Of Oracles
Google Suspends MetaMask From Its Play App Store, Citing “Deceptive Services”
Christmas Shopping: Where To Buy With Crypto This Festive Season
At 8,990,000% Gains, Bitcoin Dwarfs All Other Investments This Decade
Coinbase CEO Armstrong Wins Patent For Tech Allowing Users To Email Bitcoin
Bitcoin Has Got Society To Think About The Nature Of Money
How DeFi Goes Mainstream In 2020: Focus On Usability (#GotBitcoin?)
Dissidents And Activists Have A Lot To Gain From Bitcoin, If Only They Knew It (#GotBitcoin?)
At A Refugee Camp In Iraq, A 16-Year-Old Syrian Is Teaching Crypto Basics
Bitclub Scheme Busted In The US, Promising High Returns From Mining
Bitcoin Advertised On French National TV
Germany: New Proposed Law Would Legalize Banks Holding Bitcoin
How To Earn And Spend Bitcoin On Black Friday 2019
The Ultimate List of Bitcoin Developments And Accomplishments
Charities Put A Bitcoin Twist On Giving Tuesday
Family Offices Finally Accept The Benefits of Investing In Bitcoin
An Army Of Bitcoin Devs Is Battle-Testing Upgrades To Privacy And Scaling
Bitcoin ‘Carry Trade’ Can Net Annual Gains With Little Risk, Says PlanB
Max Keiser: Bitcoin’s ‘Self-Settlement’ Is A Revolution Against Dollar
Blockchain Can And Will Replace The IRS
China Seizes The Blockchain Opportunity. How Should The US Respond? (#GotBitcoin?)
Jack Dorsey: You Can Buy A Fraction Of Berkshire Stock Or ‘Stack Sats’
Bitcoin Price Skyrockets $500 In Minutes As Bakkt BTC Contracts Hit Highs
Bitcoin’s Irreversibility Challenges International Private Law: Legal Scholar
Bitcoin Has Already Reached 40% Of Average Fiat Currency Lifespan
Yes, Even Bitcoin HODLers Can Lose Money In The Long-Term: Here’s How (#GotBitcoin?)
Unicef To Accept Donations In Bitcoin (#GotBitcoin?)
Former Prosecutor Asked To “Shut Down Bitcoin” And Is Now Face Of Crypto VC Investing (#GotBitcoin?)
Switzerland’s ‘Crypto Valley’ Is Bringing Blockchain To Zurich
Next Bitcoin Halving May Not Lead To Bull Market, Says Bitmain CEO
Bitcoin Developer Amir Taaki, “We Can Crash National Economies” (#GotBitcoin?)
Veteran Crypto And Stocks Trader Shares 6 Ways To Invest And Get Rich
Is Chainlink Blazing A Trail Independent Of Bitcoin?
Nearly $10 Billion In BTC Is Held In Wallets Of 8 Crypto Exchanges (#GotBitcoin?)
SEC Enters Settlement Talks With Alleged Fraudulent Firm Veritaseum (#GotBitcoin?)
Blockstream’s Samson Mow: Bitcoin’s Block Size Already ‘Too Big’
Attorneys Seek Bank Of Ireland Execs’ Testimony Against OneCoin Scammer (#GotBitcoin?)
OpenLibra Plans To Launch Permissionless Fork Of Facebook’s Stablecoin (#GotBitcoin?)
Tiny $217 Options Trade On Bitcoin Blockchain Could Be Wall Street’s Death Knell (#GotBitcoin?)
Class Action Accuses Tether And Bitfinex Of Market Manipulation (#GotBitcoin?)
Sharia Goldbugs: How ISIS Created A Currency For World Domination (#GotBitcoin?)
Bitcoin Eyes Demand As Hong Kong Protestors Announce Bank Run (#GotBitcoin?)
How To Securely Transfer Crypto To Your Heirs
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
Crypto News From The Spanish-Speaking World (#GotBitcoin?)
Financial Services Giant Morningstar To Offer Ratings For Crypto Assets (#GotBitcoin?)
‘Gold-Backed’ Crypto Token Promoter Karatbars Investigated By Florida Regulators (#GotBitcoin?)
The Original Sins Of Cryptocurrencies (#GotBitcoin?)
Bitcoin Is The Fraud? JPMorgan Metals Desk Fixed Gold Prices For Years (#GotBitcoin?)
Israeli Startup That Allows Offline Crypto Transactions Secures $4M (#GotBitcoin?)
[PSA] Non-genuine Trezor One Devices Spotted (#GotBitcoin?)
Bitcoin Stronger Than Ever But No One Seems To Care: Google Trends (#GotBitcoin?)
First-Ever SEC-Qualified Token Offering In US Raises $23 Million (#GotBitcoin?)
You Can Now Prove A Whole Blockchain With One Math Problem – Really
Crypto Mining Supply Fails To Meet Market Demand In Q2: TokenInsight
$2 Billion Lost In Mt. Gox Bitcoin Hack Can Be Recovered, Lawyer Claims (#GotBitcoin?)
Fed Chair Says Agency Monitoring Crypto But Not Developing Its Own (#GotBitcoin?)
Wesley Snipes Is Launching A Tokenized $25 Million Movie Fund (#GotBitcoin?)
Mystery 94K BTC Transaction Becomes Richest Non-Exchange Address (#GotBitcoin?)
A Crypto Fix For A Broken International Monetary System (#GotBitcoin?)
Four Out Of Five Top Bitcoin QR Code Generators Are Scams: Report (#GotBitcoin?)
Waves Platform And The Abyss To Jointly Launch Blockchain-Based Games Marketplace (#GotBitcoin?)
Bitmain Ramps Up Power And Efficiency With New Bitcoin Mining Machine (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Miss Finland: Bitcoin’s Risk Keeps Most Women Away From Cryptocurrency (#GotBitcoin?)
Artist Akon Loves BTC And Says, “It’s Controlled By The People” (#GotBitcoin?)
Ledger Live Now Supports Over 1,250 Ethereum-Based ERC-20 Tokens (#GotBitcoin?)
Co-Founder Of LinkedIn Presents Crypto Rap Video: Hamilton Vs. Satoshi (#GotBitcoin?)
Crypto Insurance Market To Grow, Lloyd’s Of London And Aon To Lead (#GotBitcoin?)
No ‘AltSeason’ Until Bitcoin Breaks $20K, Says Hedge Fund Manager (#GotBitcoin?)
NSA Working To Develop Quantum-Resistant Cryptocurrency: Report (#GotBitcoin?)
Custody Provider Legacy Trust Launches Crypto Pension Plan (#GotBitcoin?)
Vaneck, SolidX To Offer Limited Bitcoin ETF For Institutions Via Exemption (#GotBitcoin?)
Russell Okung: From NFL Superstar To Bitcoin Educator In 2 Years (#GotBitcoin?)
Bitcoin Miners Made $14 Billion To Date Securing The Network (#GotBitcoin?)
Why Does Amazon Want To Hire Blockchain Experts For Its Ads Division?
Argentina’s Economy Is In A Technical Default (#GotBitcoin?)
Blockchain-Based Fractional Ownership Used To Sell High-End Art (#GotBitcoin?)
Portugal Tax Authority: Bitcoin Trading And Payments Are Tax-Free (#GotBitcoin?)
Bitcoin ‘Failed Safe Haven Test’ After 7% Drop, Peter Schiff Gloats (#GotBitcoin?)
Bitcoin Dev Reveals Multisig UI Teaser For Hardware Wallets, Full Nodes (#GotBitcoin?)
Bitcoin Price: $10K Holds For Now As 50% Of CME Futures Set To Expire (#GotBitcoin?)
Bitcoin Realized Market Cap Hits $100 Billion For The First Time (#GotBitcoin?)
Stablecoins Begin To Look Beyond The Dollar (#GotBitcoin?)
Bank Of England Governor: Libra-Like Currency Could Replace US Dollar (#GotBitcoin?)
Binance Reveals ‘Venus’ — Its Own Project To Rival Facebook’s Libra (#GotBitcoin?)
The Real Benefits Of Blockchain Are Here. They’re Being Ignored (#GotBitcoin?)
CommBank Develops Blockchain Market To Boost Biodiversity (#GotBitcoin?)
SEC Approves Blockchain Tech Startup Securitize To Record Stock Transfers (#GotBitcoin?)
SegWit Creator Introduces New Language For Bitcoin Smart Contracts (#GotBitcoin?)
You Can Now Earn Bitcoin Rewards For Postmates Purchases (#GotBitcoin?)
Bitcoin Price ‘Will Struggle’ In Big Financial Crisis, Says Investor (#GotBitcoin?)
Fidelity Charitable Received Over $100M In Crypto Donations Since 2015 (#GotBitcoin?)
Would Blockchain Better Protect User Data Than FaceApp? Experts Answer (#GotBitcoin?)
Just The Existence Of Bitcoin Impacts Monetary Policy (#GotBitcoin?)
What Are The Biggest Alleged Crypto Heists And How Much Was Stolen? (#GotBitcoin?)
IRS To Cryptocurrency Owners: Come Clean, Or Else!
Coinbase Accidentally Saves Unencrypted Passwords Of 3,420 Customers (#GotBitcoin?)
Bitcoin Is A ‘Chaos Hedge, Or Schmuck Insurance‘ (#GotBitcoin?)
Bakkt Announces September 23 Launch Of Futures And Custody
Coinbase CEO: Institutions Depositing $200-400M Into Crypto Per Week (#GotBitcoin?)
Researchers Find Monero Mining Malware That Hides From Task Manager (#GotBitcoin?)
Crypto Dusting Attack Affects Nearly 300,000 Addresses (#GotBitcoin?)
A Case For Bitcoin As Recession Hedge In A Diversified Investment Portfolio (#GotBitcoin?)
SEC Guidance Gives Ammo To Lawsuit Claiming XRP Is Unregistered Security (#GotBitcoin?)
15 Countries To Develop Crypto Transaction Tracking System: Report (#GotBitcoin?)
US Department Of Commerce Offering 6-Figure Salary To Crypto Expert (#GotBitcoin?)
Mastercard Is Building A Team To Develop Crypto, Wallet Projects (#GotBitcoin?)
Canadian Bitcoin Educator Scams The Scammer And Donates Proceeds (#GotBitcoin?)
Amazon Wants To Build A Blockchain For Ads, New Job Listing Shows (#GotBitcoin?)
Shield Bitcoin Wallets From Theft Via Time Delay (#GotBitcoin?)
Blockstream Launches Bitcoin Mining Farm With Fidelity As Early Customer (#GotBitcoin?)
Commerzbank Tests Blockchain Machine To Machine Payments With Daimler (#GotBitcoin?)
Man Takes Bitcoin Miner Seller To Tribunal Over Electricity Bill And Wins (#GotBitcoin?)
Bitcoin’s Computing Power Sets Record As Over 100K New Miners Go Online (#GotBitcoin?)
Walmart Coin And Libra Perform Major Public Relations For Bitcoin (#GotBitcoin?)
Judge Says Buying Bitcoin Via Credit Card Not Necessarily A Cash Advance (#GotBitcoin?)
Poll: If You’re A Stockowner Or Crypto-Currency Holder. What Will You Do When The Recession Comes?
1 In 5 Crypto Holders Are Women, New Report Reveals (#GotBitcoin?)
Beating Bakkt, Ledgerx Is First To Launch ‘Physical’ Bitcoin Futures In Us (#GotBitcoin?)
Facebook Warns Investors That Libra Stablecoin May Never Launch (#GotBitcoin?)
Government Money Printing Is ‘Rocket Fuel’ For Bitcoin (#GotBitcoin?)
Bitcoin-Friendly Square Cash App Stock Price Up 56% In 2019 (#GotBitcoin?)
Safeway Shoppers Can Now Get Bitcoin Back As Change At 894 US Stores (#GotBitcoin?)
TD Ameritrade CEO: There’s ‘Heightened Interest Again’ With Bitcoin (#GotBitcoin?)
Venezuela Sets New Bitcoin Volume Record Thanks To 10,000,000% Inflation (#GotBitcoin?)
Newegg Adds Bitcoin Payment Option To 73 More Countries (#GotBitcoin?)
China’s Schizophrenic Relationship With Bitcoin (#GotBitcoin?)
More Companies Build Products Around Crypto Hardware Wallets (#GotBitcoin?)
Bakkt Is Scheduled To Start Testing Its Bitcoin Futures Contracts Today (#GotBitcoin?)
Bitcoin Network Now 8 Times More Powerful Than It Was At $20K Price (#GotBitcoin?)
Crypto Exchange BitMEX Under Investigation By CFTC: Bloomberg (#GotBitcoin?)
“Bitcoin An ‘Unstoppable Force,” Says US Congressman At Crypto Hearing (#GotBitcoin?)
Bitcoin Network Is Moving $3 Billion Daily, Up 210% Since April (#GotBitcoin?)
Cryptocurrency Startups Get Partial Green Light From Washington
Fundstrat’s Tom Lee: Bitcoin Pullback Is Healthy, Fewer Searches Аre Good (#GotBitcoin?)
Bitcoin Lightning Nodes Are Snatching Funds From Bad Actors (#GotBitcoin?)
The Provident Bank Now Offers Deposit Services For Crypto-Related Entities (#GotBitcoin?)
Bitcoin Could Help Stop News Censorship From Space (#GotBitcoin?)
US Sanctions On Iran Crypto Mining — Inevitable Or Impossible? (#GotBitcoin?)
US Lawmaker Reintroduces ‘Safe Harbor’ Crypto Tax Bill In Congress (#GotBitcoin?)
EU Central Bank Won’t Add Bitcoin To Reserves — Says It’s Not A Currency (#GotBitcoin?)
The Miami Dolphins Now Accept Bitcoin And Litecoin Crypt-Currency Payments (#GotBitcoin?)
Trump Bashes Bitcoin And Alt-Right Is Mad As Hell (#GotBitcoin?)
Goldman Sachs Ramps Up Development Of New Secret Crypto Project (#GotBitcoin?)
Blockchain And AI Bond, Explained (#GotBitcoin?)
Grayscale Bitcoin Trust Outperformed Indexes In First Half Of 2019 (#GotBitcoin?)
XRP Is The Worst Performing Major Crypto Of 2019 (GotBitcoin?)
Bitcoin Back Near $12K As BTC Shorters Lose $44 Million In One Morning (#GotBitcoin?)
As Deutsche Bank Axes 18K Jobs, Bitcoin Offers A ‘Plan ฿”: VanEck Exec (#GotBitcoin?)
Argentina Drives Global LocalBitcoins Volume To Highest Since November (#GotBitcoin?)
‘I Would Buy’ Bitcoin If Growth Continues — Investment Legend Mobius (#GotBitcoin?)
Lawmakers Push For New Bitcoin Rules (#GotBitcoin?)
Facebook’s Libra Is Bad For African Americans (#GotBitcoin?)
Crypto Firm Charity Announces Alliance To Support Feminine Health (#GotBitcoin?)
Canadian Startup Wants To Upgrade Millions Of ATMs To Sell Bitcoin (#GotBitcoin?)
Trump Says US ‘Should Match’ China’s Money Printing Game (#GotBitcoin?)
Casa Launches Lightning Node Mobile App For Bitcoin Newbies (#GotBitcoin?)
Bitcoin Rally Fuels Market In Crypto Derivatives (#GotBitcoin?)
World’s First Zero-Fiat ‘Bitcoin Bond’ Now Available On Bloomberg Terminal (#GotBitcoin?)
Buying Bitcoin Has Been Profitable 98.2% Of The Days Since Creation (#GotBitcoin?)
Another Crypto Exchange Receives License For Crypto Futures
From ‘Ponzi’ To ‘We’re Working On It’ — BIS Chief Reverses Stance On Crypto (#GotBitcoin?)
These Are The Cities Googling ‘Bitcoin’ As Interest Hits 17-Month High (#GotBitcoin?)
Venezuelan Explains How Bitcoin Saves His Family (#GotBitcoin?)
Quantum Computing Vs. Blockchain: Impact On Cryptography
This Fund Is Riding Bitcoin To Top (#GotBitcoin?)
Bitcoin’s Surge Leaves Smaller Digital Currencies In The Dust (#GotBitcoin?)
Bitcoin Exchange Hits $1 Trillion In Trading Volume (#GotBitcoin?)
Bitcoin Breaks $200 Billion Market Cap For The First Time In 17 Months (#GotBitcoin?)
You Can Now Make State Tax Payments In Bitcoin (#GotBitcoin?)
Religious Organizations Make Ideal Places To Mine Bitcoin (#GotBitcoin?)
Goldman Sacs And JP Morgan Chase Finally Concede To Crypto-Currencies (#GotBitcoin?)
Bitcoin Heading For Fifth Month Of Gains Despite Price Correction (#GotBitcoin?)
Breez Reveals Lightning-Powered Bitcoin Payments App For IPhone (#GotBitcoin?)
Big Four Auditing Firm PwC Releases Cryptocurrency Auditing Software (#GotBitcoin?)
Amazon-Owned Twitch Quietly Brings Back Bitcoin Payments (#GotBitcoin?)
JPMorgan Will Pilot ‘JPM Coin’ Stablecoin By End Of 2019: Report (#GotBitcoin?)
Is There A Big Short In Bitcoin? (#GotBitcoin?)
Coinbase Hit With Outage As Bitcoin Price Drops $1.8K In 15 Minutes
Samourai Wallet Releases Privacy-Enhancing CoinJoin Feature (#GotBitcoin?)
There Are Now More Than 5,000 Bitcoin ATMs Around The World (#GotBitcoin?)
You Can Now Get Bitcoin Rewards When Booking At Hotels.Com (#GotBitcoin?)
North America’s Largest Solar Bitcoin Mining Farm Coming To California (#GotBitcoin?)
Bitcoin On Track For Best Second Quarter Price Gain On Record (#GotBitcoin?)
Bitcoin Hash Rate Climbs To New Record High Boosting Network Security (#GotBitcoin?)
Bitcoin Exceeds 1Million Active Addresses While Coinbase Custodies $1.3B In Assets
Why Bitcoin’s Price Suddenly Surged Back $5K (#GotBitcoin?)
Bitcoin’s Lightning Comes To Apple Smartwatches With New App (#GotBitcoin?)
E-Trade To Offer Crypto Trading (#GotBitcoin)
Bitfinex Used Tether Reserves To Mask Missing $850 Million, Probe Finds (#GotBitcoin?)
21-Year-Old Jailed For 10 Years After Stealing $7.5M In Crypto By Hacking Cell Phones (#GotBitcoin?)
You Can Now Shop With Bitcoin On Amazon Using Lightning (#GotBitcoin?)
Afghanistan, Tunisia To Issue Sovereign Bonds In Bitcoin, Bright Future Ahead (#GotBitcoin?)
Crypto Faithful Say Blockchain Can Remake Securities Market Machinery (#GotBitcoin?)
Disney In Talks To Acquire The Owner Of Crypto Exchanges Bitstamp And Korbit (#GotBitcoin?)
Crypto Exchange Gemini Rolls Out Native Wallet Support For SegWit Bitcoin Addresses (#GotBitcoin?)
Binance Delists Bitcoin SV, CEO Calls Craig Wright A ‘Fraud’ (#GotBitcoin?)
Bitcoin Outperforms Nasdaq 100, S&P 500, Grows Whopping 37% In 2019 (#GotBitcoin?)
Bitcoin Passes A Milestone 400 Million Transactions (#GotBitcoin?)
Future Returns: Why Investors May Want To Consider Bitcoin Now (#GotBitcoin?)
Next Bitcoin Core Release To Finally Connect Hardware Wallets To Full Nodes (#GotBitcoin?)
Major Crypto-Currency Exchanges Use Lloyd’s Of London, A Registered Insurance Broker (#GotBitcoin?)
How Bitcoin Can Prevent Fraud And Chargebacks (#GotBitcoin?)
Why Bitcoin’s Price Suddenly Surged Back $5K (#GotBitcoin?)
Zebpay Becomes First Exchange To Add Lightning Payments For All Users (#GotBitcoin?)
Coinbase’s New Customer Incentive: Interest Payments, With A Crypto Twist (#GotBitcoin?)
The Best Bitcoin Debit (Cashback) Cards Of 2019 (#GotBitcoin?)
Real Estate Brokerages Now Accepting Bitcoin (#GotBitcoin?)
Ernst & Young Introduces Tax Tool For Reporting Cryptocurrencies (#GotBitcoin?)
How Will Bitcoin Behave During A Recession? (#GotBitcoin?)
Investors Run Out of Options As Bitcoin, Stocks, Bonds, Oil Cave To Recession Fears (#GotBitcoin?)



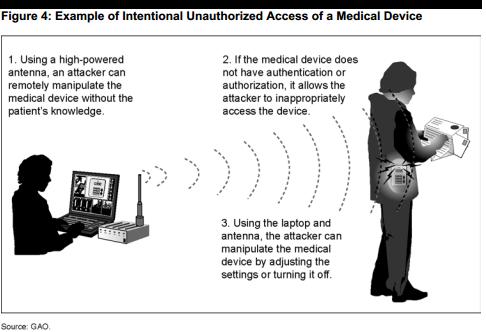


Wonderful site. Lots of useful info here. I’m sending it to several pals ans additionally sharing in delicious. And obviously, thanks to your sweat!
Thanks for the compliments.
We really appreciate that.
Monty